Advanced Purification Technology for High-Purity Camptothecin Derivatives and Commercial Scalability
Advanced Purification Technology for High-Purity Camptothecin Derivatives and Commercial Scalability
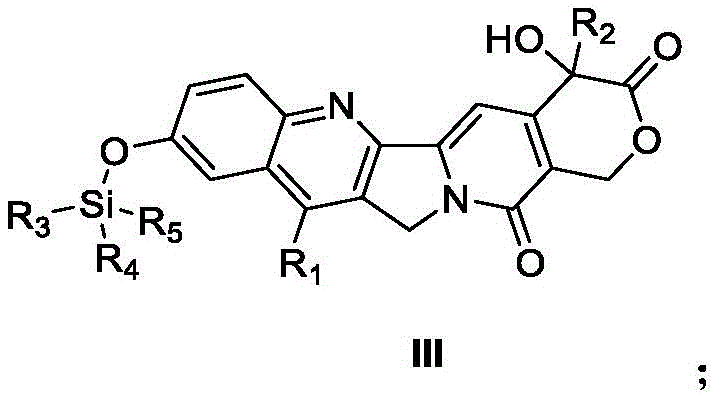
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of critical oncology intermediates, particularly camptothecin derivatives which serve as pivotal precursors for topoisomerase I inhibitors. A groundbreaking approach detailed in patent CN116354976A introduces a sophisticated purification strategy that fundamentally shifts the paradigm from labor-intensive chromatographic techniques to an efficient binary solvent crystallization process. This innovation addresses the longstanding challenge of isolating high-purity compounds, specifically those represented by Formula III, from complex reaction mixtures without compromising yield. By leveraging a precise volumetric relationship between non-polar alkanes and specific ether solvents, manufacturers can now achieve purity levels exceeding 99% while maintaining recovery rates above 95%, a metric that is crucial for cost-effective API intermediate manufacturing. This technical breakthrough not only streamlines the downstream processing workflow but also significantly mitigates the risks associated with product degradation often observed during prolonged exposure to silica gel or extensive solvent evaporation steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of complex heterocyclic compounds like camptothecin derivatives has relied heavily on silica gel column chromatography or preparative reversed-phase chromatography. These traditional methods are fraught with inherent inefficiencies that pose significant bottlenecks for commercial scale-up. The primary drawback lies in the substantial product loss incurred during the adsorption and elution phases, where valuable material remains trapped within the stationary phase or is lost during fraction collection. Furthermore, the operational complexity of column chromatography requires highly skilled personnel and extensive monitoring, leading to prolonged batch cycles and inconsistent throughput. From a supply chain perspective, the massive consumption of organic solvents required for gradient elution creates substantial waste disposal challenges and increases the overall environmental footprint of the manufacturing process. Additionally, the mechanical stress and potential exposure to acidic sites on silica gel can sometimes induce decomposition of sensitive lactone rings, thereby compromising the integrity and pharmacological potency of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to these archaic techniques, the novel purification method disclosed utilizes a strategic binary solvent system designed to exploit subtle differences in solubility profiles between the target compound and its impurities. The core of this innovation involves treating the crude camptothecin derivative with a mixture comprising Component A, selected from alkanes such as petroleum ether, n-heptane, n-pentane, or n-hexane, and Component B, chosen from ethers like diethyl ether, isopropyl ether, or methyl tert-butyl ether. The elegance of this approach lies in its simplicity; by adjusting the volume ratio of Component A to Component B within a specific range of 4:1 to 100:1, preferably between 30:1 and 60:1, the target molecule is induced to precipitate selectively while impurities remain in the supernatant. This solid-liquid separation can be executed via standard filtration equipment, drastically reducing the technical barrier for implementation. The result is a highly pure solid product obtained through a single mixing and filtration step, effectively bypassing the need for complex chromatographic columns and enabling a continuous or semi-continuous manufacturing flow that is far more amenable to industrial automation and quality control standards.
Mechanistic Insights into Binary Solvent Crystallization
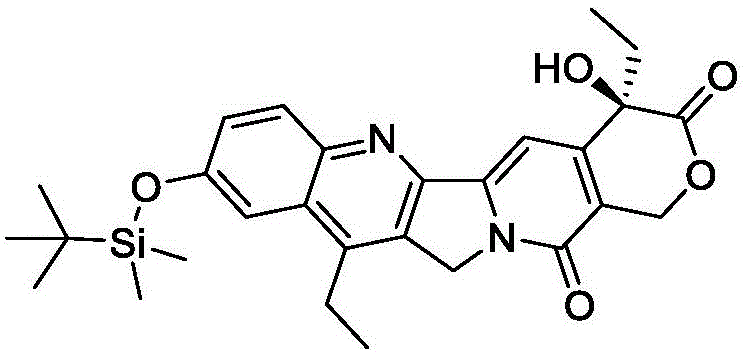
The efficacy of this purification technique is rooted in the thermodynamic principles of solubility modulation within a mixed solvent system. When the crude derivative, often containing residual starting materials, side products, and catalyst residues, is introduced into Component B (the good solvent), it dissolves completely. Subsequent addition of Component A (the anti-solvent) systematically lowers the dielectric constant and solvating power of the medium. For camptothecin derivatives, which possess a rigid pentacyclic ring system with specific polar functionalities like the lactone and hydroxyl groups, the solubility curve is extremely steep with respect to alkane concentration. By meticulously controlling the addition rate and the final ratio, typically optimizing around 50:1 for n-heptane to methyl tert-butyl ether, the system reaches a state of supersaturation exclusively for the target compound. This controlled nucleation ensures that the crystal lattice forms with high selectivity, excluding structurally similar impurities that might otherwise co-crystallize in a less discriminating solvent environment. The presence of the silyl protecting group, as seen in the specific synthesis of Compound I from SN38, further modulates the lipophilicity, making the alkane/ether system particularly effective for this class of molecules.
Furthermore, the mechanism extends to the physical morphology of the resulting solid. Unlike the amorphous powders often obtained from rapid solvent evaporation in chromatography, this crystallization method promotes the formation of well-defined crystals. This crystalline nature is advantageous not only for purity but also for downstream handling, as it improves flowability and drying characteristics. The washing step, utilizing the same binary solvent mixture or pure Component A, serves to displace the mother liquor adhering to the crystal surface, effectively removing soluble impurities without redissolving the product. This surface purification effect is critical for achieving the reported purity levels of greater than 99.5%. The robustness of this mechanism is evidenced by its tolerance to variations in crude quality; even when starting with crude material of approximately 85-90% purity, the process consistently delivers pharmaceutical-grade output. This resilience makes it an ideal candidate for a reliable camptothecin derivative supplier aiming to maintain consistent quality across multiple production batches despite minor fluctuations in upstream synthesis conditions.
How to Synthesize High-Purity Camptothecin Derivatives Efficiently
The implementation of this purification protocol requires precise adherence to solvent ratios and mixing protocols to ensure reproducibility. The process begins with the preparation of the crude material, typically obtained from the silylation of SN38 using agents like t-butyldimethylchlorosilane in dichloromethane. Once the crude solid is isolated, it is subjected to the binary solvent treatment. The operational window is relatively broad, allowing for flexibility in manufacturing, yet specific parameters such as a Component A to Component B ratio of 50:1 have been identified as optimal for maximizing both purity and yield. Detailed standard operating procedures regarding temperature control, stirring speeds, and filtration rates are essential to translate this laboratory success into a commercial reality. For a comprehensive guide on the exact step-by-step execution of this synthesis and purification workflow, please refer to the standardized instructions below.
- Dissolve the crude camptothecin derivative in Component B (e.g., methyl tert-butyl ether) to form a solution.
- Add Component A (e.g., n-heptane or petroleum ether) to the solution under stirring to induce precipitation.
- Perform solid-liquid separation via filtration and dry the filter cake under reduced pressure to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chromatographic purification to this binary solvent crystallization method represents a significant strategic advantage in terms of cost structure and operational reliability. The elimination of silica gel and the drastic reduction in solvent consumption directly correlate to a substantial decrease in raw material costs. Traditional column chromatography consumes vast quantities of eluents, the disposal of which incurs high environmental compliance costs; by replacing this with a recyclable alkane/ether system, the overall cost reduction in API intermediate manufacturing becomes profound. Moreover, the simplification of the unit operation from a multi-step column process to a single mixing and filtration step drastically shortens the batch cycle time. This efficiency gain allows for higher throughput within existing facility footprints, effectively increasing capacity without the need for capital-intensive infrastructure expansion. The reliability of supply is further enhanced by the robustness of the process, which minimizes the risk of batch failures due to column channeling or operator error, ensuring a steady flow of high-quality intermediates to downstream API synthesis lines.
- Cost Reduction in Manufacturing: The removal of expensive chromatography media and the reduction of solvent volume by orders of magnitude leads to direct savings in material costs. Additionally, the simplified workflow reduces labor hours per kilogram of product, contributing to a lower cost of goods sold (COGS). The ability to recover and recycle the alkane and ether solvents further amplifies these economic benefits, creating a closed-loop system that aligns with green chemistry principles and long-term sustainability goals.
- Enhanced Supply Chain Reliability: By decoupling production capacity from the bottleneck of chromatographic purification, manufacturers can respond more agilely to market demand fluctuations. The use of commodity chemicals like n-heptane and methyl tert-butyl ether ensures that raw material availability is never a constraint, unlike specialized chromatography resins which may have long lead times. This stability in the supply chain is critical for maintaining uninterrupted production schedules for life-saving oncology medications, reducing the risk of stockouts and ensuring timely delivery to global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from kilogram to tonne scale without the geometric limitations of column diameter and height. Filtration and drying are well-understood unit operations that can be easily automated in large-scale reactors. Furthermore, the significant reduction in hazardous waste generation simplifies regulatory compliance and lowers the burden on waste treatment facilities. This environmental stewardship not only reduces liability but also enhances the corporate reputation of the manufacturer as a responsible partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel purification technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on how this method compares to industry standards. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this process for their specific camptothecin derivative portfolios. The answers reflect the proven capabilities of the method to deliver high purity and yield consistently.
Q: Why is this purification method superior to traditional silica gel chromatography?
A: Traditional silica gel chromatography often results in significant product loss, complex operations, and difficulty in scaling. This patented binary solvent method achieves purity levels exceeding 99% with recovery rates over 95% through simple filtration, eliminating the need for expensive column packing and large solvent volumes associated with elution.
Q: What represents the optimal solvent ratio for maximum purity?
A: Experimental data indicates that a volume ratio of Component A (alkanes like n-heptane) to Component B (ethers like methyl tert-butyl ether) between 30:1 and 60:1 yields the best results. Specifically, ratios such as 50:1 have demonstrated purity greater than 99.7% with excellent recovery.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is highly scalable. By replacing batch-dependent column chromatography with a straightforward mixing and filtration protocol, the method significantly reduces labor intensity and processing time, making it ideal for commercial production of oncology intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Camptothecin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced purification technologies like the one described in CN116354976A is essential for maintaining competitiveness in the global oncology market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this binary solvent method are fully realized in practical manufacturing environments. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of camptothecin derivative meets the exacting standards required for clinical and commercial applications. We are committed to leveraging our technical expertise to optimize these processes further, ensuring maximum yield and minimal environmental impact for our clients.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this purification technology for their specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current production volumes and purity requirements. We encourage you to reach out to us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive efficiency and quality in your supply chain. Let us help you transform complex chemical challenges into streamlined commercial successes.
