Scalable Production of High-Purity Artemisinin via Optimized Chemical Oxidation Routes
The global demand for effective antimalarial therapeutics continues to drive innovation in the synthesis of key active pharmaceutical ingredients, with artemisinin remaining the gold standard for treatment protocols. A pivotal advancement in this domain is documented in patent CN103172645A, which discloses a high-efficiency synthesis method that fundamentally shifts production away from resource-intensive plant extraction toward robust chemical semisynthesis. This technological breakthrough addresses the critical bottlenecks of traditional sourcing, such as seasonal variability and ecological strain, by utilizing artemisinic acid derived from fermentation processes or extraction by-products as the primary starting material. The methodology outlined in this intellectual property represents a significant leap forward for reliable artemisinin supplier networks, offering a pathway that is not only environmentally friendlier but also capable of sustaining the rigorous purity specifications required by international regulatory bodies. By leveraging advanced catalytic oxidation techniques, this process ensures a stable supply chain for high-purity artemisinin, mitigating the risks associated with agricultural dependency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of artemisinin has been plagued by significant inefficiencies inherent to both direct plant extraction and early-generation synthetic attempts. Traditional extraction from Artemisia annua is notoriously labor-intensive, requiring vast quantities of biomass and resulting in yields that fluctuate wildly based on harvest timing and geographic location. Furthermore, early synthetic approaches, such as those described in patent US4992561, relied heavily on photochemical methods to introduce the essential peroxy bridge. These photochemical processes are inherently difficult to scale due to light penetration limitations in large reactors, leading to cumbersome operations and poor atom economy. Other prior art methods utilized ozone for oxidation, which introduces severe safety hazards and operational complexities that are ill-suited for large-scale commercial manufacturing. Consequently, these legacy techniques often suffered from low total yields, sometimes falling below 20 percent, and generated substantial waste streams, making them economically and environmentally unsustainable for modern cost reduction in API manufacturing initiatives.
The Novel Approach
In stark contrast to these legacy constraints, the novel approach detailed in the patent data utilizes a sophisticated chemical oxidation strategy that eliminates the need for hazardous ozone or complex photoreactors. This method achieves the critical introduction of the peroxygen bond through the use of accessible metal catalysts and common peroxides under controlled, mild conditions. The process is designed with modularity in mind, offering two distinct synthetic routes: a direct oxidation path and a protected intermediate path, allowing manufacturers to optimize for either speed or selectivity depending on their specific infrastructure. By replacing unstable or dangerous reagents with stable metal salts and hydrogen peroxide derivatives, the novel approach drastically simplifies the operational workflow and enhances safety profiles. This shift enables the commercial scale-up of complex pharmaceutical intermediates with significantly reduced lead times, as the reaction conditions are compatible with standard stainless steel reactor systems found in most fine chemical facilities, thereby removing the barrier to entry for widespread adoption.
Mechanistic Insights into Metal-Catalyzed Peroxidation and Rearrangement
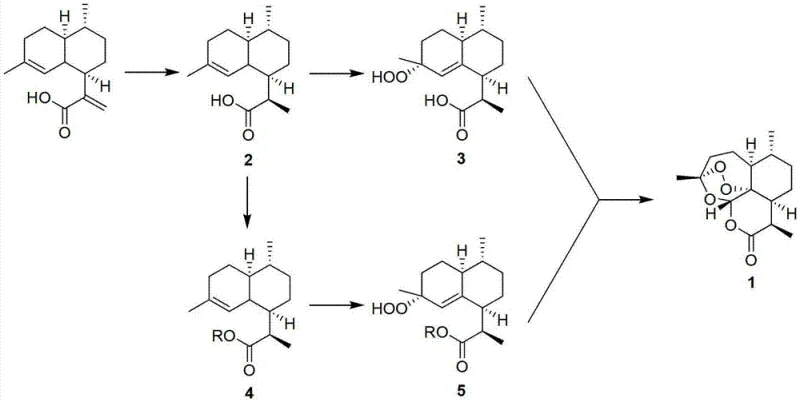
The core of this synthetic innovation lies in the precise mechanistic control over the oxidation and rearrangement steps, which are critical for establishing the unique endoperoxide bridge characteristic of artemisinin. The process begins with the highly stereoselective reduction of artemisinic acid to dihydroartemisinic acid, achieving yields approaching 99 percent through optimized hydrogenation or hydride reduction conditions. Following this, the pivotal oxidation step employs a diverse array of metal catalysts, including salts of molybdenum, tungsten, vanadium, or lanthanum, to activate peroxides such as hydrogen peroxide or tert-butyl hydroperoxide. This catalytic system facilitates the electrophilic attack on the olefinic bond, generating the hydroperoxy intermediate with high regioselectivity. In the alternative route, the carboxyl group is temporarily protected using various silyl or alkyl groups, which serves to modulate the electronic environment of the molecule, further suppressing side reactions and enhancing the stability of the peroxide intermediate before the final cyclization event occurs.
The final transformation involves an acid-catalyzed rearrangement conducted in the presence of oxygen, which triggers the intramolecular cyclization necessary to form the sesquiterpene lactone structure. This step is meticulously controlled using Bronsted or Lewis acids, such as p-toluenesulfonic acid or copper triflate, to ensure the correct stereochemical outcome while minimizing the decomposition of the sensitive peroxide moiety. The ability to tune the acid catalyst and solvent system allows for the suppression of common impurities that typically arise during peroxide handling. This level of mechanistic understanding translates directly into superior impurity control, ensuring that the final crystalline product meets stringent pharmacopeial standards without requiring excessive chromatographic purification. The integration of these chemical principles demonstrates a deep command of organic synthesis, transforming a fragile natural product structure into a robustly manufacturable commodity.
How to Synthesize Artemisinin Efficiently
Implementing this synthesis protocol requires a systematic approach to reaction engineering, beginning with the preparation of high-quality dihydroartemisinic acid from fermented artemisinic acid feedstocks. The standardized procedure involves dissolving the starting material in a polar or non-polar solvent system, such as methanol or tetrahydrofuran, and introducing the reducing agent under controlled temperature and pressure regimes to ensure complete saturation of the double bond. Following isolation of the reduced intermediate, the process diverges based on the chosen route, either proceeding directly to oxidation or undergoing a protection step to mask the carboxylic acid functionality. The subsequent oxidation phase demands careful addition of peroxide sources to maintain thermal safety while maximizing conversion to the peroxide intermediate. Detailed standardized synthesis steps see the guide below.
- Reduce artemisinic acid to dihydroartemisinic acid using catalytic hydrogenation or hydride reduction.
- Optionally protect the carboxyl group to form a dihydroartemisinic acid derivative.
- Oxidize the intermediate using metal catalysts and peroxides to introduce the peroxy bridge.
- Perform acid-catalyzed rearrangement in the presence of oxygen to cyclize and form artemisinin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology offers transformative benefits that extend far beyond simple yield improvements. The primary advantage lies in the decoupling of production from agricultural cycles, which historically caused volatile pricing and supply shortages for artemisinin. By utilizing fermentation-derived starting materials and a fully chemical synthesis pathway, manufacturers can guarantee continuous production schedules regardless of seasonal weather patterns or crop failures. This reliability is crucial for maintaining the continuity of supply for life-saving antimalarial medications, allowing pharmaceutical companies to plan long-term inventory strategies with confidence. Furthermore, the use of cheap and easily obtainable reagents, such as common metal salts and hydrogen peroxide, significantly lowers the raw material cost base compared to exotic catalysts or specialized photochemical equipment required by older methods.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and hazardous processing steps. By avoiding the need for specialized photochemical reactors or ozone generators, capital expenditure for new production lines is substantially reduced, while existing infrastructure can often be retrofitted with minimal investment. The high selectivity of the catalytic oxidation minimizes the formation of by-products, which in turn reduces the burden on downstream purification units and lowers solvent consumption. Additionally, the ability to telescope certain steps or utilize crude intermediates directly in subsequent reactions without extensive purification further drives down operational expenses, resulting in substantial cost savings throughout the value chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved through the use of stable, shelf-stable reagents that do not require complex cold-chain logistics or immediate usage upon delivery. The starting material, artemisinic acid, can be sourced from multiple fermentation facilities globally, creating a diversified supply base that mitigates the risk of single-source failure. The robustness of the chemical steps ensures that production batches are consistent in quality and quantity, reducing the incidence of failed batches that can disrupt delivery timelines. This predictability allows logistics teams to optimize shipping schedules and reduce safety stock levels, freeing up working capital while ensuring that critical API intermediates are available exactly when needed for formulation.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and regulatory expectations. The avoidance of ozone and the use of aqueous hydrogen peroxide as an oxidant result in water as the primary by-product, significantly reducing the toxic load of waste streams. The mild reaction conditions, often operating at or near ambient temperatures, lower energy consumption for heating and cooling, contributing to a reduced carbon footprint for the manufacturing site. The process is inherently scalable from kilogram to multi-ton scales without the linear increase in complexity seen in photochemical processes, making it ideal for meeting surging global demand without compromising on environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this artemisinin synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers below reflect the specific advantages and operational parameters defined within the intellectual property.
Q: Why is this chemical synthesis method preferred over traditional extraction?
A: Traditional extraction relies on seasonal plant harvesting, leading to supply instability and ecological concerns. This patented chemical route uses fermentation-derived artemisinic acid, ensuring consistent quality, higher atom economy, and independence from agricultural variables.
Q: What are the safety advantages of this oxidation method compared to prior art?
A: Previous methods often utilized hazardous ozone or complex photochemical setups which are difficult to scale safely. This method employs stable metal salt catalysts and common peroxides under mild temperatures, significantly reducing operational risks and simplifying industrial reactor requirements.
Q: How does the carboxyl protection strategy impact the final yield?
A: The patent describes an optional route where protecting the carboxyl group prior to oxidation can enhance regioselectivity. This modification minimizes side reactions during the critical peroxide formation step, often resulting in improved overall purity and facilitating easier downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Artemisinin Supplier
As the pharmaceutical industry increasingly prioritizes secure and sustainable supply chains for critical antimalarial agents, the expertise to execute complex synthetic routes like the one described in CN103172645A becomes a vital strategic asset. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing the technical acumen and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require pilot-scale quantities for clinical trials or metric tons for global distribution, we can deliver with precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of artemisinin meets the highest international standards, providing peace of mind to our partners regarding product quality and regulatory compliance.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthesis route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits tailored to your volume needs. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than projections. Together, we can secure the future of antimalarial therapy through innovation, reliability, and a shared commitment to excellence in chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
