Revolutionizing Benzisothiazole Production: Advanced Synthetic Routes for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance molecular complexity with economic viability, particularly for heterocyclic scaffolds like benzisothiazoles which serve as critical backbones in numerous therapeutic agents. Patent CN1187346C introduces a transformative approach to synthesizing 3-substituted benzisothiazole derivatives by leveraging a novel intermediate, 2-cyano-thiophenyl halogen, which effectively bypasses the historical bottlenecks associated with traditional precursor availability. This technological breakthrough addresses the persistent challenge of sourcing high-purity starting materials, offering a streamlined route that transitions smoothly from laboratory discovery to industrial application without compromising on yield or structural integrity. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms, ensuring that the supply of these essential intermediates remains stable even amidst fluctuating raw material markets. By redefining the synthetic entry point, this methodology not only enhances process safety but also opens new avenues for derivative exploration, making it a cornerstone technology for modern medicinal chemistry pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted benzisothiazole derivatives has been heavily reliant on the availability of 3-halo-1,2-benzisothiazole, a precursor that has long plagued supply chains due to its synthetic complexity and scarcity. As illustrated in the prior art reaction schemes, the conventional pathway necessitates the use of this specific halogenated heterocycle, which is notoriously difficult to procure in bulk quantities and often commands a premium price that inflates the overall cost of goods sold. 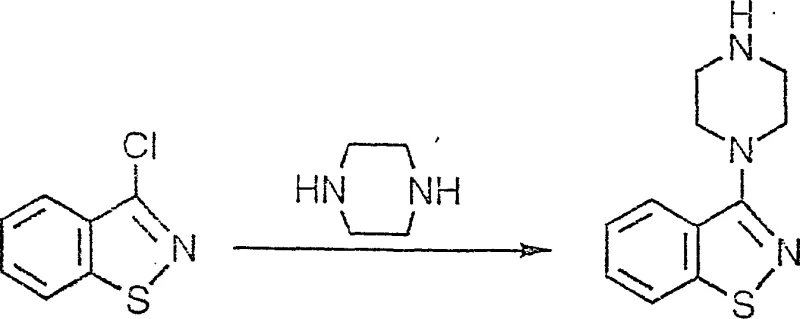 The reliance on such specialized starting materials creates a fragile supply chain vulnerability, where any disruption in the production of the halo-benzisothiazole directly halts the downstream manufacturing of vital pharmaceutical intermediates. Furthermore, the existing literature indicates that alternative methods involving thiosalicylic acid or chlorinated ketones suffer from low yields and require harsh reaction conditions that are environmentally taxing and operationally hazardous. These legacy processes often involve multiple purification steps to remove stubborn impurities, leading to significant material loss and extended production cycles that are incompatible with the demands of modern just-in-time manufacturing. Consequently, the industry has faced a persistent need for a more resilient synthetic strategy that decouples production from these scarce and expensive resources.
The reliance on such specialized starting materials creates a fragile supply chain vulnerability, where any disruption in the production of the halo-benzisothiazole directly halts the downstream manufacturing of vital pharmaceutical intermediates. Furthermore, the existing literature indicates that alternative methods involving thiosalicylic acid or chlorinated ketones suffer from low yields and require harsh reaction conditions that are environmentally taxing and operationally hazardous. These legacy processes often involve multiple purification steps to remove stubborn impurities, leading to significant material loss and extended production cycles that are incompatible with the demands of modern just-in-time manufacturing. Consequently, the industry has faced a persistent need for a more resilient synthetic strategy that decouples production from these scarce and expensive resources.
The Novel Approach
In stark contrast to the constrained legacy methods, the novel approach detailed in the patent utilizes 2-cyano-thiophenyl halogen as a versatile and accessible building block, fundamentally altering the economic landscape of benzisothiazole production. This new intermediate can be efficiently generated through the direct halogenation of readily available 2-cyano-thiophenyl derivatives, such as 2-cyano-benzenethiol or its sulfide analogues, which are abundant in the global chemical market.  By shifting the synthetic focus to this cyano-thiophenyl scaffold, manufacturers can leverage established halogenation protocols using common reagents like chlorine or bromine, thereby eliminating the dependency on exotic precursors. The process is designed to be telescoped, allowing for the continuous conversion of the thiol derivative to the halogenated intermediate and subsequently to the final benzisothiazole product without the need for isolating unstable species. This continuity not only reduces solvent consumption and waste generation but also significantly shortens the overall processing time, enhancing the throughput capacity of production facilities. The result is a robust, scalable protocol that aligns perfectly with the requirements for reliable pharmaceutical intermediate supplier networks seeking to optimize their operational efficiency.
By shifting the synthetic focus to this cyano-thiophenyl scaffold, manufacturers can leverage established halogenation protocols using common reagents like chlorine or bromine, thereby eliminating the dependency on exotic precursors. The process is designed to be telescoped, allowing for the continuous conversion of the thiol derivative to the halogenated intermediate and subsequently to the final benzisothiazole product without the need for isolating unstable species. This continuity not only reduces solvent consumption and waste generation but also significantly shortens the overall processing time, enhancing the throughput capacity of production facilities. The result is a robust, scalable protocol that aligns perfectly with the requirements for reliable pharmaceutical intermediate supplier networks seeking to optimize their operational efficiency.
Mechanistic Insights into Electrophilic Halogenation and Cyclization
The core of this innovative synthesis lies in the precise control of electrophilic halogenation, where the 2-cyano-thiophenyl derivative undergoes selective substitution to form the critical sulfur-halogen bond found in Formula I. The reaction mechanism involves the interaction of the sulfur atom with halogen molecules (Cl2 or Br2), facilitated by the electron-withdrawing nature of the adjacent cyano group which stabilizes the transition state and prevents over-halogenation of the aromatic ring. Temperature control plays a pivotal role in this step, with optimal ranges identified between -10°C and 130°C to balance reaction kinetics against the formation of side products, ensuring that the resulting 2-cyano-thiophenyl halogen maintains high chemical purity. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters, such as reagent stoichiometry and addition rates, to maximize yield while minimizing the generation of hazardous byproducts. The ability to isolate these halogenated intermediates as stable white crystals further underscores the practical utility of this method, providing a tangible checkpoint for quality control before proceeding to the final coupling stage.
Following the formation of the halogenated intermediate, the subsequent cyclization with piperazine compounds proceeds through a nucleophilic substitution mechanism that constructs the benzisothiazole ring system with remarkable efficiency. The nitrogen atom of the piperazine acts as a nucleophile, attacking the electrophilic carbon adjacent to the sulfur-halogen bond, triggering an intramolecular rearrangement that closes the heterocyclic ring. 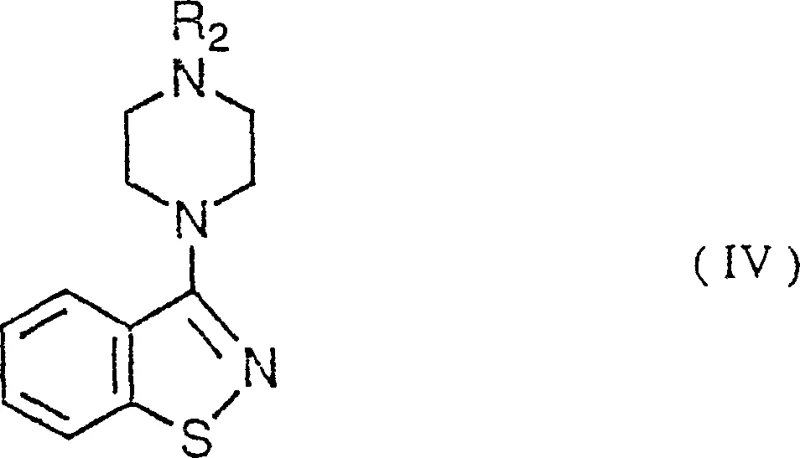 This step is notably tolerant of various substituents on the piperazine ring, allowing for the synthesis of a diverse library of 3-substituted derivatives including methyl, ethyl, and butyl variants without requiring significant changes to the reaction conditions. The impurity profile is tightly controlled during this phase, as the reaction conditions favor the desired cyclization over potential polymerization or degradation pathways, resulting in a crude product that requires minimal purification. Such mechanistic robustness is crucial for maintaining consistent batch-to-batch quality, a key metric for R&D directors evaluating the feasibility of scaling complex organic syntheses for clinical and commercial supply.
This step is notably tolerant of various substituents on the piperazine ring, allowing for the synthesis of a diverse library of 3-substituted derivatives including methyl, ethyl, and butyl variants without requiring significant changes to the reaction conditions. The impurity profile is tightly controlled during this phase, as the reaction conditions favor the desired cyclization over potential polymerization or degradation pathways, resulting in a crude product that requires minimal purification. Such mechanistic robustness is crucial for maintaining consistent batch-to-batch quality, a key metric for R&D directors evaluating the feasibility of scaling complex organic syntheses for clinical and commercial supply.
How to Synthesize 2-Cyano-Thiophenyl Halogen Efficiently
The practical implementation of this synthesis route involves a straightforward sequence that begins with the preparation of the 2-cyano-thiophenyl halogen intermediate, serving as the foundation for the entire process. Detailed standardized operating procedures for this synthesis, including specific reagent grades, mixing speeds, and thermal profiles, are essential for ensuring reproducibility across different manufacturing sites.
- Halogenation of 2-cyano-thiophenyl derivatives (Formula II) using chlorine or bromine to form 2-cyano-thiophenyl halogen (Formula I).
- Reaction of the resulting 2-cyano-thiophenyl halogen with a piperazine compound (Formula III) under heated conditions.
- Purification of the final 3-substituted benzisothiazole (Formula IV) via crystallization or acid-base extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented methodology offers substantial advantages by fundamentally restructuring the cost drivers associated with benzisothiazole manufacturing. The elimination of the need for scarce 3-halo-1,2-benzisothiazole removes a major bottleneck, allowing supply chain managers to source raw materials from a broader and more competitive vendor base, thereby mitigating the risk of supply disruptions. This shift not only stabilizes the supply chain but also introduces significant flexibility in negotiating pricing, as the new starting materials are commodity chemicals with well-established global production capacities. Furthermore, the high yields reported in the patent examples translate directly into improved material efficiency, reducing the volume of waste that requires disposal and lowering the environmental compliance burden on manufacturing facilities. These factors combine to create a more resilient and economically attractive production model that supports long-term business continuity.
- Cost Reduction in Manufacturing: The transition to 2-cyano-thiophenyl derivatives as starting materials drastically simplifies the raw material portfolio, removing the premium costs associated with specialized heterocyclic precursors. By utilizing common halogenating agents and avoiding expensive transition metal catalysts, the process inherently lowers the variable cost per kilogram of the final product. Additionally, the high conversion rates minimize the loss of valuable intermediates, ensuring that a greater proportion of input materials are converted into saleable product, which drives down the overall cost of goods. This economic efficiency is further amplified by the potential for solvent recovery and reuse, given the simplicity of the reaction mixture compared to traditional multi-step syntheses.
- Enhanced Supply Chain Reliability: Sourcing 2-cyano-benzenethiol and related sulfides is significantly more reliable than procuring niche halogenated benzisothiazoles, as these chemicals are produced by multiple suppliers globally. This diversification of the supply base reduces the risk of single-source dependency, ensuring that production schedules can be maintained even if one vendor faces operational issues. The stability of the intermediate also allows for strategic stockpiling, enabling manufacturers to buffer against market volatility and ensure continuous delivery to downstream customers. Consequently, lead times for high-purity intermediates can be reduced, providing a competitive edge in fast-moving pharmaceutical development timelines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily managed in large-scale reactors without requiring exotic high-pressure or cryogenic equipment. The absence of heavy metal catalysts simplifies the purification process and reduces the environmental footprint, aligning with increasingly stringent global regulations on chemical manufacturing emissions. Waste streams are less complex and easier to treat, lowering the operational costs associated with environmental compliance and waste disposal. This green chemistry profile not only enhances corporate sustainability metrics but also future-proofs the manufacturing process against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's technical disclosures and beneficial effects. These insights are intended to clarify the operational benefits and feasibility for stakeholders evaluating this technology for integration into their supply chains.
Q: What is the primary advantage of using 2-cyano-thiophenyl halogen over traditional precursors?
A: The primary advantage lies in the availability and cost of raw materials. Traditional methods rely on 3-halo-1,2-benzisothiazole, which is difficult to obtain and expensive. The novel route utilizes readily available 2-cyano-thiophenyl derivatives, significantly simplifying the supply chain and reducing production costs.
Q: What are the typical reaction conditions for the halogenation step?
A: The halogenation is typically conducted at temperatures ranging from -10°C to 130°C, depending on the specific derivative. Solvents such as chlorobenzene or hydrocarbons can be used, though solvent-free conditions are also viable. The process demonstrates high efficiency with yields exceeding 95% in optimized examples.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly highlights industrial usability. The reactions utilize common reagents like chlorine or bromine and standard solvents, avoiding exotic catalysts. The high yields and straightforward purification steps (crystallization/distillation) make it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-Thiophenyl Halogen Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN1187346C to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications and maintaining rigorous QC labs to guarantee that every batch of 2-cyano-thiophenyl halogen and its derivatives meets the exacting standards required by top-tier drug manufacturers. Our infrastructure is specifically designed to handle complex heterocyclic chemistry, providing a secure and compliant environment for the production of high-value intermediates.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific project needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits and supply chain improvements this technology offers for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your manufacturing operations.
