Scalable Synthesis of Beta-Azole-Phenyl Ketone Derivatives for Next-Generation Antifungal Therapeutics
The escalating threat of drug-resistant fungal infections necessitates the continuous development of novel therapeutic agents with improved efficacy and safety profiles. Patent CN109485607B introduces a significant advancement in this field by disclosing a series of beta-azole-phenyl ketone derivatives, represented by General Formula I, which exhibit potent antifungal activity against both superficial and deep-seated fungal pathogens. Unlike traditional azole antifungals that often suffer from narrow spectra or hepatotoxicity, these novel derivatives leverage a unique beta-ketone backbone linked to varied aryl and heteroaryl moieties to enhance target binding affinity. The structural versatility allows for extensive SAR optimization, making them highly valuable candidates for next-generation antifungal drug development. 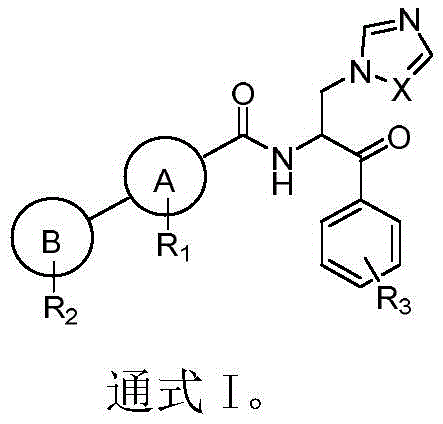
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of azole-based antifungal intermediates frequently relies on harsh alkylation conditions or the use of expensive transition metal catalysts for C-N bond formation, which can introduce heavy metal impurities difficult to remove to ppm levels required for API manufacturing. Furthermore, conventional routes often involve multiple protection-deprotection sequences that significantly reduce overall yield and increase waste generation, posing challenges for green chemistry compliance. The reliance on specialized reagents can also create supply chain bottlenecks, leading to inconsistent availability and inflated costs for pharmaceutical manufacturers seeking reliable sources of high-purity intermediates.
The Novel Approach
The methodology outlined in the patent overcomes these hurdles by employing a modular synthetic strategy centered on robust nucleophilic substitution and amide coupling reactions. By utilizing urotropine as a safe nitrogen source and Boc-protection strategies, the process ensures high chemoselectivity without requiring exotic catalysts. This approach not only simplifies the purification workflow, typically relying on standard crystallization or chromatography, but also enhances the scalability of the process. The ability to introduce diverse substituents at the R1, R2, and R3 positions in the later stages of synthesis allows for rapid library generation, accelerating the lead optimization phase for R&D teams targeting specific fungal CYP51 enzymes.
Mechanistic Insights into the Modular Synthetic Pathway
The core of this synthesis involves a carefully orchestrated sequence beginning with the functionalization of alpha-bromoacetophenone. The initial step utilizes a Delepine-like reaction where urotropine acts as a nucleophile to displace the bromide, followed by acidic hydrolysis to reveal the primary amine. This amine is subsequently protected with di-tert-butyl dicarbonate ((Boc)2O) to prevent side reactions during the critical hydroxymethylation step. The introduction of the hydroxymethyl group via formaldehyde condensation sets the stage for the installation of the azole ring. 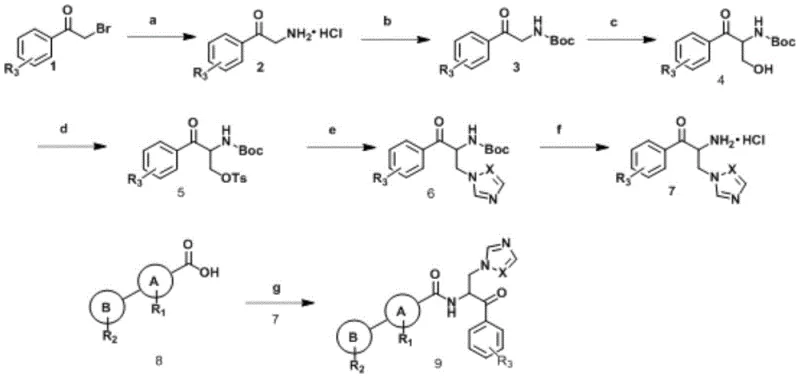
Following tosylation to activate the hydroxyl group, a nucleophilic substitution with imidazole or triazole installs the pharmacophore essential for antifungal activity. The final assembly involves deprotection of the amine and coupling with various substituted benzoic acids using standard peptide coupling reagents like EDCI and HOBt. This mechanistic pathway is designed to minimize racemization and ensure high regioselectivity, crucial for maintaining the biological potency of the final chiral or pseudo-chiral molecules. The use of mild bases like potassium carbonate and solvents like DMF or DCM ensures compatibility with a wide range of functional groups, allowing for the synthesis of complex derivatives without compromising structural integrity.
How to Synthesize Beta-Azole-Phenyl Ketone Derivatives Efficiently
To achieve optimal yields and purity, the synthesis should be conducted under strictly anhydrous conditions during the coupling and substitution steps. The process begins with the preparation of the key amine intermediate, which serves as the convergent point for introducing diversity. Careful control of reaction temperatures, particularly during the exothermic hydroxymethylation and tosylation steps, is essential to prevent byproduct formation. 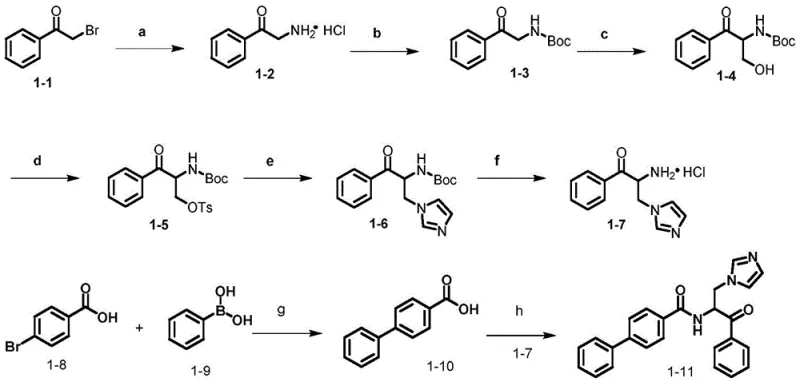
- React alpha-bromoacetophenone with urotropine followed by acidic hydrolysis to generate the amino-ketone intermediate.
- Protect the amine with Boc anhydride, followed by hydroxymethylation using formaldehyde and subsequent tosylation.
- Perform nucleophilic substitution with imidazole or triazole, deprotect the amine, and couple with the desired benzoic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by utilizing commodity chemicals that are readily available in the global market. Starting materials such as alpha-bromoacetophenone, urotropine, and imidazole are produced at massive scales for other industries, ensuring a stable supply chain and mitigating the risk of raw material shortages. The elimination of precious metal catalysts not only reduces the direct cost of goods sold but also simplifies the regulatory filing process by removing the need for extensive heavy metal clearance testing, thereby shortening the time to market for new drug applications.
- Cost Reduction in Manufacturing: The process avoids the use of expensive palladium or rhodium catalysts, relying instead on cost-effective organic reagents and standard coupling agents. This shift significantly lowers the raw material costs and reduces the complexity of waste treatment, as there are no toxic metal residues to manage. The high atom economy of the substitution reactions further contributes to overall cost efficiency, making the commercial production of these intermediates economically viable even at competitive market prices.
- Enhanced Supply Chain Reliability: By depending on widely sourced bulk chemicals rather than specialized custom synthons, manufacturers can secure long-term supply contracts with multiple vendors. This diversification reduces dependency on single-source suppliers and enhances resilience against geopolitical or logistical disruptions. The robustness of the chemical steps means that production can be easily transferred between different manufacturing sites without significant re-validation, ensuring continuous supply for downstream API production.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-ambient temperatures for several key steps, reducing energy consumption associated with heating or cooling large reactors. The solvents used, such as ethanol, ethyl acetate, and dichloromethane, are well-understood in terms of environmental impact and can be efficiently recovered and recycled. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly stringent environmental regulations in major pharmaceutical manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these beta-azole-phenyl ketone derivatives. The answers are derived directly from the experimental data and synthesis protocols detailed in the patent documentation, providing clarity on feasibility and performance.
Q: What is the primary advantage of this synthetic route for antifungal intermediates?
A: The route utilizes mild reaction conditions and readily available starting materials like alpha-bromoacetophenone and urotropine, avoiding expensive transition metal catalysts and enabling easier purification.
Q: Are these derivatives effective against resistant fungal strains?
A: Yes, the patent data indicates strong inhibitory activity against both superficial and deep fungi, including strains that may show resistance to older azole classes, due to the novel beta-ketone structural motif.
Q: Can this process be scaled for commercial API production?
A: Absolutely. The synthesis relies on standard unit operations such as reflux, extraction, and column chromatography, which are easily adaptable from gram-scale laboratory synthesis to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Azole-Phenyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex laboratory pathways into robust commercial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of beta-azole-phenyl ketone intermediate meets the highest quality standards required for pharmaceutical applications.
We invite you to discuss your specific requirements with our technical procurement team to receive a Customized Cost-Saving Analysis tailored to your project volume. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can optimize your supply chain for next-generation antifungal therapies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
