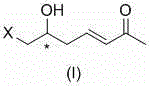
Scalable Synthesis of Optically Active 7-Halo-6-Hydroxyhept-3-En-2-One for Statin Manufacturing
The pharmaceutical industry's relentless pursuit of efficient pathways for statin production has identified a critical breakthrough in the synthesis of key chiral building blocks. Patent CN103145540A details a robust and economically viable preparation method for optically active 7-halogenated-6-hydroxyhept-3-en-2-one, a pivotal intermediate in the asymmetric synthesis of blockbuster drugs like Atorvastatin and Rosuvastatin. This technology represents a significant paradigm shift from traditional transition-metal catalyzed routes, offering a streamlined approach that leverages ozonolysis and Horner-Wadsworth-Emmons (HWE) chemistry. By utilizing readily available optically active 5-halo-4-hydroxy-1-pentene as a starting material, the process achieves total yields exceeding 90% under remarkably mild conditions. For R&D directors and procurement strategists, this patent outlines a pathway that not only ensures high stereochemical fidelity but also drastically simplifies the supply chain by removing dependency on precious metal catalysts. The structural integrity and functional group compatibility of this intermediate make it an ideal candidate for downstream elaboration into complex lipid-lowering agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon skeleton required for statin side chains has relied heavily on olefin metathesis reactions catalyzed by ruthenium complexes, specifically the second-generation Hoveyda-Grubbs catalyst. While chemically elegant, this conventional approach presents severe bottlenecks for industrial application. The primary constraint is the exorbitant cost of the catalyst itself, which contains precious ruthenium, coupled with the difficulty in recovering and recycling the metal from the reaction mixture. Furthermore, these metathesis reactions demand rigorous exclusion of moisture and oxygen, necessitating specialized equipment and inert atmosphere operations that drive up capital expenditure and operational complexity. The sensitivity of the catalyst often leads to variable batch-to-batch consistency, and the removal of trace metal residues to meet pharmaceutical standards requires additional, costly purification steps. These factors collectively render the traditional metathesis route economically unfeasible for multi-ton commercial production, creating a pressing need for a more robust alternative.
The Novel Approach
The methodology disclosed in the patent circumvents these challenges by employing a two-step sequence that relies on abundant, non-precious reagents. The novel approach initiates with the oxidative cleavage of a chiral terminal alkene via ozonolysis, a transformation that is highly atom-economical and scalable. This is followed by a Horner-Wadsworth-Emmons olefination using simple phosphonate esters, which proceeds efficiently under basic conditions. Unlike the sensitive metathesis catalysts, the reagents used here—such as sodium sulfite, dimethyl sulfide, and potassium tert-butoxide—are commodity chemicals with stable supply chains. The reaction conditions are notably mild, operating between -80°C and 50°C, which reduces energy consumption and safety risks associated with high-temperature processes. This strategic pivot from noble metal catalysis to classical organic transformations ensures that the process is not only chemically efficient but also commercially resilient, offering a reliable [Pharmaceutical Intermediates] supplier solution for long-term manufacturing needs.
Mechanistic Insights into Ozonolysis and HWE Olefination
The core of this synthetic strategy lies in the precise control of oxidation states and carbon-carbon bond formation while preserving the chiral information encoded in the starting material. The first step involves the ozonolysis of optically active 5-halo-4-hydroxy-1-pentene in a solvent like dichloromethane at temperatures ranging from -80°C to -20°C. Ozone selectively attacks the terminal double bond to form a molozonide, which rearranges to a stable ozonide. Crucially, the subsequent reductive workup using agents like sodium thiosulfate or dimethyl sulfide cleaves the ozonide to yield the corresponding aldehyde, optically active 4-halo-3-hydroxybutyraldehyde, without racemizing the adjacent stereocenter. The mildness of the reductive quench is vital, as harsher reducing conditions could compromise the halogen or hydroxyl functionalities. This step effectively shortens the carbon chain while installing the reactive aldehyde handle necessary for the subsequent coupling.
Following the isolation of the chiral aldehyde, the second mechanistic phase employs the Horner-Wadsworth-Emmons reaction to extend the carbon framework. The aldehyde reacts with a 2-oxopropyl phosphate ester in the presence of a base such as potassium tert-butoxide or potassium carbonate. The base deprotonates the alpha-carbon of the phosphonate to generate a nucleophilic carbanion, which attacks the carbonyl carbon of the aldehyde. The resulting betaine intermediate collapses to eliminate a phosphate byproduct, forming the desired alpha,beta-unsaturated ketone with high E-selectivity. This mechanism is particularly advantageous because the byproducts are water-soluble phosphates, facilitating easy separation during aqueous workup. The preservation of optical purity throughout this sequence is paramount, as the chiral center at the C6 position (relative to the final ketone) dictates the biological activity of the final statin drug. The use of mild bases and moderate temperatures ensures that no epimerization occurs, delivering a high-purity [Pharmaceutical Intermediates] product ready for further enzymatic or chemical reduction.
How to Synthesize Optically Active 7-Halo-6-Hydroxyhept-3-En-2-One Efficiently
The execution of this synthesis requires careful attention to temperature control during the ozonolysis step and stoichiometry during the HWE coupling to maximize yield and purity. The process is designed to be telescoped where possible, minimizing the handling of the unstable aldehyde intermediate. Operators should utilize standard industrial reactors equipped for low-temperature gas-liquid reactions and ensure efficient mixing to manage the exotherm of the ozone addition. The choice of solvent plays a critical role, with dichloromethane preferred for the oxidation step due to its ability to dissolve ozone and the organic substrates effectively, while tetrahydrofuran or acetonitrile is optimal for the subsequent condensation reaction. Detailed standardized operating procedures regarding the quenching of excess ozone and the neutralization of basic waste streams are essential for safe scale-up. For a comprehensive breakdown of the specific reaction parameters and workup protocols, please refer to the technical guide below.
- Perform ozonolysis on optically active 5-halo-4-hydroxy-1-pentene in dichloromethane at low temperatures (-80°C to -20°C) followed by reductive workup.
- Isolate the resulting optically active 4-halo-3-hydroxybutyraldehyde intermediate without extensive purification to maintain yield.
- Conduct Horner-Wadsworth-Emmons reaction with 2-oxopropyl phosphate esters using alkali bases in THF or acetonitrile to form the final enone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers transformative benefits that directly impact the bottom line and supply chain stability for generic and proprietary drug manufacturers. By shifting away from precious metal catalysis, the method eliminates a major cost driver and a significant supply risk associated with fluctuating ruthenium prices. The reliance on commodity chemicals means that raw material sourcing is decentralized and robust, reducing the likelihood of production stoppages due to reagent shortages. Furthermore, the simplified purification profile, driven by the formation of water-soluble byproducts, reduces the consumption of chromatography media and solvents, leading to substantial cost savings in downstream processing. This efficiency translates into a more competitive pricing structure for the final intermediate, enabling cost reduction in [Pharmaceutical Intermediates] manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of the second-generation Hoveyda-Grubbs catalyst removes the need for expensive noble metals and the complex ligand systems associated with them. This substitution with inexpensive phosphonate esters and inorganic bases drastically lowers the bill of materials. Additionally, the avoidance of strict anhydrous and oxygen-free environments reduces the engineering controls required, lowering both capital investment and utility costs. The high overall yield of over 90% minimizes raw material waste, further enhancing the economic viability of the process for large-scale production runs.
- Enhanced Supply Chain Reliability: The starting materials, such as optically active epoxyhalopropanes and simple phosphates, are widely available from multiple global suppliers, ensuring a continuous and reliable [Pharmaceutical Intermediates] supplier network. The process does not depend on single-source specialty catalysts that often have long lead times and limited availability. This diversification of the supply base mitigates the risk of bottlenecks and allows for flexible procurement strategies. The robustness of the reaction conditions also means that the process can be transferred between manufacturing sites with minimal validation overhead, ensuring business continuity.
- Scalability and Environmental Compliance: The use of recyclable solvents like dichloromethane and ethyl acetate, combined with the generation of non-toxic phosphate salts as waste, aligns well with modern environmental regulations. The mild reaction temperatures reduce the energy footprint of the manufacturing process, contributing to sustainability goals. The scalability is proven by the wide operating windows for temperature and time, allowing the process to be safely expanded from pilot plant to multi-ton commercial production. This ease of scale-up ensures that reducing lead time for high-purity [Pharmaceutical Intermediates] is achievable even during periods of surging market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational parameters detailed in the patent literature, providing clarity for technical teams evaluating this technology. Understanding these nuances is critical for assessing the feasibility of integrating this intermediate into existing production lines. The answers reflect the consensus on best practices for handling chiral halogenated intermediates and optimizing the HWE reaction conditions.
Q: What are the advantages of this method over Grubbs catalyst metathesis?
A: This method eliminates the need for expensive second-generation Hoveyda-Grubbs catalysts, avoids strict anhydrous/oxygen-free conditions, and utilizes cheap, recyclable solvents, significantly reducing production costs.
Q: What is the optical purity of the final product?
A: The process starts with optically active raw materials and maintains stereochemical integrity throughout the mild ozonolysis and HWE steps, ensuring high optical purity suitable for asymmetric synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the reaction conditions are mild (-80°C to 50°C), use common industrial solvents like DCM and THF, and achieve total yields over 90%, making it highly scalable for commercial API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Halo-6-Hydroxyhept-3-En-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral intermediates in the global pharmaceutical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering this optically active intermediate with stringent purity specifications, leveraging our rigorous QC labs to verify optical rotation and chemical purity at every stage. Our infrastructure is designed to handle the specific safety requirements of ozonolysis and the precise stoichiometry needed for HWE reactions, guaranteeing a consistent supply of material that meets the exacting standards of the statin market.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your manufacturing costs and supply security. By requesting a Customized Cost-Saving Analysis, you can quantify the economic benefits of switching to this non-precious metal route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development pipeline remains robust and competitive in a dynamic market environment.
