Advanced Two-Step Synthesis of Polymethoxy Phenylacetic Acid for Pharmaceutical Applications
Advanced Two-Step Synthesis of Polymethoxy Phenylacetic Acid for Pharmaceutical Applications
The pharmaceutical industry continuously demands efficient, safe, and scalable routes for critical organic intermediates, particularly those serving as backbones for antitumor agents and bioactive molecules. Patent CN101274888B introduces a groundbreaking preparation method for dual or poly-methoxy substituted phenylacetic acid, addressing significant safety and cost bottlenecks found in legacy synthetic pathways. This technology leverages a sophisticated two-step sequence starting from readily available methoxylated benzaldehydes, utilizing phase transfer catalysis to drive the initial carbon-carbon bond formation under mild alkaline conditions. By circumventing the use of hazardous cyanide reagents and high-pressure carbonylation equipment, this methodology offers a robust alternative for producing high-value intermediates used in the synthesis of compounds like Pterostilbene and the potent antitumor drug Combretastatin A4 (CA4). The strategic integration of environmentally benign solvents and accessible inorganic salts further underscores the commercial viability of this approach for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of methoxylated phenylacetic acids has relied heavily on methodologies that present substantial operational hazards and environmental liabilities. The traditional cyanide process, for instance, necessitates the use of highly toxic prussiate or sodium cyanide, creating severe risks for operator safety and requiring complex, costly wastewater treatment systems to neutralize cyanide residues before discharge. Similarly, the Grignard reagent method involves reacting benzyl chlorides with magnesium metal followed by carbon dioxide insertion, a procedure that demands strictly anhydrous conditions and often suffers from low yields due to side reactions and the sensitivity of organometallic intermediates. Furthermore, oxo synthesis routes utilizing carbon monoxide require high-pressure reactors and expensive noble metal catalysts that are prone to poisoning and deactivation, thereby inflating capital expenditure and maintenance costs. These conventional pathways are increasingly untenable in a regulatory landscape that prioritizes green chemistry principles and minimizes the use of substances of very high concern (SVHC).
The Novel Approach
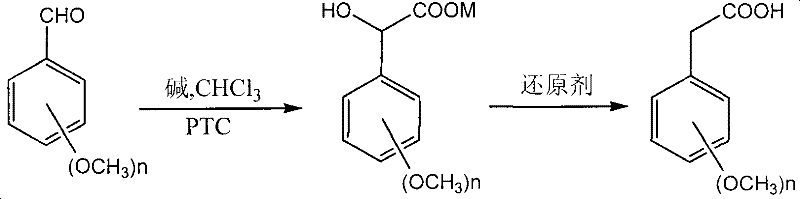
In stark contrast, the innovative route disclosed in the patent employs a phase-transfer catalyzed reaction between methoxylated benzaldehydes and chloroform under alkaline conditions to generate mandelic acid salts as key intermediates. This transformation occurs at moderate temperatures ranging from 30°C to 80°C, eliminating the need for cryogenic conditions or high-pressure vessels typically associated with carbonylation reactions. The subsequent reduction of the mandelic acid derivative to the target phenylacetic acid is achieved using common reducing agents like stannous chloride or sodium pyrosulfite in an acidic medium, ensuring high selectivity and ease of purification. This two-step strategy not only simplifies the operational workflow by avoiding the isolation of unstable intermediates but also utilizes cheap, industrially available raw materials that are easy to source domestically. The overall process design inherently reduces the E-factor by minimizing solvent usage and enabling straightforward aqueous workups, making it an ideal candidate for sustainable manufacturing.
Mechanistic Insights into Phase Transfer Catalyzed Carboxylation and Reduction
The core of this synthetic strategy lies in the efficient generation of dichlorocarbene or its equivalent species in the presence of a phase transfer catalyst (PTC) such as tetrabutylammonium bromide (TBAB). Under strong alkaline conditions provided by sodium or potassium hydroxide, chloroform undergoes deprotonation at the interface of the organic and aqueous phases, facilitated by the quaternary ammonium cation which transports the hydroxide ion into the organic layer. This reactive species then attacks the carbonyl carbon of the dimethoxy or polymethoxy substituted benzaldehyde, forming a trichloromethyl carbinol intermediate which subsequently hydrolyzes to yield the corresponding mandelic acid salt. The choice of PTC is critical; catalysts like cetyltriethylammonium bromide (CTMAB) or beta-cyclodextrin derivatives can significantly enhance the reaction rate and yield by stabilizing the transition state and improving mass transfer between the immiscible phases. This mechanistic pathway avoids the direct use of carbon monoxide or cyanide ions, relying instead on the intrinsic reactivity of the haloform reaction adapted for synthetic utility.
Following the formation of the mandelic acid salt, the second stage involves a reductive dehydroxylation to convert the alpha-hydroxy acid moiety into the methylene group of the phenylacetic acid. This reduction is typically carried out using stannous chloride (SnCl2) in the presence of concentrated hydrochloric acid, which serves both as the acid source and a ligand to activate the tin reagent. The mechanism likely proceeds through the formation of a tin-alkoxide complex followed by hydride transfer or radical-mediated cleavage of the C-O bond, effectively removing the hydroxyl group without affecting the sensitive methoxy substituents on the aromatic ring. Alternative reducing agents such as sodium pyrosulfite or phosphorous acid offer flexibility depending on the specific substrate sensitivity and cost constraints. The reaction conditions are optimized to proceed between 60°C and 120°C, ensuring complete conversion while minimizing the formation of over-reduced byproducts or polymerization of the aromatic system, thus securing a high-purity profile for the final API intermediate.
How to Synthesize Polymethoxy Phenylacetic Acid Efficiently
To implement this synthesis effectively, operators must carefully control the stoichiometry of the base and the phase transfer catalyst to maximize the interfacial reaction rate. The patent details specific molar ratios, recommending an alkali to aldehyde ratio of 1:1 to 1:8, with a preference for a 1:5 ratio to ensure complete conversion without excessive salt formation that could complicate downstream processing. Detailed standardized synthetic steps, including precise temperature ramps and workup procedures for isolating the mandelic acid intermediate, are outlined in the technical guide below to ensure reproducibility across different batch sizes.
- React dimethoxy or polymethoxy substituted benzaldehyde with chloroform under alkaline conditions (NaOH/KOH) and phase transfer catalyst at 30-80°C for 2-12 hours to form mandelic acid salts.
- Treat the isolated mandelic acid salt with a reducing agent (e.g., stannous chloride) under acidic conditions at 60-120°C for 1-6 hours to yield the final phenylacetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers profound advantages by decoupling production from volatile supply chains associated with specialized organometallic reagents or high-pressure gases. The reliance on commodity chemicals such as chloroform, sodium hydroxide, and stannous chloride ensures that raw material costs remain stable and predictable, shielding manufacturing budgets from the price fluctuations often seen with noble metal catalysts or toxic cyanide salts. Furthermore, the elimination of heavy metal catalysts like palladium or rhodium, which are common in carbonylation reactions, removes the need for expensive and technically demanding metal scavenging steps to meet stringent pharmaceutical purity specifications. This simplification of the purification train directly translates to reduced processing time and lower consumption of specialty adsorbents, driving down the overall cost of goods sold (COGS) for the final intermediate.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive, bulk-grade solvents and reagents that are widely available in the global chemical market. By avoiding the use of proprietary ligands or high-pressure equipment, capital expenditure for plant setup is drastically reduced, allowing for faster ROI on production assets. The ability to perform the reaction in aqueous or mixed aqueous-organic systems further lowers solvent recovery costs compared to purely anhydrous processes required for Grignard reactions.
- Enhanced Supply Chain Reliability: Sourcing risk is minimized as the key starting materials, such as veratraldehyde or syringaldehyde derivatives, are produced on a multi-ton scale for the flavor and fragrance industry, ensuring a steady supply even during market disruptions. The robustness of the reaction conditions means that production is less susceptible to minor variations in raw material quality, reducing the frequency of batch failures and ensuring consistent on-time delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The methodology is inherently scalable, having been demonstrated to work efficiently from gram to kilogram scales without loss of yield or selectivity. The use of less hazardous reagents simplifies the permitting process for new manufacturing lines and reduces the burden on waste treatment facilities, aligning perfectly with corporate sustainability goals and increasingly strict environmental regulations regarding toxic effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway, drawing directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is essential for process chemists aiming to adapt this technology for specific analogues or larger production campaigns.
Q: Why is this method preferred over traditional cyanide processes?
A: Traditional cyanide processes utilize highly toxic raw materials like potassium cyanide, posing severe environmental and safety risks. This novel method replaces toxic cyanides with chloroform and benign inorganic bases, significantly improving operator safety and waste treatment protocols while maintaining high product quality.
Q: What are the typical reaction conditions for the reduction step?
A: The reduction of the mandelic acid intermediate is conducted under acidic conditions using agents such as stannous chloride or sodium pyrosulfite. The reaction typically proceeds at temperatures between 60°C and 120°C for 1 to 6 hours, ensuring complete dehydroxylation without requiring high-pressure equipment.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial scalability. It utilizes cheap, domestically available raw materials and simple aqueous workup procedures. The elimination of complex purification steps and the use of standard reactor materials make it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polymethoxy Phenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polymethoxy phenylacetic acid meets the exacting standards required for GMP API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthetic route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring your supply chain is built on a foundation of reliability, quality, and scientific excellence.
