Advanced One-Step Catalytic Synthesis of 2,3-Dihydropyrido[2,3-d]pyrimidin-4-one Derivatives for Commercial Scale-up
Introduction to Next-Generation Heterocyclic Synthesis
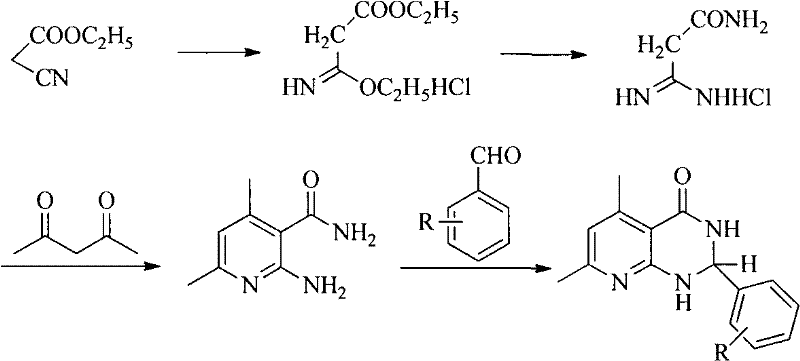
The pharmaceutical and agrochemical industries continuously demand efficient access to complex nitrogen-containing heterocycles, particularly the 2,3-dihydropyrido[2,3-d]pyrimidin-4-(3H)-one scaffold, which serves as a critical backbone for diuretics, antibacterial agents, and CXCR3 antagonists. Patent CN102127076A introduces a transformative methodology that bypasses the historical bottlenecks associated with constructing this fused ring system. By leveraging a direct condensation between readily available 2-amino-3-cyanopyridine derivatives and diverse carbonyl compounds, this technology enables the rapid assembly of high-value intermediates in a single operational step. The strategic advantage lies in the elimination of precarious multi-step sequences, thereby enhancing overall process robustness and atom economy.
This innovative approach utilizes a broad spectrum of catalysts, ranging from inexpensive Lewis acids like anhydrous zinc chloride to simple alkoxides, facilitating reaction conditions that are remarkably mild compared to legacy protocols. The versatility of the substrate scope allows for the incorporation of various substituents (R groups such as halogens, alkyls, or nitro groups) and different carbonyl partners (ketones or aldehydes), providing a modular platform for library synthesis. For procurement and R&D teams, this represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering complex scaffolds with reduced lead times and improved cost structures.
![General reaction scheme showing one-step condensation of 2-amino-3-cyanopyridine with carbonyl compounds to form 2,3-dihydropyrido[2,3-d]pyrimidin-4-one](/insights/img/pyrido-pyrimidinone-synthesis-catalyst-pharma-supplier-20260305051706-04.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrido[2,3-d]pyrimidin-4-one core has been plagued by inefficient synthetic routes that rely on scarce and expensive precursors. Traditional methods often necessitate the preparation of 2-amino-3-formylpyridine, a unstable intermediate that requires a cumbersome three-step synthesis involving harsh conditions and extended reaction times. As illustrated in prior art pathways, such as the conversion of ethyl cyanoacetate through multiple protection and deprotection stages, the cumulative yield suffers significantly at each transformation. Furthermore, alternative routes involving N-(benzyloxycarbonyl)glycyl chloride introduce toxic reagents like hydrobromic acid, creating severe environmental and safety liabilities for large-scale manufacturing facilities.
These conventional processes are not only economically burdensome due to the high cost of starting materials but also operationally risky. The requirement for high temperatures and prolonged heating periods increases energy consumption and the potential for thermal degradation of sensitive functional groups. Additionally, the purification of intermediates in these multi-step sequences often demands extensive column chromatography or repeated recrystallizations, generating substantial solvent waste. For supply chain managers, reliance on such fragile synthetic pathways translates to unpredictable delivery schedules and vulnerability to raw material shortages, highlighting the urgent need for a more streamlined and resilient manufacturing strategy.
The Novel Approach
The methodology disclosed in CN102127076A fundamentally redefines the synthetic landscape by enabling a direct, one-pot cyclization. By reacting 2-amino-3-cyanopyridine directly with ketones or aldehydes in the presence of a catalyst, the formation of the target heterocycle is achieved without isolating unstable intermediates. This telescoped process drastically simplifies the workflow, reducing the number of unit operations and minimizing the exposure of reactive species to potentially degrading conditions. The ability to use liquid carbonyl compounds as both reactants and solvents further enhances the concentration efficiency, leading to higher space-time yields in commercial reactors.
Moreover, the new approach offers exceptional flexibility in catalyst selection, accommodating both acidic and basic conditions depending on the electronic nature of the substrates. This adaptability ensures that even sensitive substrates bearing electron-withdrawing groups can be processed effectively without side reactions. The result is a robust protocol that delivers high-purity products with simplified work-up procedures, typically involving straightforward filtration or extraction. For organizations focused on cost reduction in pharmaceutical intermediates manufacturing, this shift from multi-step linear synthesis to convergent one-step cyclization represents a paradigm shift in process economics and operational simplicity.
Mechanistic Insights into Lewis Acid and Base Catalyzed Cyclization
The core of this synthetic breakthrough relies on the activation of the nitrile group and the carbonyl functionality through cooperative catalysis. Under Lewis acid conditions, such as with anhydrous aluminum trichloride or zinc chloride, the electrophilicity of the carbonyl carbon is enhanced, facilitating nucleophilic attack by the exocyclic amino group of the pyridine derivative. Subsequent intramolecular cyclization involves the attack of the ring nitrogen on the activated nitrile carbon, forming the fused pyrimidine ring. Alternatively, under basic conditions using sodium methoxide or potassium tert-butoxide, the mechanism proceeds via the deprotonation of the alpha-carbon of the ketone, generating an enolate that attacks the nitrile group, followed by amidine formation and ring closure. This dual mechanistic pathway allows chemists to tune the reaction environment to match the specific pKa and steric requirements of the substrates.
Impurity control is inherently superior in this one-step design because the avoidance of isolated intermediates prevents the accumulation of byproducts from previous steps. In traditional multi-step syntheses, impurities from the formation of 2-amino-3-formylpyridine often carry through to the final cyclization, requiring rigorous purification. In contrast, the direct condensation minimizes side reactions such as polymerization or hydrolysis, as the reaction time is significantly shortened (ranging from seconds in microwave conditions to a few hours under reflux). The use of mild temperatures (room temperature to 200°C) further preserves the integrity of sensitive substituents, ensuring a cleaner crude profile that simplifies downstream purification and enhances the overall yield of the high-purity pharmaceutical intermediates.
How to Synthesize 2,3-Dihydropyrido[2,3-d]pyrimidin-4-one Efficiently
To implement this technology effectively, precise control over stoichiometry and reaction parameters is essential. The patent outlines a flexible protocol where the molar ratio of the aminopyridine cyanide to the ketone can vary from 1:1 to 1:200, allowing the carbonyl compound to serve as the solvent in cases where it is liquid at room temperature. Catalyst loading is equally adaptable, ranging from 0.1 to 50 equivalents, providing leeway to optimize for speed versus cost. The reaction can be driven by conventional heating or accelerated via microwave irradiation, offering scalability options from laboratory discovery to pilot plant operations. Detailed standardized synthesis steps for specific derivatives are provided in the guide below to ensure reproducibility and quality consistency.
- Mix 2-amino-3-cyanopyridine derivative and ketone/aldehyde in a molar ratio of 1: 1 to 1:200 with a suitable solvent.
- Add catalyst (e.g., ZnCl2, NaOMe) and heat to 200°C or use microwave promotion until reaction completion.
- Quench reaction, extract with organic solvent, dry, and purify via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound strategic benefits for procurement and supply chain stakeholders, primarily by de-risking the sourcing of critical heterocyclic building blocks. The elimination of complex, multi-step precursor synthesis removes several potential points of failure in the supply chain, such as the availability of specialized reagents like N-(benzyloxycarbonyl)glycyl chloride. By relying on commodity chemicals like acetone, cyclohexanone, and simple pyridine derivatives, manufacturers can secure raw materials from multiple global vendors, ensuring continuity of supply even during market fluctuations. This diversification of the supply base is crucial for maintaining production schedules for downstream API manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this one-step process is driven by the drastic reduction in processing time and solvent usage. By collapsing three or four synthetic steps into a single operation, labor costs, utility consumption, and waste disposal fees are significantly lowered. The absence of expensive transition metal catalysts or toxic reagents further reduces the cost of goods sold (COGS), while the high atom economy ensures that raw material spend is converted efficiently into product mass rather than waste byproducts.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to greater predictability in manufacturing lead times. Since the process tolerates a wide range of temperatures and solvents (including green solvents like ethanol and water mixtures), it is less susceptible to batch failures caused by minor deviations in operating parameters. This reliability allows supply chain planners to optimize inventory levels and reduce the need for excessive safety stock, freeing up working capital and improving overall logistics efficiency.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method aligns with green chemistry principles by minimizing hazardous waste generation. The ability to perform the reaction without high-pressure equipment or cryogenic conditions simplifies the engineering requirements for scale-up, allowing for rapid transition from kilogram to ton-scale production. Furthermore, the simplified work-up procedure reduces the volume of organic solvents required for extraction and purification, lowering the facility's environmental footprint and regulatory compliance burden.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic cyclization technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project requirements. Understanding these nuances is key to making informed decisions about process adoption and vendor qualification.
Q: What are the advantages of this one-step method over traditional multi-step synthesis?
A: This method eliminates the need for pre-synthesizing expensive intermediates like 2-amino-3-formylpyridine, reducing reaction time from days to hours and significantly lowering raw material costs.
Q: Can this process be scaled for industrial production?
A: Yes, the process uses common solvents like ethanol and toluene and operates under mild conditions (RT to 200°C), making it highly adaptable for large-scale reactors without specialized high-pressure equipment.
Q: What types of catalysts are compatible with this reaction?
A: The reaction is versatile and supports Lewis acids (ZnCl2, AlCl3), protonic acids (HCl, H2SO4), and bases (NaOH, NaOMe), allowing flexibility based on substrate sensitivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydropyrido[2,3-d]pyrimidin-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient heterocyclic synthesis plays in accelerating drug development timelines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant with stringent purity specifications. We leverage advanced analytical capabilities in our rigorous QC labs to verify the identity and quality of every batch, guaranteeing that our high-purity pharmaceutical intermediates meet the exacting standards required by global regulatory agencies.
We invite you to collaborate with us to evaluate the feasibility of this one-step catalytic route for your specific pipeline candidates. Our experts are ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined process. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can drive value for your organization.
