Advanced Cobalt Coordination Compound Synthesis for High-Efficiency Catalytic Applications
Advanced Cobalt Coordination Compound Synthesis for High-Efficiency Catalytic Applications
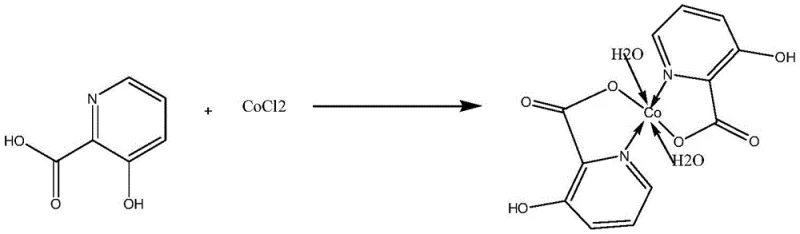
The landscape of transition metal catalysis is continuously evolving, driven by the demand for more efficient and selective catalysts in the synthesis of fine chemicals and pharmaceutical intermediates. A significant advancement in this field is documented in patent CN108840821B, which details the preparation and synthesis of a novel cobalt complex, specifically the dihydrate bis[3-hydroxy-2-pyridinecarboxylic acid]cobalt(II) complex. This coordination compound represents a strategic development in metal-organic chemistry, offering a robust platform for catalyzing key organic transformations such as nitrile silylation and amide bond formation. For R&D directors and procurement specialists in the fine chemical sector, understanding the structural integrity and synthetic accessibility of this complex is paramount. The patent outlines a streamlined methodology that bypasses the need for intricate ligand design, utilizing readily available 3-hydroxy-2-pyridine carboxylic acid to form a stable, crystalline catalyst that exhibits promising conversion rates in model reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the development of effective cobalt-based catalysts has often been hindered by the complexity of ligand synthesis and the instability of the resulting metal centers under reaction conditions. Many conventional protocols require multi-step organic synthesis to generate specialized chiral or achiral ligands, which significantly increases the cost of goods sold (COGS) and extends the lead time for catalyst production. Furthermore, older methods frequently involve harsh reaction conditions or toxic solvents that complicate waste management and pose safety risks in a manufacturing environment. The purification of these traditional catalysts can also be problematic, often requiring chromatography or recrystallization from difficult solvent systems, which reduces overall yield and operational efficiency. These factors collectively create bottlenecks in the supply chain for high-purity catalytic materials needed for API intermediate manufacturing.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the method described in patent CN108840821B introduces a remarkably simple and direct synthesis pathway. The process involves a straightforward one-step coordination reaction between 3-hydroxy-2-pyridine carboxylic acid and cobalt chloride in anhydrous methanol. By heating the mixture to reflux for 48 hours, the system achieves thermodynamic equilibrium, allowing for the spontaneous self-assembly of the coordination sphere around the cobalt ion. This approach eliminates the need for pre-functionalized ligands and avoids the use of exotic reagents. The resulting product precipitates as high-quality crystals upon natural volatilization of the solvent, facilitating easy isolation through simple filtration and washing with non-polar solvents like petroleum ether. This operational simplicity translates directly into reduced processing time and lower energy consumption.
Mechanistic Insights into Cobalt-Catalyzed Organic Transformations
The efficacy of this cobalt complex stems from its unique coordination geometry and electronic properties. Structural analysis reveals that the cobalt center is coordinated by nitrogen and oxygen atoms from the pyridine-carboxylate ligands, forming a stable chelate ring system. This N,O-chelation mode enhances the Lewis acidity of the metal center while protecting it from premature deactivation or aggregation. In the context of nitrile silylation, the cobalt complex likely acts as a Lewis acid catalyst, activating the carbonyl group of the aldehyde towards nucleophilic attack by trimethylsilyl cyanide (TMSCN). The specific arrangement of the ligands creates a steric environment that favors the formation of the silylated product, achieving a conversion rate of 68% under mild conditions. This mechanistic pathway highlights the potential of the complex to facilitate carbon-carbon bond-forming reactions essential for building complex molecular scaffolds in drug discovery.
Beyond silylation, the complex demonstrates versatile catalytic activity in condensation reactions, specifically between benzaldehyde and benzamide. In this transformation, the catalyst promotes the formation of amide bonds, a fundamental linkage in peptide chemistry and polymer science. The observed conversion rate of 88% indicates a high turnover frequency and robustness of the catalyst in the presence of nucleophilic amines. The stability of the complex is further evidenced by its thermal properties, with a melting point exceeding 220°C, suggesting it can withstand elevated temperatures often required in industrial batch processes. This dual functionality makes it a valuable tool for synthetic chemists looking to diversify their catalytic toolkit without investing in multiple specialized metal systems.

How to Synthesize Dihydrate Bis[3-hydroxy-2-pyridinecarboxylic acid]Cobalt(II) Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The procedure relies on standard glassware and heating equipment, making it accessible for most chemical manufacturing facilities. The key to success lies in maintaining anhydrous conditions during the initial dissolution and ensuring sufficient reflux time to drive the coordination equilibrium to completion. The crystallization step is critical; allowing the filtrate to volatilize slowly ensures the growth of large, well-defined crystals which are easier to filter and wash free of unreacted starting materials. Detailed standardized synthesis steps follow below for technical implementation.
- Dissolve 3-hydroxy-2-pyridine carboxylic acid in anhydrous methanol within a round-bottom flask under stirring.
- Add cobalt chloride to the solution and heat the mixture to reflux for 48 hours to ensure complete coordination.
- Filter the hot reaction solution, allow the filtrate to volatilize naturally to form crystals, then wash and vacuum dry the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cobalt complex synthesis method offers substantial advantages for supply chain optimization and cost management. The reliance on commodity chemicals—specifically 3-hydroxy-2-pyridine carboxylic acid and cobalt chloride—mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Since these precursors are produced on a large scale globally, their pricing remains stable and predictable, allowing for accurate long-term budgeting. Furthermore, the elimination of complex purification steps such as column chromatography significantly reduces the consumption of silica gel and organic solvents, leading to a drastic reduction in waste disposal costs and environmental compliance burdens.
- Cost Reduction in Manufacturing: The one-step nature of the synthesis inherently lowers labor and utility costs. By avoiding multi-step ligand synthesis, manufacturers save on reactor time, intermediate isolation, and quality control testing for multiple intermediates. The high yield of 70% reported in the patent indicates an efficient atom economy, minimizing the loss of valuable cobalt resources. Additionally, the use of methanol as a solvent is economically favorable compared to more expensive aprotic solvents, and its recovery for reuse is straightforward via distillation. These factors combine to create a highly cost-effective production profile for the final catalyst.
- Enhanced Supply Chain Reliability: The robustness of the crystallization process ensures consistent product quality batch after batch. Unlike amorphous powders which may vary in bulk density and handling characteristics, crystalline materials offer superior flow properties and stability during storage and transport. This consistency is crucial for downstream users who require precise dosing in their catalytic reactions. The simplified workflow also reduces the likelihood of human error or process deviations, thereby enhancing the reliability of delivery schedules. Suppliers can maintain higher inventory levels with confidence, knowing that the production cycle is short and reproducible.
- Scalability and Environmental Compliance: Scaling this process from grams to tons involves linear expansion of reactor volume without the need for new technology or hazardous reagents. The absence of heavy metal contaminants other than the intended cobalt simplifies the regulatory approval process for use in pharmaceutical applications, provided residual levels are managed. The washing protocol using petroleum ether and hexane effectively removes organic impurities, ensuring high purity standards are met. Moreover, the solid nature of the final product facilitates safe packaging and shipping, reducing the risks associated with transporting liquid catalyst solutions. This scalability supports the growing demand for green and sustainable catalytic solutions in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this cobalt coordination compound. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation. They serve to clarify the operational parameters and potential benefits for industrial partners considering the integration of this catalyst into their processes.
Q: What are the primary catalytic applications of this cobalt complex?
A: This complex demonstrates significant catalytic activity in the nitrile silylation of benzaldehyde (68% conversion) and the condensation reaction between benzaldehyde and benzamide (88% conversion).
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes a simple one-step reflux in methanol with commercially available starting materials, making it highly amenable to scale-up without requiring complex purification techniques.
Q: What is the structural stability of the complex?
A: X-ray diffraction analysis confirms a stable dihydrate structure with robust Co-N and Co-O coordination bonds, ensuring thermal stability with a melting point exceeding 220°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cobalt Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalysts play in accelerating drug discovery and optimizing chemical manufacturing. Our team of expert chemists is well-versed in the nuances of metal-organic coordination chemistry and stands ready to support your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art rigorous QC labs guarantee that every batch of catalyst meets stringent purity specifications, providing you with the confidence to proceed with your synthesis campaigns without delay.
We invite you to explore the potential of this novel cobalt complex for your specific applications. Whether you require custom synthesis modifications or bulk supply agreements, our technical procurement team is equipped to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments. Let us partner with you to drive innovation and efficiency in your chemical supply chain, leveraging our deep technical expertise to solve your most challenging synthesis problems.
