Scalable Synthesis of Novel C-Ring Fused Abietane Lactones for Oncology Applications
Scalable Synthesis of Novel C-Ring Fused Abietane Lactones for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer improved bioavailability and potency, particularly in the realm of natural product-derived oncology agents. Patent CN111606917B introduces a groundbreaking methodology for constructing abietane compounds featuring a novel C-ring-fused lactone ring skeleton. This innovation addresses long-standing limitations in modifying tanshinone-type diterpenoids, traditionally restricted to A-ring or D-ring functionalization. By shifting the focus to the C-ring o-quinone fragment, this technology effectively enhances the non-planarity of the molecular structure, resulting in derivatives with significantly superior pharmacological profiles, including potent anti-tumor and cardioprotective activities. For procurement and R&D teams, this represents a critical opportunity to access high-value pharmaceutical intermediates that bridge the gap between natural product complexity and synthetic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, structural modifications of abietane diterpenoids, such as tanshinones, have been largely confined to the A and D rings due to the perceived inertness of the C-ring framework. Conventional strategies often involve introducing sulfonic groups to the D-ring to improve water solubility, as seen with tanshinone IIA sodium sulfonate. However, this approach frequently compromises the intrinsic drug efficacy because the resulting ionic compounds struggle to traverse lipid-based biomembranes. Furthermore, modifications on the A-ring are limited by the requirement for an alicyclic structure, restricting the chemical diversity available to medicinal chemists. These traditional pathways often result in derivatives with limited activity improvements or unfavorable pharmacokinetic properties, creating a bottleneck in the development of next-generation therapeutics derived from Salvia miltiorrhiza.
The Novel Approach
The patented technology overcomes these hurdles by targeting the C-ring o-quinone fragment for structural reconstruction. Through a metal-catalyzed condensation with acid anhydrides, the process constructs a fused lactone ring directly onto the C-ring, fundamentally altering the steric and electronic properties of the core skeleton. This novel architecture not only preserves the pharmacophore responsible for biological activity but also optimizes the spatial configuration for better receptor binding. The result is a series of compounds (Types I, II, and III) that demonstrate remarkably enhanced anti-tumor and cardiovascular activities compared to their parent compounds. This strategic shift allows for the creation of high-purity abietane derivatives that maintain potency while offering new avenues for formulation and delivery.
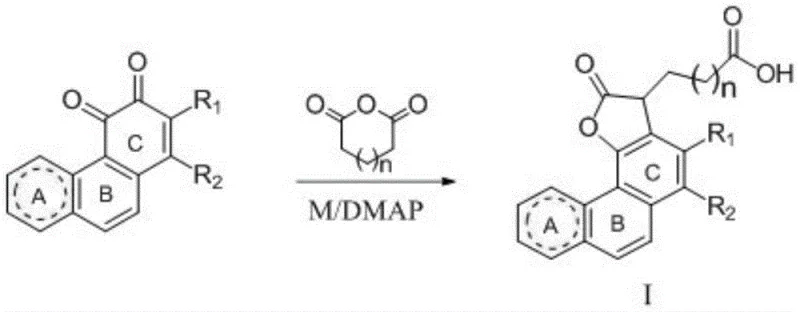
Mechanistic Insights into Zn/DMAP-Catalyzed Reductive Acylation
The core of this synthesis lies in a sophisticated reductive acylation mechanism facilitated by elemental zinc and 4-dimethylaminopyridine (DMAP). In the initial step, the carbonyl group at the 10-position of the tanshinone substrate undergoes reduction by zinc powder, generating a reactive intermediate. Simultaneously, DMAP activates the cyclic acid anhydride, promoting a nucleophilic attack that forms an acylated species. This cascade is meticulously controlled to ensure regioselectivity, preventing unwanted side reactions on the sensitive furan D-ring. The subsequent removal of zinc as zinc hydroxide and the intramolecular condensation of the ester carbonyl with the 11-position carbonyl lead to aromatization and the formation of the stable lactone ring. This mechanism highlights the elegance of using simple reducing metals to drive complex skeletal rearrangements without the need for harsh acidic or basic conditions that could degrade the sensitive diterpene backbone.
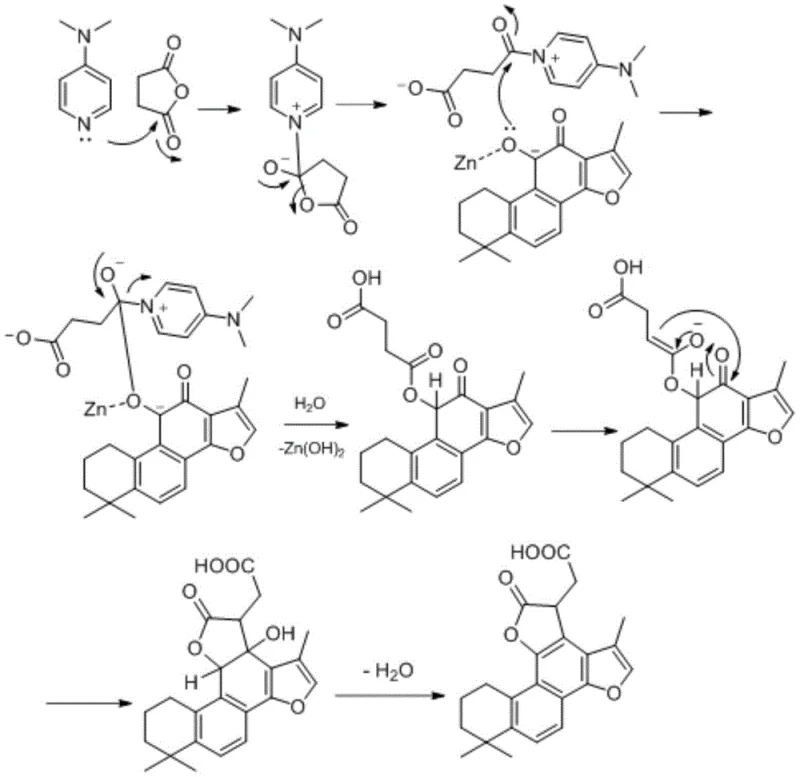
Impurity control is inherently built into this mechanistic pathway. The use of stoichiometric amounts of acid anhydride and the specific redox potential of zinc minimize the formation of over-reduced byproducts or polymerized species. The reaction proceeds through a well-defined transition state where the metal catalyst coordinates with the oxygen atoms, directing the cyclization specifically to the C-ring. This precision ensures that the final product mixture is clean, significantly reducing the burden on downstream purification processes. For manufacturing partners, this translates to a robust process where critical quality attributes can be consistently maintained, ensuring that the commercial scale-up of complex natural product derivatives remains feasible and economically viable.
How to Synthesize C-Ring Fused Abietane Lactones Efficiently
The synthesis protocol outlined in the patent provides a clear, two-stage pathway suitable for industrial adaptation. The first stage involves the condensation of the quinone precursor with an anhydride under reflux, while the second stage optional cyclization allows for further structural diversification. The process utilizes common solvents like tetrahydrofuran and dichloromethane, which are easily recovered and recycled in a GMP environment. Detailed standard operating procedures regarding temperature gradients, addition rates, and quenching protocols are essential to maximize yield and safety.
- Condense abietane diterpene quinones with cyclic acid anhydrides using a metal catalyst (Zn/Fe/Mg) and DMAP in THF under reflux.
- Filter off the metal residue and concentrate the filtrate to isolate the Type I intermediate via recrystallization.
- Convert Type I intermediates into Type II or III lactones using DCC and DMAP in dichloromethane with an organic base.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers substantial advantages by leveraging widely available starting materials and eliminating dependency on scarce resources. The reliance on elemental metals like zinc or iron powder instead of precious metal catalysts (such as palladium or platinum) drastically reduces the raw material cost profile. Furthermore, the workup procedure involves simple filtration to remove metal residues, avoiding complex extraction or chromatography steps typically associated with transition metal removal. This simplicity directly contributes to cost reduction in API manufacturing by shortening cycle times and minimizing waste generation. The ability to produce these high-value intermediates with fewer processing steps enhances overall throughput and operational efficiency.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for specialized scavenging resins and extensive purification protocols, leading to significant operational savings. Additionally, the high atom economy of the condensation reaction ensures that raw materials are efficiently converted into the desired product, minimizing waste disposal costs. The use of recyclable solvents and standard laboratory equipment further lowers the capital expenditure required for setting up production lines, making this route highly attractive for budget-conscious procurement strategies.
- Enhanced Supply Chain Reliability: The primary raw materials, including tanshinone quinones and cyclic anhydrides, are commodity chemicals with stable global supply chains, reducing the risk of shortages. Unlike processes dependent on bespoke reagents or biologically extracted enzymes, this chemical synthesis is resilient to seasonal or biological variations. This stability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to market demands and maintain consistent inventory levels without the fear of supply disruption.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate under standard pressure, facilitating easy translation from bench scale to multi-ton production. The absence of toxic heavy metals simplifies environmental compliance and wastewater treatment, aligning with modern green chemistry principles. The robust nature of the reaction allows for flexible batch sizing, enabling manufacturers to scale production up or down based on clinical trial phases or commercial demand without re-validating the entire process, thus ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is crucial for evaluating the feasibility of integrating these intermediates into your existing drug development pipelines. The answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation.
Q: What is the primary advantage of modifying the C-ring over the A or D rings in tanshinones?
A: Modifying the C-ring o-quinone fragment significantly improves the non-planarity of the molecule, which enhances membrane permeability and biological activity compared to traditional A or D-ring modifications that often compromise efficacy for solubility.
Q: Are the raw materials for this synthesis readily available for large-scale production?
A: Yes, the process utilizes commercially available abietane-type diterpenoid quinones (such as Tanshinone IIA) and common cyclic anhydrides (succinic, glutaric), ensuring a robust and continuous supply chain for commercial manufacturing.
Q: Does this method require expensive transition metal catalysts?
A: No, the protocol employs inexpensive elemental reducing metals like zinc, iron, or magnesium powder, which eliminates the need for costly noble metal catalysts and simplifies the removal of metal residues during workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Abietane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this C-ring fused lactone technology in advancing oncology and cardiovascular therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of abietane intermediate meets the highest international standards. We are committed to supporting your R&D goals with reliable supply and technical expertise.
We invite you to collaborate with us to optimize your supply chain for these next-generation intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
