Advanced Synthesis of Gadobenate Dimeglumine Intermediates for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Gadobenate Dimeglumine Intermediates for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry's demand for high-purity MRI contrast agents has driven significant innovation in synthetic methodology, particularly for complex chelating agents like Gadobenate Dimeglumine. Patent CN102408348A presents a groundbreaking preparation method that fundamentally restructures the synthesis pathway to address critical safety and purity bottlenecks inherent in prior art. This novel approach replaces hazardous high-pressure chlorination steps with a safer addition reaction between 2-chloroacrylonitrile and benzyl alcohol, catalyzed by sodium metal. By shifting the synthetic strategy to utilize esterification and controlled N-alkylation, the process effectively eliminates the generation of toxic chlorine gas and minimizes the formation of cyclic lactam impurities. For R&D directors and procurement specialists seeking a reliable MRI contrast agent intermediate supplier, this technology represents a pivotal shift towards greener, more controllable manufacturing protocols that ensure consistent product quality suitable for regulatory submission.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gadobenate Dimeglumine and its precursors relied heavily on processes disclosed in patents such as EP230893 and EP9904478, which utilized 2-chloro-3-benzyloxy propionic acid prepared via direct chlorination. These conventional routes suffer from severe technical deficiencies that complicate cost reduction in pharmaceutical intermediate manufacturing. The primary drawback is the requirement for high-pressure operations involving toxic chlorine gas, creating substantial safety hazards and necessitating expensive, specialized containment infrastructure that increases capital expenditure. Furthermore, the purification of intermediates in these older methods often depends on ion exchange resin techniques, which consume large quantities of hydrochloric acid and are prone to generating six-membered lactam byproducts through unwanted cyclization. These impurities are notoriously difficult to remove and can compromise the final drug substance's safety profile, leading to batch failures and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The innovative methodology described in the patent data circumvents these issues by introducing a multi-step sequence starting with the addition of benzyl alcohol to 2-chloroacrylonitrile. This reaction, followed by hydrolysis, yields a highly purified 2-chloro-3-benzyloxy propionic acid salt without the need for elemental chlorine. A critical advancement in this route is the subsequent esterification with tert-butanol to form a sterically hindered tert-butyl ester. This structural modification is not merely a protecting group strategy but a kinetic control mechanism that physically blocks the intramolecular nucleophilic attack responsible for lactam formation. By stabilizing the intermediate against cyclization, the process ensures that the subsequent N-alkylation steps proceed with high selectivity. This results in a cleaner reaction profile that simplifies downstream processing and aligns perfectly with the requirements for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into N-Alkylation and Esterification Strategy
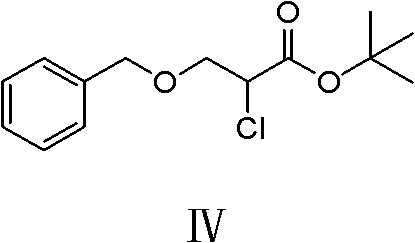
The core of this synthetic breakthrough lies in the precise manipulation of steric and electronic effects during the construction of the polyamine backbone. The initial formation of the tert-butyl ester (Compound IV) serves as a crucial pivot point in the synthesis. As illustrated in the structure below, the bulky tert-butyl group adjacent to the carbonyl functionality creates significant steric hindrance. This physical barrier prevents the nitrogen atom of the incoming diethylenetriamine from attacking the carbonyl carbon of the same molecule, a side reaction that plagues traditional syntheses and leads to the aforementioned lactam impurities. Consequently, the nucleophilic substitution proceeds exclusively at the chloro-substituted carbon, ensuring the linear growth of the molecular chain required for the final chelating agent structure.
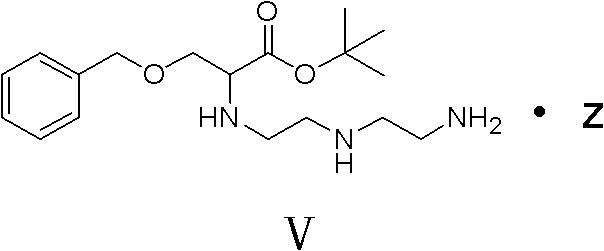
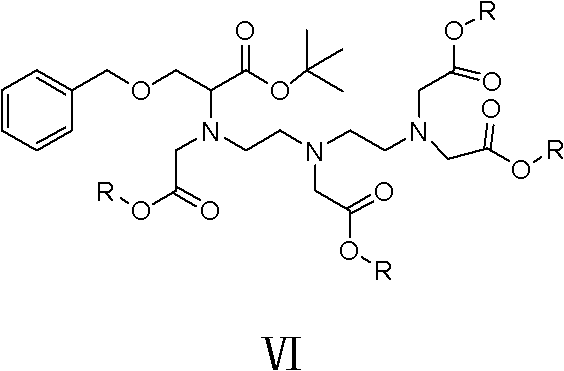
Following the formation of the protected acid, the synthesis advances through sequential N-alkylation reactions. The intermediate Compound IV reacts with diethylenetriamine under alkaline conditions to yield Compound V, introducing the necessary amine functionalities for metal chelation. The patent specifies the use of organic solvents like ethyl acetate or toluene and mild bases such as potassium carbonate, which facilitate the displacement of the chloride leaving group while maintaining the integrity of the ester protection. The resulting amine intermediate is then further alkylated with bromoacetic acid esters to install the carboxymethyl arms. This stepwise construction allows for rigorous quality control at each stage, as each intermediate can be isolated and purified via recrystallization rather than complex chromatographic or resin-based methods. The final deprotection and hydrolysis steps reveal the free carboxylic acids needed for gadolinium coordination, completing the transformation into the BOPTA ligand.
The final stages of the synthesis involve the hydrolysis of the ester groups and the chelation with gadolinium sesquioxide. The use of the tert-butyl ester proves advantageous here as well, as it can be cleaved under specific acidic conditions (such as trifluoroacetic acid) or basic hydrolysis without degrading the sensitive polyamine core. The resulting ligand, BOPTA, is then complexed with gadolinium in the presence of meglumine to form the final contrast agent. The entire pathway is designed to minimize the introduction of heavy metal contaminants and organic impurities, which is critical for injectable pharmaceutical products. The ability to purify every intermediate through simple recrystallization from solvents like ethanol or isopropanol demonstrates a robust understanding of process chemistry that prioritizes scalability and reproducibility over theoretical yield alone.
How to Synthesize Gadobenate Dimeglumine Intermediates Efficiently
The implementation of this novel synthesis route requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the initial addition and alkylation steps. The patent outlines a clear progression from raw materials to the final chelated product, emphasizing the importance of intermediate purification to prevent impurity carryover. Operators must strictly adhere to the specified molar ratios, such as the 1:0.5 to 1:5 ratio between 2-chloroacrylonitrile and benzyl alcohol, to maximize conversion while minimizing side reactions. The detailed standardized synthesis steps provided below offer a comprehensive guide for laboratory and pilot-scale execution, ensuring that the theoretical advantages of the process are realized in practical application.
- React 2-chloroacrylonitrile with benzyl alcohol using sodium metal as a catalyst, followed by hydrolysis and recrystallization to obtain highly purified 2-chloro-3-benzyloxy propionic acid salt.
- Perform esterification of the acid salt with tert-butanol under DCC conditions to form the sterically hindered tert-butyl ester intermediate (Compound IV).
- Conduct N-alkylation reactions sequentially with diethylenetriamine and bromoacetic acid esters under alkaline conditions to build the polyamine backbone (Compounds V and VI).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic benefits that extend beyond simple unit cost calculations. The elimination of high-pressure chlorination equipment drastically reduces the capital expenditure required for facility setup and maintenance, directly impacting the overall cost structure of the manufacturing process. Furthermore, by removing the need for handling toxic chlorine gas, the operational risk profile is significantly lowered, leading to reduced insurance premiums and fewer regulatory hurdles related to environmental safety compliance. The simplified purification workflow, which relies on recrystallization rather than ion exchange resins, decreases the consumption of auxiliary chemicals like hydrochloric acid and reduces the volume of hazardous waste generated, contributing to substantial cost savings in waste disposal and environmental management.
- Cost Reduction in Manufacturing: The removal of expensive high-pressure reactors and toxic gas handling systems translates to lower fixed costs and depreciation expenses for the manufacturing facility. Additionally, the replacement of ion exchange resin purification with solvent-based recrystallization reduces the recurring cost of consumables and the energy load associated with resin regeneration and desalination processes. This streamlined approach minimizes the number of unit operations required, thereby lowering labor costs and increasing overall equipment effectiveness through faster batch turnover times.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials like 2-chloroacrylonitrile and benzyl alcohol, the supply chain becomes less vulnerable to the fluctuations and shortages often associated with specialized chlorinating agents. The robustness of the recrystallization purification steps ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical customers who require strict adherence to delivery schedules for their own clinical and commercial programs.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial amplification, avoiding the bottlenecks of millimicro-straining installations and complex desalination steps found in older technologies. The use of common organic solvents like ethyl acetate and toluene facilitates easier solvent recovery and recycling, aligning with modern green chemistry principles. This environmental compatibility not only future-proofs the manufacturing site against tightening regulations but also enhances the corporate sustainability profile, a key factor for multinational pharmaceutical partners evaluating potential suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Gadobenate Dimeglumine intermediates using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the new process. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does the new synthesis method avoid the formation of lactam byproducts?
A: The novel process utilizes a tert-butyl ester protection group on the propionic acid chain. This steric hindrance and electronic effect significantly reduce the probability of intramolecular cyclization that typically forms unwanted 6-membered lactam rings during the alkylation steps.
Q: Why is the elimination of chlorine gas important for industrial scale-up?
A: Traditional methods require high-pressure chlorination using toxic chlorine gas, which poses severe safety risks and environmental hazards. The new method uses 2-chloroacrylonitrile and benzyl alcohol in an addition reaction, removing the need for pressurized toxic gas handling and specialized containment equipment.
Q: What purification advantages does this route offer over ion exchange resin methods?
A: Instead of complex ion exchange resin separation which requires large amounts of hydrochloric acid and specialized nanofiltration, this process allows for purification via simple recrystallization and organic solvent extraction at each step, ensuring stable quality control and easier waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gadobenate Dimeglumine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex MRI contrast agents depends on a partner who can bridge the gap between innovative patent chemistry and robust industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate steps of this novel synthesis are executed with precision and consistency. We maintain stringent purity specifications across all our facilities, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the absence of critical impurities like lactams and heavy metals at every stage of the value chain.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply security and cost efficiency. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive your contrast agent projects forward with confidence and speed.
