Advanced Manufacturing of Dopamine-Beta-Hydroxylase Inhibitors for Hypertension Therapy
Introduction to Novel Dopamine-Beta-Hydroxylase Inhibitors
The landscape of antihypertensive pharmacology has long sought effective agents capable of modulating catecholamine biosynthesis without the severe side effects associated with early-generation inhibitors. Patent CN87103808A introduces a significant breakthrough in this domain through the discovery of substituted 1-aralkyl-1,2,4-triazole-5-thiols and their alkylthio analogs. These compounds function as high-efficiency inhibitors of dopamine-beta-hydroxylase (DBH), the enzyme responsible for converting dopamine into norepinephrine. By selectively inhibiting this enzymatic step, the technology offers a dual therapeutic benefit: lowering blood pressure through reduced norepinephrine levels while simultaneously increasing dopamine concentrations, which can provide selective vasodilatory effects. The core structural innovation lies in the specific substitution patterns on the triazole ring, which optimize binding affinity and metabolic stability compared to prior art such as fusaric acid derivatives.
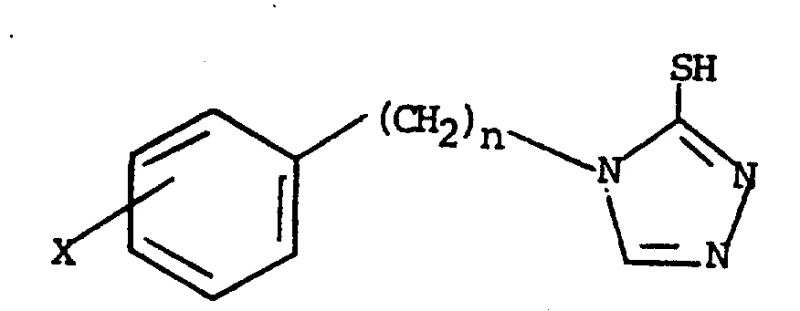
The structural versatility of this chemical class is defined by the variable aralkyl chain length (n=0-5) and the diverse substituents (X) allowable on the aromatic ring, ranging from halogens to electron-withdrawing groups like nitro and cyano. This flexibility allows medicinal chemists to fine-tune the physicochemical properties of the final active pharmaceutical ingredient (API) to meet specific bioavailability targets. For procurement specialists and supply chain managers, understanding the robustness of the synthetic pathway behind these structures is critical, as it directly impacts the reliability of sourcing these high-value pharmaceutical intermediates. The patent details a scalable, solution-phase synthesis that avoids the use of exotic reagents, relying instead on commodity chemicals like substituted benzaldehydes and tert-butyl carbazinate, thereby ensuring a stable supply chain foundation for commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of DBH inhibitors was plagued by compounds that lacked specificity or exhibited unacceptable toxicity profiles. Early generations of inhibitors, such as 5-alkylpicolinic acids and fusaric acid derivatives, operated primarily through non-specific metal chelation mechanisms. While effective at inhibiting the copper-dependent DBH enzyme, these agents often chelated essential metals in other biological systems, leading to significant hepatotoxicity and other systemic side effects that precluded their clinical utility. Furthermore, phenethylamine analogs, another class of known inhibitors, often suffered from poor metabolic stability or required complex multi-step syntheses involving hazardous reagents that were difficult to scale safely. The reliance on these older scaffolds created a bottleneck in the development of safe antihypertensive therapies, necessitating a search for new chemical entities that could offer potent inhibition without the liability of heavy metal interaction or liver damage.
The Novel Approach
The methodology described in CN87103808A represents a paradigm shift by utilizing a 1,2,4-triazole-5-thiol scaffold that inhibits DBH through a mechanism distinct from simple metal chelation. This novel approach leverages the specific electronic properties of the thiol group positioned on the triazole ring to interact with the enzyme's active site more selectively. The synthetic route is designed to be modular, allowing for the easy introduction of various substituents (X) on the benzene ring to modulate potency; for instance, the 3'-fluoro derivative demonstrated an IC50 of 1.4×10^-5 M, significantly outperforming the unsubstituted analog. From a manufacturing perspective, this approach eliminates the need for complex fermentation processes or the isolation of natural products, which are often subject to batch-to-batch variability. Instead, it employs a fully synthetic organic chemistry route that is amenable to strict quality control, ensuring consistent purity and impurity profiles essential for regulatory approval of high-purity pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Triazole Cyclization
The core of this manufacturing process is the formation of the 1,2,4-triazole ring, a heterocyclic system known for its thermal stability and resistance to metabolic degradation. The mechanism proceeds through a thiosemicarbazide intermediate, which undergoes intramolecular cyclodehydration in the presence of a strong acid. A critical aspect of this chemistry is the tautomeric equilibrium of the final product. As illustrated in the patent data, the compound exists in equilibrium between the thione and thiol forms. This tautomerism is not merely a structural curiosity but has profound implications for the compound's reactivity and solubility during downstream processing. Understanding this equilibrium is vital for R&D teams aiming to optimize crystallization conditions, as the dominant tautomer in the solid state may differ from that in solution, affecting the final polymorphic form of the API.

Impurity control is managed strategically through the use of tert-butoxycarbonyl (Boc) protecting groups during the early stages of the synthesis. The initial condensation of the benzaldehyde with tert-butyl carbazinate forms a protected hydrazone, which prevents premature oligomerization or side reactions at the hydrazine nitrogen. This protection strategy is maintained through the hydrogenation and isothiocyanate coupling steps, ensuring that the reactive amine functionality is only revealed at the final cyclization stage. By delaying the exposure of the free hydrazine until the thiosemicarbazide is fully formed, the process minimizes the formation of urea byproducts and symmetrical hydrazine dimers. This level of mechanistic control is essential for achieving the stringent purity specifications required for commercial scale-up of complex pharmaceutical intermediates, reducing the burden on downstream purification units.
How to Synthesize 1-Aralkyl-1,2,4-Triazole-5-Thiols Efficiently
The synthesis of these potent DBH inhibitors follows a logical four-step sequence that balances yield with operational simplicity. The process begins with the condensation of a substituted benzaldehyde and tert-butyl carbazinate, followed by catalytic hydrogenation to reduce the C=N bond. The resulting hydrazine is then coupled with tert-butyl isothiocyanate to install the sulfur-containing side chain. The final and most critical step is the acid-mediated cyclization, which simultaneously closes the triazole ring and removes the acid-labile protecting groups. This telescoped deprotection-cyclization strategy reduces the total number of isolation steps, thereby improving overall throughput and reducing solvent consumption. Detailed standard operating procedures for each unit operation, including specific temperature ramps and quench protocols, are essential for maintaining safety and consistency at scale.
- Condensation of substituted benzaldehydes with tert-butyl carbazinate in hexane to form protected hydrazones.
- Catalytic hydrogenation using 10% Pd/C in methanol to reduce hydrazones to hydrazines.
- Reaction with tert-butyl isothiocyanate in ethyl acetate to generate thiosemicarbazide intermediates.
- Acid-catalyzed cyclization using formic acid to close the triazole ring and remove protecting groups.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from legacy DBH inhibitor technologies to this novel triazole-based synthesis offers substantial strategic advantages. The primary driver for cost optimization lies in the raw material selection; the process utilizes substituted benzaldehydes, which are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the synthetic route avoids the use of expensive transition metal catalysts beyond standard palladium on carbon, which can be recovered and recycled, significantly lowering the cost of goods sold (COGS). The elimination of fermentation-based steps also removes the variability associated with biological processes, ensuring a more predictable production schedule and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to maximize atom economy and minimize waste generation. By employing a convergent synthesis strategy where the aromatic and heterocyclic components are assembled late in the sequence, the process avoids the loss of valuable chiral or functionalized intermediates in early failed steps. The use of common solvents like hexane, ethyl acetate, and methanol facilitates solvent recovery and recycling, further driving down operational expenses. Additionally, the final cyclization step uses formic acid, a relatively inexpensive reagent compared to specialized Lewis acids or coupling agents, contributing to a leaner manufacturing cost structure without compromising yield.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent supply continuity. Unlike natural product extraction which is subject to seasonal and agricultural variables, this fully synthetic approach can be executed year-round in standard chemical reactors. The intermediates, such as the protected hydrazones and thiosemicarbazides, are stable solids that can be stockpiled if necessary, providing a buffer against demand fluctuations. This stability is crucial for maintaining the supply of reliable pharmaceutical intermediate supplier networks, ensuring that downstream formulation partners receive materials on time and within specification, regardless of external market pressures.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram-scale laboratory synthesis to multi-kilogram pilot batches. The unit operations involved—reflux, filtration, and crystallization—are standard in the fine chemical industry, meaning existing infrastructure can be utilized without major capital expenditure. From an environmental standpoint, the avoidance of heavy metal chelators reduces the toxic load of the effluent, simplifying wastewater treatment requirements. The ability to recycle palladium catalysts and recover organic solvents aligns with modern green chemistry principles, facilitating easier regulatory compliance and enhancing the sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these DBH inhibitors. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding the nuances of the synthesis and the biological activity of the final compounds is essential for partners looking to integrate these molecules into their development pipelines. The answers below reflect the current state of the art as described in the intellectual property, highlighting both the potential and the specific processing requirements of this chemical class.
Q: What is the primary therapeutic application of these triazole thiol compounds?
A: These compounds act as potent inhibitors of dopamine-beta-hydroxylase (DBH), making them valuable candidates for treating hypertension, gastric ulcers, and conditions requiring reduced norepinephrine levels.
Q: How does this synthesis route improve upon traditional DBH inhibitors?
A: Unlike earlier metal chelators or fusaric acid derivatives which exhibited liver toxicity, this novel triazole-thiol scaffold offers specific enzyme inhibition with a potentially safer toxicological profile.
Q: What are the critical process parameters for the cyclization step?
A: The cyclization requires strong acidic conditions, preferably using 98% formic acid under reflux for approximately 4 hours, to ensure complete ring closure and deprotection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Aralkyl-1,2,4-Triazole-5-Thiol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of novel antihypertensive therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to full-scale market availability. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that every gram of 1-aralkyl-1,2,4-triazole-5-thiol we produce meets the exacting standards required for pharmaceutical applications, minimizing the risk of delays in your regulatory filings.
We invite you to engage with our technical procurement team to discuss your specific requirements for cost reduction in pharmaceutical intermediates manufacturing. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogs, we are prepared to provide the support you need. Contact us today to explore how our manufacturing expertise can accelerate your development timeline and secure a reliable supply of these vital dopamine-beta-hydroxylase inhibitors.
