Revolutionizing Glucal Production: A Technical Deep Dive into Scalable Manufacturing
The landscape of carbohydrate chemistry is constantly evolving, driven by the relentless demand for efficient access to key building blocks like glucal, a pivotal intermediate in the synthesis of complex glycosides and nucleoside analogs. Patent CN100579973C presents a significant technological breakthrough by introducing a novel one-pot preparation method that fundamentally alters the economic and operational dynamics of glucal manufacturing. Unlike traditional multi-step sequences that often suffer from low atom economy and cumbersome isolation procedures, this invention leverages a zinc-mediated reductive elimination strategy in an aqueous environment. This approach not only streamlines the synthetic pathway but also addresses critical pain points regarding environmental sustainability and cost-efficiency, making it a highly attractive option for a reliable pharmaceutical intermediates supplier seeking to optimize their production portfolio. The strategic implementation of this technology allows manufacturers to bypass the logistical complexities associated with handling sensitive organometallic reagents, thereby securing a more robust supply chain for high-value sugar derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glucal and its acetylated derivatives has relied heavily on methodologies that impose significant burdens on both research budgets and production timelines. Conventional routes frequently utilize expensive transition metal catalysts, such as titanocene dichloride or chromium-based reagents, which require stringent anhydrous conditions and inert atmosphere handling to prevent decomposition. As illustrated in prior art examples like the lithium perchlorate mediated elimination shown below, these processes often involve toxic organic solvents and generate substantial amounts of hazardous waste that require costly disposal protocols.
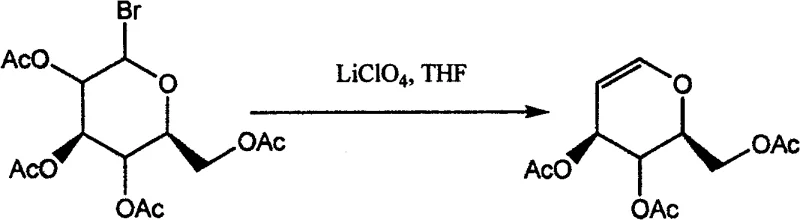
Furthermore, the necessity for cryogenic temperatures or prolonged reflux periods in these legacy methods increases energy consumption and limits the throughput capacity of manufacturing facilities. The separation of heavy metal residues from the final product adds another layer of complexity, often necessitating specialized chromatography or extensive washing steps that erode overall yield. For procurement managers, these factors translate into volatile pricing and potential supply disruptions, as the availability of high-purity specialty catalysts can be inconsistent. Consequently, the industry has long sought a more resilient alternative that maintains high stereochemical fidelity without the operational overhead of traditional organometallic chemistry.
The Novel Approach
The methodology disclosed in CN100579973C represents a paradigm shift by utilizing earth-abundant zinc powder in conjunction with simple phosphate buffers or sodium dihydrogen phosphate solutions. This innovative route effectively replaces costly and toxic reagents with commodity chemicals that are readily available on the global market, drastically simplifying the procurement process. The reaction proceeds smoothly at room temperature, eliminating the need for energy-intensive heating or cooling systems, which directly contributes to cost reduction in API manufacturing. By operating in an aqueous or semi-aqueous medium, the process inherently reduces the volume of volatile organic compounds (VOCs) released into the environment, aligning with modern green chemistry principles and regulatory compliance standards. This simplification of the reaction matrix not only accelerates the reaction kinetics but also facilitates easier workup procedures, allowing for higher recovery rates of the target molecule with minimal purification effort.
Mechanistic Insights into Zinc-Mediated Reductive Elimination
At the heart of this technological advancement lies a sophisticated yet elegant mechanistic pathway involving the single-electron transfer capabilities of activated zinc species. In the presence of copper sulfate or within a phosphate buffer system, the zinc surface facilitates the reductive cleavage of the carbon-bromine bond at the anomeric position of the 1-bromoacetylglucose substrate. This initial activation generates a transient organozinc intermediate or a radical species that rapidly undergoes beta-elimination to form the characteristic double bond of the glucal scaffold. The presence of copper ions acts as a crucial mediator, potentially enhancing the electron transfer rate and preventing the passivation of the zinc surface, which ensures consistent reaction performance throughout the batch cycle. Understanding this catalytic cycle is essential for R&D directors aiming to replicate these results, as the precise ratio of zinc to copper and the pH of the buffer solution play pivotal roles in controlling the reaction trajectory and minimizing side reactions such as hydrogenation or epimerization.
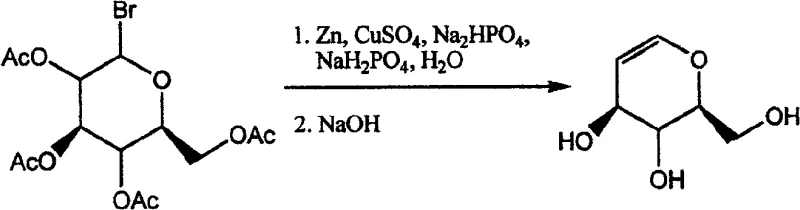
Following the formation of the acetylated glucal intermediate, the process seamlessly transitions into a deacetylation phase through the addition of a strong base such as sodium hydroxide or potassium hydroxide. This tandem operation is critical for impurity control, as performing the deprotection in situ avoids the isolation of unstable acetylated intermediates that might degrade upon storage or exposure to atmospheric moisture. The mild basic conditions employed are sufficient to cleave the ester bonds without compromising the integrity of the enol ether functionality, which is notoriously sensitive to acid-catalyzed hydrolysis. This careful balance of reactivity ensures that the final product exhibits a superior purity profile, with significantly reduced levels of halogenated byproducts or over-reduced sugar alcohols. For quality assurance teams, this inherent selectivity reduces the burden on analytical testing and simplifies the specification release process for commercial batches.
How to Synthesize Glucal Efficiently
To implement this high-yielding synthesis route effectively, operators must adhere to a precise sequence of reagent addition and mixing parameters that maximize the interaction between the solid zinc surface and the dissolved substrate. The patent outlines a robust protocol where the stoichiometry of zinc powder relative to the bromo-sugar is maintained at a specific weight ratio to ensure complete conversion while minimizing excess metal waste. Detailed standard operating procedures regarding the preparation of the phosphate buffer and the controlled addition of the base are critical for reproducibility on a larger scale. The following guide summarizes the essential steps derived from the experimental examples to assist technical teams in adopting this methodology.
- Prepare a phosphate buffer solution containing zinc powder and copper sulfate, or alternatively use an aqueous sodium dihydrogen phosphate solution with zinc powder.
- Introduce 1-bromoacetylglucose to the reaction mixture and stir at room temperature for 0.5 to 8 hours to facilitate reductive elimination.
- Add sodium hydroxide or potassium hydroxide to the reaction liquid and stir for an additional 0.5 to 3 hours to complete deacetylation and isolate the final glucal product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this zinc-mediated synthesis offers profound benefits that extend far beyond the laboratory bench, directly impacting the bottom line of chemical manufacturing enterprises. The substitution of precious metal catalysts with inexpensive zinc powder results in a dramatic decrease in raw material expenditures, allowing for more competitive pricing structures in the global marketplace. Moreover, the elimination of complex solvent recovery systems and hazardous waste treatment protocols associated with heavy metals leads to substantial cost savings in operational overhead. For supply chain heads, the reliance on universally available commodities like zinc and phosphate salts mitigates the risk of supply disruptions caused by geopolitical tensions or niche vendor shortages, ensuring a steady flow of materials for continuous production schedules.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the drastic simplification of the reagent list. By removing the dependency on specialized organometallic catalysts that require cold chain shipping and inert storage, companies can significantly reduce their inventory holding costs and logistics expenses. The ability to run the reaction at ambient temperature further lowers utility bills related to heating and cooling, contributing to a leaner manufacturing footprint. Additionally, the high yields reported in the patent examples imply that less starting material is wasted, optimizing the overall material balance and maximizing the return on investment for every kilogram of 1-bromoacetylglucose processed.
- Enhanced Supply Chain Reliability: Stability in the supply of critical intermediates is paramount for downstream pharmaceutical production, and this method enhances reliability by decoupling production from fragile supply chains. Since the key reagents are bulk chemicals produced by multiple vendors worldwide, procurement managers are not locked into single-source agreements that can lead to price gouging or delivery delays. The robustness of the aqueous reaction system also means that production is less susceptible to fluctuations in ambient conditions, reducing the likelihood of batch failures due to environmental factors. This resilience translates into shorter lead times for high-purity pharmaceutical intermediates, enabling customers to maintain lower safety stock levels and improve their cash flow efficiency.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but the simplicity of this one-pot design facilitates a smooth transition from pilot plant to full commercial scale. The absence of exothermic spikes or gas evolution hazards simplifies reactor design and safety engineering, allowing for larger batch sizes without proportional increases in risk. Furthermore, the reduced generation of toxic waste aligns with increasingly stringent environmental regulations, protecting the manufacturer from potential fines and reputational damage. This eco-friendly profile is a significant asset when bidding for contracts with multinational corporations that prioritize sustainable sourcing and green chemistry initiatives in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucal synthesis technology, providing clarity for stakeholders evaluating its potential integration into their existing workflows. These insights are derived directly from the patent specifications and practical considerations for industrial application, ensuring that decision-makers have accurate information to assess feasibility. Understanding these nuances is vital for aligning R&D capabilities with commercial objectives and ensuring a successful technology transfer.
Q: What are the primary advantages of the zinc-mediated glucal synthesis method over traditional titanium-based routes?
A: The zinc-mediated method described in patent CN100579973C eliminates the need for expensive and moisture-sensitive transition metal catalysts like titanocene dichloride. This shift significantly reduces raw material costs and simplifies the operational requirements, as the reaction can proceed in aqueous buffer systems rather than strictly anhydrous organic solvents, thereby enhancing process safety and scalability.
Q: How does this one-pot methodology impact the impurity profile of the final glucal intermediate?
A: By combining the reductive elimination and deacetylation steps into a streamlined sequence, the exposure of reactive intermediates to harsh conditions is minimized. The use of mild room temperature conditions and aqueous workups helps prevent the formation of degradation byproducts often associated with prolonged heating or strong acidic conditions found in older methodologies, resulting in a cleaner crude product that requires less intensive purification.
Q: Is this synthesis route suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process is explicitly designed for industrial feasibility. The reliance on inexpensive, commodity-grade reagents such as zinc powder and phosphate salts, coupled with the ability to operate at room temperature without specialized high-pressure equipment, makes it highly adaptable for multi-kilogram to ton-scale manufacturing, ensuring consistent supply continuity for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glucal Supplier
As the demand for high-quality carbohydrate intermediates continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in navigating the complexities of modern chemical synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch without supply bottlenecks. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest international standards, guaranteeing the consistency required for regulatory filings. By leveraging our expertise in zinc-mediated chemistries and aqueous processing, we can help you realize the full cost and efficiency potential of this patented technology.
We invite you to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific volume requirements and quality targets. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits for your organization, and let us provide you with specific COA data and route feasibility assessments that demonstrate our capability to deliver. Together, we can optimize your supply chain and secure a competitive edge in the fast-paced pharmaceutical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
