Advanced Synthesis Of Alkylsulfinylbenzamides And 1 2 Benzisothiazol 3 Ones For Commercial Scale Up
The pharmaceutical and agrochemical industries continuously demand high-purity intermediates that can be manufactured safely and economically. Patent CN1042530C introduces a groundbreaking methodology for the production of alkylsulfinylbenzamides and their subsequent conversion into 1,2-benzisothiazol-3-ones, which are critical scaffolds for antibacterial and antifungal agents. This technology addresses significant limitations in prior art by replacing hazardous oxidants with a controlled halogenation process in a heterogeneous solvent system. By leveraging phase-transfer catalysis, this invention enables the transformation of readily available halobenzamides into valuable sulfinyl derivatives with exceptional efficiency. For global procurement teams, this represents a shift towards more sustainable and cost-effective supply chains for complex fine chemical intermediates. The robustness of this chemical pathway ensures consistent quality and reliability, essential for maintaining uninterrupted production schedules in regulated environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkylsulfinylbenzamides relied heavily on oxidation methods using periodic acid or meta-chloroperbenzoic acid (m-CPBA), as illustrated in older literature references. These conventional reagents present substantial challenges for industrial application due to their high cost, inherent instability, and significant safety risks associated with handling strong oxidizers on a large scale. Furthermore, processes involving 2-(methylthio)benzoyl chloride often suffer from raw material stability issues, complicating storage and logistics for manufacturing facilities. The multi-step nature of traditional routes, frequently requiring isolation of unstable intermediates, leads to increased operational complexity and lower overall throughput. Consequently, these factors contribute to elevated production costs and extended lead times, creating bottlenecks for companies seeking reliable sources of high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in this patent utilizes a heterogeneous solvent system comprising water and water-immiscible organic solvents like toluene or dichloromethane. This method reacts halobenzamides directly with thioalkanols in the presence of a base and a phase-transfer catalyst, bypassing the need for unstable acid chlorides. The subsequent oxidation step employs simple halogens such as bromine or chlorine, which are far more economical and easier to handle than periodic acid. This strategic shift not only simplifies the reaction workflow but also enhances safety profiles significantly. The ability to conduct these reactions under relatively mild conditions, typically between 0°C and 150°C, allows for greater flexibility in process engineering. This innovation paves the way for substantial cost reduction in pharmaceutical intermediate manufacturing by streamlining the synthetic route and minimizing waste disposal requirements.
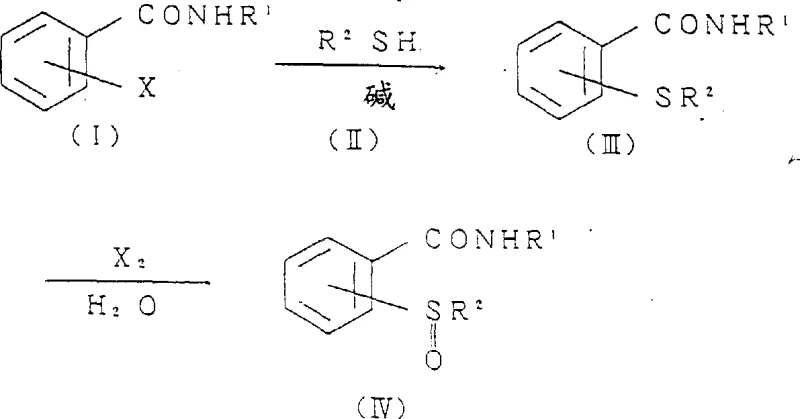
Mechanistic Insights into Phase-Transfer Catalyzed Halogenation
The core of this technological advancement lies in the efficient nucleophilic substitution facilitated by phase-transfer catalysts such as tetra-n-butylammonium bromide. In this mechanism, the halobenzamide, which is insoluble in water, reacts with the thioalkanol salt generated in the aqueous phase at the interface of the heterogeneous solvent system. The catalyst shuttles the reactive anionic species into the organic phase, accelerating the formation of the alkylthiobenzamide intermediate with high selectivity. Following this, the introduction of a halogen source allows for the controlled oxidation of the sulfur atom to the sulfinyl state without over-oxidation to the sulfone. This precision is critical for maintaining the integrity of the molecular structure required for downstream biological activity. Understanding this mechanistic nuance is vital for R&D directors aiming to replicate or adapt this chemistry for specific analog synthesis.
Furthermore, the process offers a direct route to 1,2-benzisothiazol-3-ones by reacting the 2-(alkylthio)benzamide intermediate with a halogenating agent. This cyclization step occurs smoothly under mild thermal conditions, avoiding the harsh environments typical of older methods. The reaction mechanism involves the formation of a sulfonium halide species which subsequently undergoes intramolecular nucleophilic attack by the amide nitrogen. This results in the closure of the heterocyclic ring with the elimination of hydrogen halide, which is neutralized by the base present in the system. The impurity profile is significantly improved as side reactions are minimized by the controlled addition of reagents and the use of specific solvent ratios. This level of control ensures that the final product meets stringent purity specifications required for active pharmaceutical ingredient (API) synthesis.

How to Synthesize Alkylsulfinylbenzamides Efficiently
To implement this synthesis effectively, manufacturers must first prepare the reaction vessel with a mixture of water and an organic solvent such as toluene, ensuring a distinct biphasic system is established. The halobenzamide starting material is introduced along with a catalytic amount of a quaternary ammonium salt, followed by the slow addition of the thioalkanol and aqueous base solution while maintaining strict temperature control. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during scale-up operations.
- React halobenzamide with thioalkanol in a heterogeneous solvent system containing a base and phase transfer catalyst to form alkylthiobenzamide.
- Oxidize the resulting alkylthiobenzamide with a halogen in a heterogeneous solvent to produce alkylsulfinylbenzamide with high selectivity.
- Cyclize the 2-(alkylthio)benzamide intermediate directly with a halogenating agent to obtain 1,2-benzisothiazol-3-one derivatives in a one-pot process.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits for supply chain stability and cost management. By eliminating the dependency on expensive and hazardous oxidizing agents, manufacturers can achieve significant cost savings in raw material procurement. The use of common halogens and inorganic bases reduces the volatility of input costs, allowing for more predictable budgeting and pricing strategies for long-term contracts. Additionally, the heterogeneous nature of the reaction simplifies work-up procedures, as the product can often be isolated directly from the organic layer while the aqueous phase containing the catalyst is recycled. This reduces the volume of wastewater generated and lowers environmental compliance costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The replacement of high-cost reagents like periodic acid with economical halogens drastically lowers the bill of materials for each production batch. Moreover, the ability to recycle the aqueous phase containing the phase-transfer catalyst minimizes the consumption of auxiliary chemicals, further driving down operational expenses. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as by-products. These factors combine to create a leaner manufacturing process that enhances profit margins without compromising on product quality or safety standards.
- Enhanced Supply Chain Reliability: Sourcing stable and safe raw materials is a critical priority for supply chain heads, and this method utilizes widely available commodity chemicals like bromine and sodium hydroxide. This reduces the risk of supply disruptions associated with specialized or regulated reagents. The robustness of the reaction conditions allows for flexible scheduling and faster turnaround times, ensuring that delivery commitments to downstream pharmaceutical clients are met consistently. The simplified purification process also shortens the overall production cycle time, enabling quicker response to market demand fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as the exothermic nature of the halogenation can be managed effectively through controlled dosing in a heterogeneous system. The reduction in hazardous waste streams simplifies disposal protocols and reduces the environmental footprint of the manufacturing facility. Compliance with increasingly strict environmental regulations is easier to achieve when avoiding heavy metal catalysts or persistent organic pollutants. This sustainability advantage strengthens the market position of suppliers adopting this technology, appealing to eco-conscious partners in the global value chain.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity for potential partners. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of this patent process over conventional periodic acid methods?
A: This process eliminates the need for expensive and dangerous oxidants like periodic acid or m-CPBA, utilizing safer halogens and base systems instead, which significantly reduces raw material costs and safety hazards.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method employs a heterogeneous solvent system that facilitates easy separation of products and allows for the recycling of the aqueous phase containing the phase transfer catalyst, making it highly scalable and economically viable.
Q: What kind of yields can be expected from this halogenation and cyclization route?
A: The patent data demonstrates consistently high yields, often exceeding 90 percent for the final 1,2-benzisothiazol-3-one products, ensuring efficient material utilization and minimal waste generation during manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkylsulfinylbenzamides Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing complex reactions like the phase-transfer catalyzed halogenation described in CN1042530C to meet stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure every batch of alkylsulfinylbenzamides and 1,2-benzisothiazol-3-ones conforms to the highest industry standards. Our commitment to quality and safety makes us an ideal partner for multinational corporations seeking a dependable source of critical pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency. Let us help you secure a competitive advantage through superior chemical synthesis and reliable delivery performance.
