Advanced Synthesis of Olmesartan Medoxomil Intermediates: Safety, Purity, and Commercial Scalability
The pharmaceutical landscape for Angiotensin II Receptor Blockers (ARBs) continues to evolve, with Olmesartan Medoxomil remaining a cornerstone therapy for hypertension management. Recent intellectual property developments, specifically patent CN113214215B, disclose a groundbreaking preparation method for key intermediates such as 4,5-Dimethyl-1,3-dioxol-2-one (DMDO) and its derivatives. This technology addresses critical bottlenecks in traditional manufacturing, shifting from hazardous gaseous reagents to safer solid alternatives while integrating advanced purification techniques. For global procurement leaders, this represents a pivotal opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with enhanced supply chain continuity. The innovation lies not merely in chemical transformation but in the holistic redesign of the production workflow to prioritize operator safety, environmental compliance, and commercial viability without sacrificing yield or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DMDO and its chlorinated or hydroxylated derivatives has been plagued by severe safety and efficiency constraints that hinder cost reduction in API manufacturing. Traditional routes frequently rely on gaseous phosgene, a highly toxic substance that necessitates expensive containment infrastructure and rigorous safety protocols, thereby inflating operational expenditures and limiting production capacity scaling. Alternative methods utilizing trichloromethyl chloroformate suffer from reagent instability, leading to inconsistent batch quality and potential safety incidents during storage. Furthermore, chlorination processes employing chlorine gas generate significant acidic waste streams that corrode equipment and require complex neutralization treatments, creating substantial environmental liabilities. These legacy methodologies often result in low atom economy, excessive byproduct formation, and difficult purification challenges, ultimately compromising the economic feasibility of large-scale production for high-purity ARB intermediates.
The Novel Approach
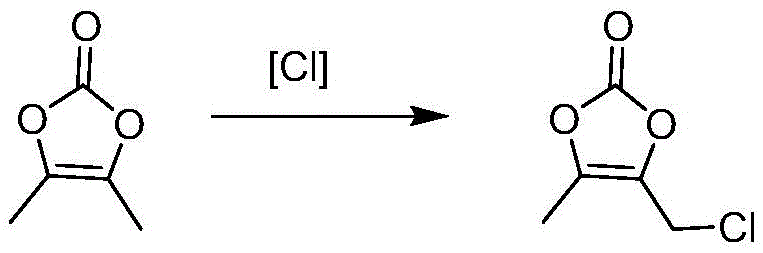
The patented methodology introduces a paradigm shift by substituting gaseous phosgene with solid phosgene (triphosgene), effectively mitigating storage and transportation risks while maintaining high reaction efficiency under mild conditions ranging from 0°C to 40°C. This strategic substitution allows for precise stoichiometric control, typically utilizing 1.0 to 1.1 equivalents, which minimizes waste and enhances the overall sustainability profile of the synthesis. In the subsequent chlorination step, the process employs chlorotriazine under UV irradiation, which not only avoids the use of corrosive chlorine gas but also generates s-triazine as a recoverable byproduct that can be purified and sold as a secondary pharmaceutical intermediate. The integration of molecular distillation technology further distinguishes this approach, enabling the isolation of thermally sensitive products like DMDO-Cl and DMDO-OH without polymerization, ensuring consistent quality suitable for stringent regulatory requirements.
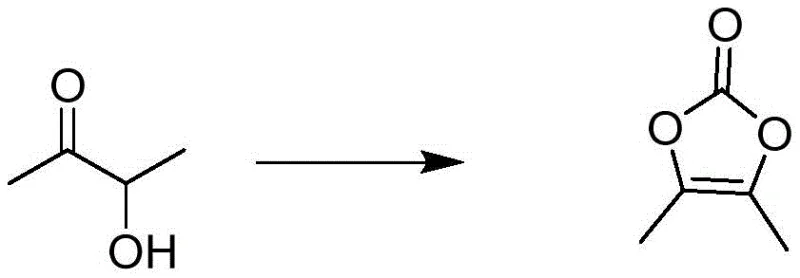
Mechanistic Insights into Solid Phosgene Carbonylation and Radical Functionalization
The core chemical innovation involves a carefully orchestrated carbonylation mechanism where solid phosgene decomposes in situ to release phosgene equivalents in a controlled manner, reacting with 3-hydroxy-2-butanone to form the cyclic carbonate structure of DMDO. This controlled release mechanism prevents the localized exotherms often associated with gaseous reagent addition, thereby reducing the formation of thermal degradation byproducts and ensuring a cleaner reaction profile. The subsequent functionalization steps leverage radical chemistry, specifically utilizing UV light to initiate the homolytic cleavage of chlorotriazine, generating chlorine radicals that selectively attack the methyl group of the DMDO ring. This selectivity is crucial for minimizing ring-opening side reactions, which are common pitfalls in conventional chlorination processes, and ensures that the structural integrity of the dioxolone core is preserved throughout the transformation.
Further mechanistic refinement is observed in the hydroxylation step, where a lithium ferrous phosphate catalyst facilitates a radical combination reaction with hydrogen peroxide. This iron-catalyzed system offers a greener alternative to selenium dioxide oxidation, eliminating the generation of toxic selenium waste and improving the overall environmental footprint of the process. The catalyst promotes the selective abstraction of hydrogen atoms, allowing for the efficient introduction of the hydroxymethyl group while maintaining high stereochemical control. Impurity control is rigorously managed through the use of specific solvent systems, such as mixtures of polar and non-polar solvents during recrystallization, which effectively separate unreacted starting materials and isomeric byproducts. The final application of two-stage molecular distillation operates under high vacuum conditions below 20 Pa, physically separating components based on boiling point differences without thermal stress, resulting in final product purity exceeding 97% with negligible solvent residues.
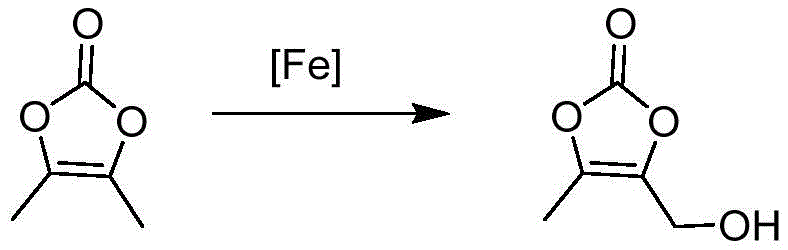
How to Synthesize Olmesartan Medoxomil Intermediate Efficiently
Implementing this advanced synthesis route requires precise adherence to the patented operational parameters to maximize yield and safety across all three stages of production. The process begins with the controlled addition of solid phosgene to a solution of 3-hydroxy-2-butanone in dichloromethane, maintaining temperatures between 15°C and 20°C to optimize reaction kinetics while preventing runaway exotherms. Following the initial carbonylation, the crude DMDO is subjected to a specialized recrystallization protocol using isopropanol and cyclohexane mixtures, which ensures the removal of trace impurities before proceeding to the chlorination stage. The subsequent steps involve strict control of UV irradiation intensity and catalyst loading to drive the functionalization reactions to completion, followed by immediate purification via molecular distillation to lock in product quality. Detailed standardized synthesis steps see the guide below.
- Synthesize DMDO using solid phosgene and 3-hydroxy-2-butanone at 0-40°C, followed by recrystallization in mixed solvents.
- Convert DMDO to DMDO-Cl using chlorotriazine under UV irradiation at 40°C, purifying via two-stage molecular distillation.
- Produce DMDO-OH via lithium ferrous phosphate catalyzed radical reaction with hydrogen peroxide, ensuring high purity through vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented technology offers compelling advantages that directly address the pain points of cost volatility and supply reliability in the pharmaceutical intermediate market. By eliminating the need for specialized gas handling infrastructure required for gaseous phosgene, manufacturers can significantly reduce capital expenditure and ongoing maintenance costs associated with safety compliance. The ability to recover and sell s-triazine as a valuable byproduct creates an additional revenue stream that offsets raw material costs, contributing to substantial cost savings in the overall production economics. Furthermore, the use of stable solid reagents and mild reaction conditions enhances batch-to-batch consistency, reducing the risk of production delays caused by safety incidents or quality failures, thus ensuring a more predictable supply timeline for downstream API manufacturers.
- Cost Reduction in Manufacturing: The transition from hazardous gaseous reagents to stable solid alternatives eliminates the need for expensive containment systems and reduces insurance premiums related to chemical storage. Additionally, the high selectivity of the chlorotriazine-mediated chlorination minimizes raw material waste, while the recovery of s-triazine provides a financial credit that further lowers the net cost of goods sold. The implementation of molecular distillation reduces energy consumption compared to traditional high-temperature distillation methods, contributing to long-term operational efficiency and reduced utility expenses without compromising throughput.
- Enhanced Supply Chain Reliability: Utilizing solid phosgene and commercially available chlorotriazine ensures that raw material sourcing is not dependent on restricted or highly regulated gas supplies, which are often subject to logistical bottlenecks. The robustness of the reaction conditions allows for flexible production scheduling, as the process is less sensitive to minor fluctuations in ambient temperature or pressure compared to gas-phase reactions. This stability translates to reduced lead time for high-purity ARB intermediates, enabling suppliers to respond more agilely to market demand spikes and ensuring continuous availability for critical medication production lines.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex pharmaceutical intermediates by avoiding the engineering challenges associated with handling large volumes of toxic gases. The reduction in acidic waste generation and the elimination of heavy metal catalysts like selenium simplify wastewater treatment protocols, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also aligns with the sustainability goals of major multinational pharmaceutical companies, making the supply chain more resilient and future-proof against evolving green chemistry mandates.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and benefits of this novel synthesis pathway, based on the specific data points and claims outlined in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks or supply partnerships. The answers reflect the practical implications of the chemical innovations on quality, safety, and operational efficiency.
Q: Why is solid phosgene preferred over gaseous phosgene for DMDO synthesis?
A: Solid phosgene (triphosgene) significantly reduces storage and transportation hazards associated with toxic gaseous phosgene, allowing for safer commercial scale-up of complex pharmaceutical intermediates without compromising reaction efficiency.
Q: How does the new chlorination method improve impurity profiles?
A: Using chlorotriazine instead of chlorine gas minimizes equipment corrosion and acidic waste generation. The byproduct, s-triazine, is a valuable pharmaceutical intermediate itself, simplifying waste treatment and enhancing overall atom economy.
Q: What purification technology ensures high purity for these intermediates?
A: The process utilizes two-stage molecular distillation technology, which operates under high vacuum and controlled temperatures to prevent thermal polymerization, consistently achieving product content above 97% with minimal solvent residue.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olmesartan Medoxomil Intermediate Supplier
As the global demand for cardiovascular medications continues to rise, securing a partner with the technical capability to execute complex synthetic routes safely and efficiently is paramount for your supply chain strategy. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. Our facility is equipped with state-of-the-art rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of Olmesartan Medoxomil intermediate meets the exacting standards required for global regulatory filings and patient safety.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing technology can be integrated into your supply base to drive value and security. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements, and let us provide you with specific COA data and route feasibility assessments that demonstrate our commitment to excellence. Together, we can build a resilient supply chain that supports the delivery of life-saving medications to patients worldwide while optimizing your operational bottom line through scientific innovation.
