Advanced One-Pot Manufacturing of Cephalosporin Esters for Scalable API Production
Advanced One-Pot Manufacturing of Cephalosporin Esters for Scalable API Production
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of beta-lactam antibiotics, particularly cephalosporins, which remain a cornerstone in the treatment of infectious diseases. Patent CN1009732B introduces a transformative approach to the manufacture of cephalosporin ester derivatives, specifically addressing the challenges associated with traditional multi-step esterification and acylation protocols. This technology leverages a sophisticated one-pot synthesis strategy that utilizes partially chlorinated lower alkanes as the reaction medium, enabling the seamless conversion of 7-amino-3-methyl-3-cephem-4-carboxylic acid (7-ADCA) into high-value ester intermediates. By integrating salt formation, esterification, and acylation into a single continuous process, this method not only enhances the overall chemical yield but also drastically simplifies the operational workflow required for commercial-scale manufacturing of oral cephalosporin prodrugs.
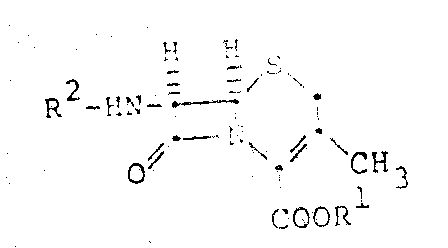
The core innovation lies in the strategic selection of reagents and conditions that preserve the integrity of the sensitive beta-lactam ring while ensuring high regioselectivity. The process begins with the suspension of 7-ADCA in solvents such as dichloromethane or chloroform, followed by treatment with a bicyclic amidine, typically 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU). This specific base is crucial as it forms a soluble salt with the acidic starting material without inducing the notorious Delta-2 isomerization that often plagues cephalosporin chemistry. Subsequent addition of a halide, such as pivaloyloxymethyl iodide, effects the esterification in situ. This intermediate is then directly subjected to acylation using activated thioesters, bypassing the need for isolating the unstable ester intermediate. This seamless integration of reaction steps represents a significant leap forward in process chemistry, offering a reliable pathway for producing complex antibiotic intermediates with improved purity profiles and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for cephalosporin esters have historically been fraught with inefficiencies that hinder large-scale production and increase the cost of goods sold. Conventional methods often rely on stepwise procedures where the esterification of the carboxylic acid and the acylation of the amino group are performed as distinct, isolated operations. This fragmentation necessitates the isolation and purification of intermediate species, which are frequently unstable and prone to degradation under standard workup conditions. Furthermore, classical acylation strategies frequently require the use of protecting groups for amino functionalities, particularly when dealing with aromatic amines found in modern third-generation cephalosporins. The installation and subsequent removal of these protecting groups add substantial complexity, requiring additional reagents, solvents, and processing time, all of which contribute to higher waste generation and lower overall atom economy. Moreover, the use of harsh conditions or incompatible reagents in sequential steps can lead to the formation of impurities, such as Delta-2 isomers or ring-opened byproducts, complicating downstream purification and potentially compromising the safety profile of the final Active Pharmaceutical Ingredient (API).
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in patent CN1009732B offers a streamlined, cohesive solution that addresses these bottlenecks through intelligent process design. By employing a one-pot methodology, the process eliminates the need for intermediate isolation, thereby maintaining the reactive species in an optimal environment throughout the transformation. The use of DBU as a salt-forming agent is particularly advantageous; it creates a highly reactive nucleophile from the 7-ADCA substrate while simultaneously buffering the reaction medium to prevent acid-catalyzed degradation of the beta-lactam core. The subsequent esterification with alkyl halides proceeds smoothly under mild conditions, typically at room temperature, avoiding the thermal stress that can trigger decomposition. Crucially, the direct acylation using thioesters allows for the modification of the amino group without the prerequisite of protecting group chemistry. This not only shortens the synthetic timeline but also removes entire classes of impurities associated with protection and deprotection cycles. The result is a cleaner reaction profile, higher crude purity, and a manufacturing process that is inherently more amenable to scale-up and regulatory compliance.
Mechanistic Insights into DBU-Mediated Salt Formation and Thioester Acylation
The mechanistic elegance of this process centers on the dual role of the bicyclic amidine base and the unique reactivity of the thioester acylating agents. When 7-ADCA is treated with DBU in a chlorinated solvent, the amidine nitrogen abstracts the proton from the carboxylic acid moiety, generating a carboxylate anion paired with the protonated DBU cation. This salt formation is critical because it solubilizes the zwitterionic 7-ADCA in organic media, making the carboxylate oxygen available for nucleophilic attack on the alkyl halide. Unlike smaller amines or inorganic bases, the bulky, non-nucleophilic nature of DBU prevents it from attacking the beta-lactam carbonyl or the ester linkage, thereby preserving the structural integrity of the molecule. Furthermore, the basicity is tuned perfectly to avoid the base-catalyzed isomerization of the double bond from the Delta-3 position to the thermodynamically more stable but biologically inactive Delta-2 position. This precise control over the reaction microenvironment ensures that the stereochemical fidelity of the cephalosporin nucleus is maintained throughout the esterification phase.
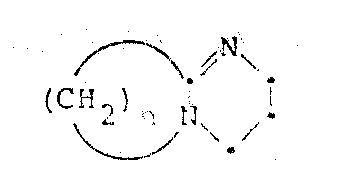
Following esterification, the introduction of the acyl side chain is achieved using activated thioesters, such as 2-phenylthiazole thioesters. The mechanism here involves the nucleophilic attack of the amino group at the 7-position on the carbonyl carbon of the thioester. Thioesters are significantly more reactive than their oxygen-ester counterparts due to the poorer orbital overlap between the carbon and sulfur atoms, which makes the carbonyl carbon more electrophilic. This heightened reactivity allows the acylation to proceed efficiently even without the activation typically required for carboxylic acids, such as carbodiimides or acid chlorides. A profound advantage of this specific mechanistic pathway is its compatibility with unprotected aromatic amines. In conventional peptide coupling, aromatic amines often require protection to prevent side reactions or oxidation; however, the mild yet effective nature of the thioester acylation permits the direct formation of the amide bond. This eliminates the need for orthogonal protection strategies, reducing the number of synthetic steps and minimizing the generation of hazardous byproducts, ultimately leading to a more sustainable and cost-effective manufacturing process for complex cephalosporin derivatives.
How to Synthesize Cephalosporin Esters Efficiently
The practical implementation of this synthesis route requires careful attention to stoichiometry and reaction conditions to maximize yield and minimize impurity formation. The process initiates by suspending the 7-ADCA starting material in a suitable volume of dichloromethane, ensuring a uniform dispersion before the addition of the base. The molar ratio of DBU to 7-ADCA is critical; it should be slightly less than stoichiometric or exactly stoichiometric to prevent excess base from catalyzing isomerization. Once the salt is formed, the alkyl halide is introduced to drive the esterification to completion. The reaction mixture is then treated directly with the thioester acylating agent, allowing the acylation to occur in the same pot. Temperature control is maintained between 0°C and 40°C, with room temperature being optimal for balancing reaction rate and stability. The final product is typically isolated as a pharmaceutically acceptable acid addition salt, such as the hydrochloride, through crystallization from a mixed solvent system. For a detailed, step-by-step breakdown of the standardized operating procedure, please refer to the technical guide below.
- Suspend 7-ADCA in dichloromethane and treat with DBU to form the salt intermediate.
- Add pivaloyloxymethyl iodide to effect esterification without isolation.
- Directly acylate with a thioester reagent to yield the final cephalosporin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors in the pharmaceutical sector. The primary value driver is the significant reduction in manufacturing costs achieved through process intensification. By consolidating three distinct chemical transformations—salt formation, esterification, and acylation—into a single reactor vessel, the process drastically reduces the consumption of solvents, which are often a major cost component in fine chemical manufacturing. Furthermore, the elimination of intermediate isolation steps means that filtration, drying, and re-dissolution operations are removed from the workflow. This not only saves on utility costs such as energy for drying and vacuum generation but also reduces the labor hours required per batch. The qualitative improvement in process efficiency translates to a lower cost of goods sold (COGS), allowing for more competitive pricing strategies in the global market for antibiotic intermediates without compromising on quality margins.
- Cost Reduction in Manufacturing: The economic benefits of this route are derived principally from the simplification of the unit operations. Traditional multi-step syntheses incur costs at every transfer and isolation point, including equipment cleaning, quality control testing of intermediates, and material handling losses. By adopting a telescoped one-pot approach, these overheads are substantially minimized. Additionally, the use of thioesters negates the expense associated with purchasing and managing protecting group reagents and the chemicals required for their removal. The avoidance of expensive transition metal catalysts or harsh coupling reagents further contributes to a leaner bill of materials. Consequently, manufacturers can achieve a more favorable cost structure, providing a buffer against raw material price volatility and enhancing the overall profitability of the cephalosporin supply chain.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the described chemistry. Complex synthetic routes with numerous steps are inherently more prone to failure modes, where a deviation in an early step can compromise the entire batch. The streamlined nature of this one-pot process reduces the number of potential failure points, leading to higher batch success rates and more predictable production schedules. Moreover, the reagents employed, such as DBU and common alkyl halides, are commercially available commodities with stable supply lines, reducing the risk of raw material shortages. The ability to produce high-purity intermediates with fewer purification steps also shortens the overall cycle time from raw material intake to finished goods. This agility enables suppliers to respond more rapidly to fluctuations in market demand, ensuring continuity of supply for downstream API manufacturers and mitigating the risk of stockouts.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden challenges, but this methodology is inherently designed for scalability. The reaction conditions are mild, operating near ambient temperature and pressure, which reduces the engineering demands on the production facility and lowers capital expenditure requirements for specialized equipment. From an environmental standpoint, the reduction in solvent usage and the elimination of waste streams associated with protecting group chemistry align with green chemistry principles. Lower waste generation simplifies effluent treatment and reduces the environmental compliance burden. The process produces a cleaner crude product, which facilitates easier crystallization and purification, further minimizing solvent waste during the final isolation. These factors collectively make the technology highly attractive for manufacturers seeking to expand capacity while adhering to increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cephalosporin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical rationale behind the process. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers provided reflect the consensus on best practices for handling beta-lactam intermediates and optimizing one-pot reactions for maximum efficiency and yield.
Q: What is the primary advantage of using DBU in this cephalosporin synthesis?
A: DBU acts as a non-nucleophilic base that forms a stable salt with 7-ADCA, preventing unwanted Delta-2 isomerization of the cephalosporin skeleton while facilitating efficient esterification.
Q: How does the one-pot method impact production costs?
A: By eliminating the isolation and purification of intermediate salts, the one-pot method significantly reduces solvent consumption, processing time, and waste generation, leading to substantial operational cost savings.
Q: Why are thioesters preferred as acylating agents in this process?
A: Thioesters, particularly 2-phenylthiazole thioesters, allow for direct acylation of aromatic amino groups without the need for complex protection and deprotection steps, streamlining the synthetic route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. The technology disclosed in patent CN1009732B exemplifies the type of innovative process chemistry that we specialize in translating from intellectual property to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of beta-lactam chemistry, including dedicated containment for potent compounds and rigorous QC labs that enforce stringent purity specifications. We are committed to delivering cephalosporin derivatives that meet the highest international standards, supporting our partners in bringing life-saving antibiotics to market faster and more cost-effectively.
We invite procurement leaders and R&D directors to explore how our expertise in one-pot synthesis can optimize your supply chain. By leveraging our technical capabilities, you can achieve significant operational efficiencies and secure a competitive advantage in the marketplace. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can support your long-term strategic goals. Let us partner with you to engineer a more resilient and profitable future for your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
