Advanced Synthesis of Quinolinyl-Benzylsulfonylureas for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Quinolinyl-Benzylsulfonylureas for High-Purity Pharmaceutical Intermediates
The pharmaceutical landscape continuously demands novel scaffolds capable of modulating specific enzymatic pathways with high selectivity and potency. Patent CN1049656A introduces a significant advancement in this domain by disclosing a series of substituted N-(quinolin-2-yl-methoxy)benzylsulfonylureas, which exhibit remarkable efficacy as 5-lipoxygenase inhibitors. These compounds represent a critical class of pharmaceutical intermediates designed to intervene in the arachidonic acid metabolism cascade, offering potential therapeutic benefits for respiratory conditions such as allergic asthma and bronchitis. The structural versatility of this chemical series allows for extensive optimization of pharmacokinetic properties, making it a highly attractive target for process research and development teams aiming to secure a reliable pharmaceutical intermediate supplier for next-generation anti-inflammatory drugs.
The core innovation lies in the specific substitution patterns on the quinoline and benzyl rings, which are meticulously defined to enhance biological activity while maintaining synthetic feasibility. As illustrated in the general structural formula, the molecule integrates a quinoline moiety linked via a methoxy bridge to a benzylsulfonylurea backbone. This architecture is not merely theoretical; it is supported by robust experimental data demonstrating high in vitro activity against leukotriene synthesis. For procurement specialists and supply chain managers, understanding the synthetic accessibility of this scaffold is paramount, as it directly influences the cost reduction in API manufacturing and the ability to scale production from laboratory grams to commercial metric tons without compromising quality or purity standards.
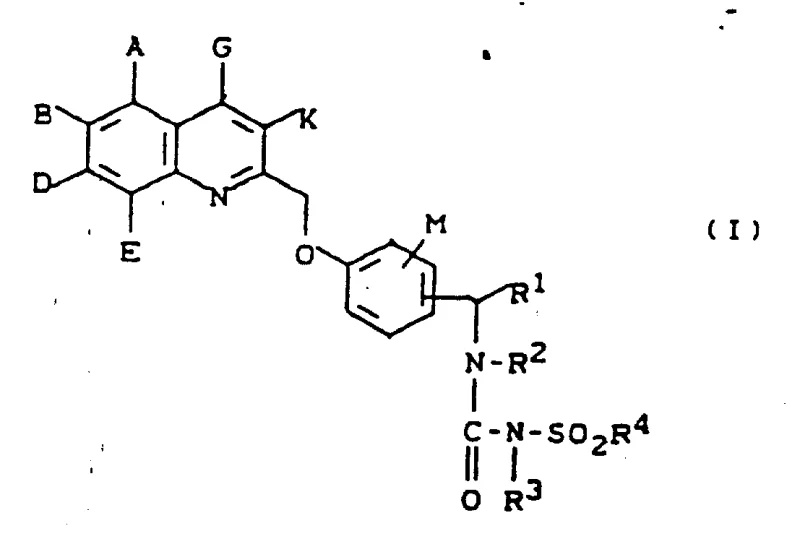
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex sulfonylurea derivatives has been plagued by inefficiencies related to harsh reaction conditions and poor atom economy. Traditional methods often rely on the reaction of amines with sulfonyl chlorides followed by further derivatization, which can generate substantial amounts of salt waste and require rigorous purification steps to remove toxic byproducts. Furthermore, achieving the specific regioselectivity required for the quinoline-benzyl linkage often necessitates multiple protection and deprotection cycles, drastically increasing the number of unit operations and extending the overall lead time. These conventional routes frequently suffer from thermal instability of intermediates, leading to decomposition and reduced yields, which poses a significant challenge for commercial scale-up of complex pharmaceutical intermediates. The reliance on aggressive reagents also complicates waste management, creating environmental compliance burdens that modern green chemistry initiatives seek to eliminate.
The Novel Approach
In stark contrast, the methodology described in CN1049656A offers a streamlined and highly efficient pathway that circumvents these traditional bottlenecks. The novel approach utilizes a direct coupling reaction between a specifically substituted benzylamine and a sulfonyl isocyanate in an inert solvent. This strategy is advantageous because sulfonyl isocyanates are highly electrophilic, allowing the reaction to proceed rapidly under mild conditions, typically ranging from -80°C to 0°C. This low-temperature protocol minimizes thermal degradation and suppresses the formation of unwanted side products, thereby simplifying the downstream purification process. By eliminating the need for excessive protecting groups and reducing the number of synthetic steps, this method significantly enhances the overall process mass intensity (PMI). For a reliable pharmaceutical intermediate supplier, this translates to a more robust manufacturing process that is easier to control and validate, ensuring consistent batch-to-batch quality essential for regulatory approval.
Mechanistic Insights into Sulfonyl Isocyanate Coupling and Amine Synthesis
The chemical elegance of this synthesis is rooted in the precise mechanistic control of nucleophilic attacks and rearrangement reactions. The formation of the sulfonylurea linkage proceeds through a nucleophilic addition of the primary amine nitrogen to the electrophilic carbon of the sulfonyl isocyanate group. This reaction is highly exothermic, which necessitates the controlled addition of reagents and strict temperature maintenance within the -80°C to 0°C window to prevent runaway reactions. The choice of solvent plays a critical role in stabilizing the transition state; polar aprotic solvents like dimethylformamide (DMF) or moderately polar solvents like dichloromethane are preferred to solubilize both the organic amine and the isocyanate while facilitating the proton transfer required for urea formation. Understanding this mechanism allows process chemists to fine-tune stoichiometry, typically using a slight excess of the isocyanate (1.0 to 1.2 equivalents) to drive the reaction to completion without generating excessive impurities that are difficult to purge.
Equally important is the synthesis of the amine precursor, which often involves a sophisticated rearrangement strategy to install the nitrogen atom with high fidelity. The patent details a route where a carboxylic acid derivative is converted into the corresponding amine via an acyl azide intermediate, utilizing diphenyl phosphoryl azide (DPPA) as the activating agent. This transformation, akin to the Curtius rearrangement, proceeds through an isocyanate intermediate which is subsequently hydrolyzed under acidic conditions to yield the primary amine. This method is particularly valuable for constructing sterically hindered amines, such as those containing cyclopentyl or pentyl chains adjacent to the benzylic position, which are difficult to access via direct alkylation. The ability to generate these complex amine scaffolds with high regiochemical purity is a key factor in ensuring the final drug substance meets the stringent purity specifications required for clinical applications.
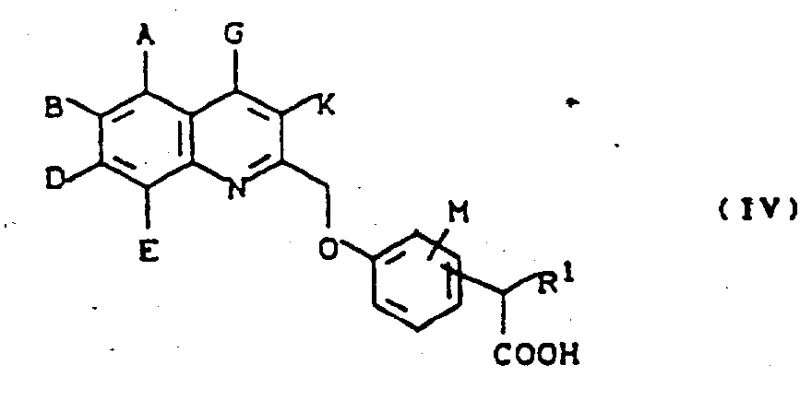
How to Synthesize Substituted N-(quinolin-2-yl-methoxy)benzylsulfonylureas Efficiently
Executing this synthesis at a commercial scale requires a disciplined approach to reaction engineering and process safety. The workflow begins with the preparation of the key amine intermediate, ensuring that all moisture is excluded to prevent premature hydrolysis of sensitive reagents like DPPA. Following the isolation of the amine, the coupling step must be performed under an inert atmosphere, such as nitrogen or argon, to protect the isocyanate functionality from atmospheric moisture. The detailed standardized synthesis steps below outline the critical parameters for temperature, stoichiometry, and workup procedures that have been validated to produce high-quality material suitable for further pharmaceutical development.
- Synthesize the amine precursor by reacting the corresponding carboxylic acid with diphenyl phosphoryl azide in an inert solvent like DMF, followed by acid hydrolysis to yield the primary amine.
- React the purified amine intermediate with the appropriate sulfonyl isocyanate in a dry inert solvent such as dichloromethane or toluene at temperatures between -80°C and 0°C.
- Isolate the final sulfonylurea product through solvent evaporation and purification via silica gel chromatography or recrystallization to ensure high pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the significant simplification of the supply chain for raw materials. The starting materials, including various substituted quinolines and sulfonyl isocyanates, are either commercially available or can be synthesized from commodity chemicals using well-established industrial processes. This availability reduces the risk of supply disruptions and mitigates the volatility associated with sourcing exotic or custom-made reagents. Furthermore, the high yields reported in the patent examples, such as the 92.6% yield in the amine synthesis and 80% in the coupling step, indicate a process with excellent material throughput. High throughput directly correlates to lower cost of goods sold (COGS), enabling more competitive pricing strategies for the final active pharmaceutical ingredient (API).
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in the number of synthetic steps drastically lower the operational expenditure associated with this manufacturing process. By avoiding expensive palladium or nickel catalysts, the need for costly metal scavenging and removal steps is completely removed, which simplifies the purification train and reduces the consumption of specialized resins. Additionally, the ability to run reactions at near-ambient or mildly cooled temperatures reduces the energy load on HVAC and chilling systems compared to cryogenic processes, contributing to substantial cost savings in utility consumption. The high atom economy of the direct coupling reaction ensures that a greater proportion of raw material mass ends up in the final product, minimizing waste disposal fees and maximizing resource efficiency.
- Enhanced Supply Chain Reliability: The robustness of this chemical pathway enhances supply chain resilience by reducing dependency on single-source suppliers for complex intermediates. Since the synthesis relies on fundamental organic transformations like etherification and nucleophilic substitution, it can be easily replicated across multiple manufacturing sites with standard glass-lined or stainless steel reactors. This flexibility allows for dual-sourcing strategies, ensuring continuity of supply even if one facility faces operational challenges. Moreover, the stability of the intermediates allows for safer storage and transportation, reducing the logistical complexities and insurance costs associated with shipping hazardous or unstable chemical species. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this process aligns well with modern green chemistry principles by minimizing the generation of hazardous waste. The solvents used, such as ethyl acetate and dichloromethane, are widely recycled in industrial settings, and the absence of heavy metal residues simplifies the wastewater treatment process. The scalability of the reaction is evidenced by the straightforward workup procedures involving extraction and crystallization, which are unit operations that translate seamlessly from kilogram to ton-scale production. This ease of scale-up reduces the time and capital investment required for technology transfer, allowing new products to reach the market faster. Compliance with environmental regulations is thus achieved not through end-of-pipe treatments but through inherent process design, future-proofing the manufacturing asset against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these quinolinyl-benzylsulfonylurea derivatives. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders assess the feasibility of integrating this chemistry into their existing portfolios and anticipate potential challenges during process validation.
Q: What is the primary pharmacological application of these compounds?
A: These substituted N-(quinolin-2-yl-methoxy)benzylsulfonylureas function primarily as potent 5-lipoxygenase inhibitors, making them valuable intermediates for developing anti-inflammatory and antiallergic medications targeting respiratory diseases like asthma.
Q: What are the optimal reaction conditions for the coupling step?
A: The coupling reaction between the amine and sulfonyl isocyanate is best conducted in inert organic solvents such as dichloromethane or DMF at low temperatures ranging from -80°C to 0°C to minimize side reactions and maximize yield.
Q: How is the amine precursor synthesized from the carboxylic acid?
A: The amine precursor is efficiently generated via a Curtius-type rearrangement using diphenyl phosphoryl azide in the presence of a base like triethylamine, followed by acidic hydrolysis to convert the intermediate isocyanate into the desired amine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(quinolin-2-yl-methoxy)benzylsulfonylureas Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a supply chain partner who understands the intricacies of complex heterocyclic chemistry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering stringent purity specifications through our rigorous QC labs, which are equipped with state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles. Whether you require custom synthesis of specific analogues or large-scale production of the core scaffold, our infrastructure is designed to support your timeline and quality requirements with precision and reliability.
We invite you to engage with our technical procurement team to discuss how we can optimize this synthetic route for your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into reduced costs for your project. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics rather than theoretical projections. Let us be your trusted partner in bringing these innovative lipoxygenase inhibitors from the laboratory to the patients who need them most.
