Scalable Manufacturing of Edoxaban Tosylate Intermediates via Novel Cyclization
The global demand for direct oral anticoagulants continues to surge, driven by an aging population and the increasing prevalence of venous thromboembolism. At the heart of this therapeutic class lies Edoxaban Tosylate, a potent Factor Xa inhibitor marketed under the brand name Lixiana®. As depicted in the molecular structure below, the efficacy of this drug relies heavily on the purity and structural integrity of its core heterocyclic scaffold. For pharmaceutical manufacturers and procurement strategists, securing a reliable supply chain for the critical intermediate, 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid, is paramount. Recent intellectual property developments, specifically patent CN114456194A, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in production safety and cost efficiency. This technical insight report analyzes the breakthrough disclosed in CN114456194A, offering a deep dive into how this novel route outperforms legacy technologies while ensuring robust supply continuity for high-purity pharmaceutical intermediates.
![Chemical structure of the key Edoxaban intermediate 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid](/insights/img/edoxaban-intermediate-synthesis-pharma-supplier-20260315054239-02.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of this pivotal thiazolo-pyridine intermediate has been plagued by significant safety hazards and economic inefficiencies. As illustrated in the reaction scheme from US2005119486A1, traditional routes often rely on the use of 4-aminopyridine derivatives which require multiple protection and deprotection steps involving Boc-anhydrides. More critically, these legacy processes frequently employ n-butyllithium, a pyrophoric reagent that poses severe fire risks and requires specialized cryogenic equipment to maintain temperatures well below zero. Furthermore, the use of elemental sulfur in cyclization steps introduces explosion risks and generates difficult-to-handle waste streams. The reliance on methyl iodide, a toxic and volatile alkylating agent, further complicates the environmental compliance profile of these older methods. From a supply chain perspective, the instability of intermediates generated under such harsh conditions often leads to batch-to-batch variability, forcing quality control teams to implement rigorous and costly purification protocols that erode profit margins.
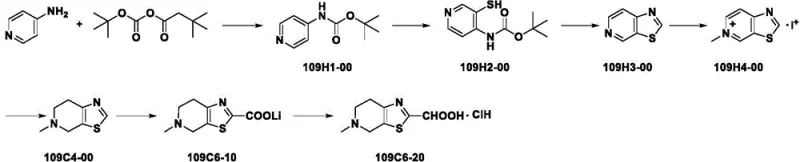
The Novel Approach
In stark contrast, the methodology detailed in CN114456194A represents a paradigm shift towards greener and safer chemical manufacturing. This innovative route initiates with the readily available N-methyl-4-piperidone, bypassing the need for complex nitrogen protection strategies. The core innovation lies in the substitution of hazardous elemental sulfur with sodium sulfide for the thiolation step, effectively mitigating explosion risks while maintaining high reaction selectivity. The process operates under remarkably mild conditions, with key steps proceeding efficiently at ambient temperatures ranging from 20°C to 25°C. This thermal leniency not only reduces energy consumption but also simplifies the engineering requirements for reactor cooling systems. By eliminating the need for pyrophoric organolithium reagents and toxic methylating agents, the new pathway drastically lowers the barrier to entry for safe commercial production. The result is a streamlined synthesis that delivers the target carboxylic acid intermediate with superior stability, facilitating easier storage and transport logistics for global supply chains.
Mechanistic Insights into Fe2(SO4)3-Catalyzed Oxidative Cyclization
The chemical elegance of this new process is best understood through its mechanistic progression, particularly the formation of the thiazole ring and subsequent aromatization. The synthesis begins with a controlled electrophilic substitution where N-methyl-4-piperidone reacts with bromine in an acetic acid medium to yield 3-bromo-N-methyl-4-piperidone. This brominated ketone serves as a highly reactive electrophile for the subsequent nucleophilic attack by sulfide ions derived from sodium sulfide. Unlike the chaotic radical mechanisms often associated with elemental sulfur, this ionic substitution proceeds with high fidelity, generating a stable mercapto-ketone intermediate. The cyclization step involves a condensation reaction between this thiol intermediate and glyoxylic acid in the presence of ammonia. This multicomponent reaction efficiently constructs the fused thiazolo-pyridine skeleton in a single pot, forming the dihydro-intermediate (IV-1). The final aromatization to the fully conjugated system is achieved through an oxidative dehydrogenation step utilizing ferric sulfate (Fe2(SO4)3). This oxidation is crucial as it establishes the aromatic character of the thiazole ring, which is essential for the biological activity of the final API. The use of iron salts as oxidants is particularly advantageous from a regulatory standpoint, as residual iron levels are easier to control and remove compared to heavy metal catalysts like palladium or platinum.
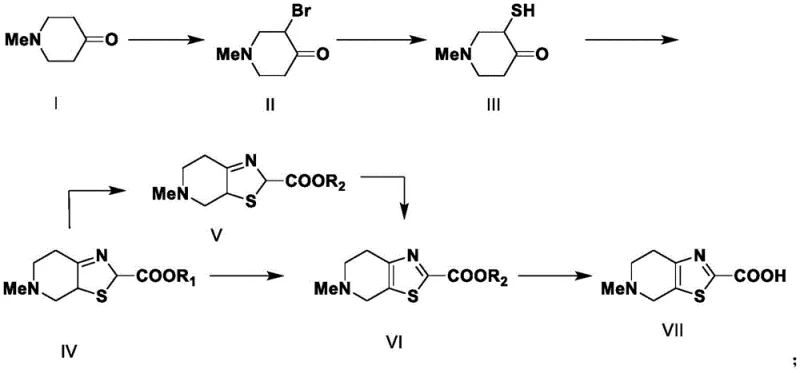
Impurity control is another critical aspect where this mechanism excels. In conventional routes, the use of strong bases like n-butyllithium often leads to over-lithiation or side reactions at the piperidine nitrogen, generating hard-to-remove regioisomers. The acidic to neutral conditions employed in the CN114456194A route minimize these basicity-driven side reactions. Furthermore, the intermediate ammonium salt (IV-1) formed during cyclization exhibits excellent crystallinity, allowing for purification via simple filtration before proceeding to the esterification and oxidation steps. This 'purge point' in the synthesis ensures that impurities do not carry through to the final stages, thereby enhancing the overall purity profile of the final carboxylic acid. The robustness of the ferric sulfate oxidation step also ensures complete conversion of the dihydro-species, preventing the accumulation of partially oxidized byproducts that could complicate downstream coupling reactions in the final API synthesis.
How to Synthesize 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid Efficiently
The implementation of this synthesis requires precise control over reaction parameters to maximize yield and safety. The process is designed to be operationally simple, utilizing standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. The initial bromination must be conducted with careful temperature monitoring to prevent poly-bromination, while the thiolation step benefits from slight cooling to manage the exotherm associated with sulfide addition. The cyclization with glyoxylic acid is a time-dependent process that reaches completion over 18 to 36 hours at room temperature, requiring minimal energy input. Following the formation of the core scaffold, the esterification and oxidation steps can be telescoped or performed sequentially depending on the desired throughput. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Bromination of N-methyl-4-piperidone using bromine in acetic acid to form 3-bromo-N-methyl-4-piperidone.
- Thiolation reaction with sodium sulfide to generate the key thiol intermediate.
- Cyclization with glyoxylic acid and ammonia, followed by esterification and oxidation to yield the final carboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing exotic and hazardous reagents like n-butyllithium and methyl iodide with commodity chemicals such as bromine, sodium sulfide, and glyoxylic acid, manufacturers can significantly reduce their exposure to volatile raw material markets. This shift not only stabilizes input costs but also broadens the supplier base, reducing the risk of single-source dependency. Moreover, the elimination of cryogenic requirements means that production can be executed in standard facilities without the need for specialized low-temperature infrastructure, thereby lowering capital expenditure barriers for contract manufacturing organizations (CMOs). The enhanced stability of the intermediates also translates to reduced waste during storage and transportation, ensuring that more of the manufactured product reaches the final formulation stage.
- Cost Reduction in Manufacturing: The economic impact of removing pyrophoric reagents cannot be overstated. Handling n-butyllithium requires inert atmosphere gloveboxes or specialized dosing systems, which incur high operational and maintenance costs. By switching to aqueous or alcoholic solutions of sodium sulfide and ammonia, the process utilizes standard pumping and dosing equipment. Additionally, the mild reaction temperatures eliminate the need for expensive cryogenic cooling media like liquid nitrogen or specialized chillers, leading to substantial savings in utility costs. The high yield reported in the patent examples suggests a more efficient atom economy, meaning less raw material is wasted as byproduct, further driving down the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and safety of raw materials. Reagents like elemental sulfur and n-butyllithium are often subject to strict transportation regulations due to their hazardous nature, which can cause delays at customs or during domestic transit. The new route relies on non-hazardous or lower-hazard classification chemicals that are easier to ship and store in bulk. This logistical flexibility allows for larger inventory buffers to be held safely on-site, protecting against upstream supply disruptions. Furthermore, the robustness of the synthesis reduces the likelihood of batch failures due to sensitive reaction conditions, ensuring a consistent and predictable output schedule for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology is inherently designed for scale. The absence of explosive sulfur dust and flammable organolithiums removes major process safety management (PSM) hurdles, facilitating faster regulatory approvals for new production lines. From an environmental perspective, the process generates less hazardous waste; the iron-based oxidant produces iron oxide sludge which is easier to treat than heavy metal catalyst waste. The use of water and alcohols as primary solvents aligns with green chemistry principles, reducing the volume of volatile organic compounds (VOCs) emitted. This cleaner profile simplifies wastewater treatment and helps manufacturers meet increasingly stringent environmental, social, and governance (ESG) targets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new intermediate is crucial for stakeholders evaluating its integration into their supply chain. The following questions address common concerns regarding process safety, scalability, and quality assurance based on the data provided in the patent literature. These insights are intended to assist technical teams in making informed decisions about vendor qualification and process adoption.
Q: Why is the new synthesis route safer than conventional methods?
A: The novel route eliminates the use of highly flammable n-butyllithium and explosive elemental sulfur, replacing them with stable sodium sulfide and mild bromination conditions, significantly reducing industrial safety hazards.
Q: What are the cost advantages of this manufacturing process?
A: By avoiding expensive pyrophoric reagents and complex low-temperature cryogenic steps, the process utilizes readily available raw materials and standard reactor equipment, leading to substantial operational cost reductions.
Q: Is this intermediate suitable for large-scale API production?
A: Yes, the process features high stability of intermediates, mild reaction temperatures (20-25°C), and simplified workup procedures, making it highly amenable to commercial scale-up for anticoagulant manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,5,6,7-tetrahydro-5-methyl-thiazolo[5,4-c]pyridine-2-carboxylic acid Supplier
As the pharmaceutical industry moves towards more sustainable and cost-effective manufacturing paradigms, the ability to execute complex heterocyclic syntheses safely is a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced process chemistry to deliver high-value intermediates like the Edoxaban precursor. Our technical team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for global regulatory filings. Our commitment to quality ensures that the impurity profiles of our intermediates are fully characterized and controlled, providing peace of mind for your API synthesis.
We invite procurement leaders and R&D directors to explore how our optimized manufacturing capabilities can enhance your supply chain resilience. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure a stable, high-quality supply of critical anticoagulant intermediates that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
