Scalable Green Synthesis of Acetylated Vinyl Sugars for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Vinyl Sugar Manufacturing
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective synthetic routes. Patent CN101747304B introduces a groundbreaking methodology for the preparation of acetylated vinyl sugars, acetylated disaccharide vinyl sugars, and benzoylated vinyl sugars, which serve as critical building blocks in the synthesis of bioactive natural products and complex glycoconjugates. This innovation addresses the longstanding challenges associated with traditional glycal synthesis by leveraging a benign Zinc/PEG/Water system that operates efficiently at room temperature. For R&D directors and process chemists, this represents a paradigm shift away from harsh conditions toward a more pragmatic, green chemistry approach that maintains high stereochemical integrity while drastically simplifying the operational workflow. The ability to generate these valuable intermediates with such simplicity opens new avenues for the rapid assembly of O-glycosides, C-glycosides, and other pharmacologically active scaffolds essential for modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of vinyl sugars or glycals has relied heavily on elimination reactions starting from 1-substituted sugars, often necessitating the use of expensive reagents and toxic organic solvents that complicate downstream processing. Traditional protocols frequently involve strong bases, heavy metal catalysts, or anhydrous conditions that are not only costly to maintain but also pose significant safety and environmental hazards during scale-up. Furthermore, many existing methods suffer from moderate to low yields and require cumbersome purification steps to remove residual metals or solvent impurities, which can be detrimental to the quality of sensitive pharmaceutical intermediates. The reliance on volatile organic compounds (VOCs) also creates a substantial burden on waste management systems, increasing the overall cost of goods sold (COGS) and limiting the sustainability profile of the manufacturing process. These factors collectively create a bottleneck for procurement teams seeking reliable sources of high-purity glycals without incurring excessive environmental compliance costs.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a remarkably simple system comprising Zinc powder, Polyethylene Glycol (PEG), and water to effect the reductive elimination of acetylated bromo-sugars. This method eliminates the need for expensive transition metal catalysts or strictly anhydrous environments, allowing the reaction to proceed smoothly at room temperature with impressive reaction rates. The use of PEG as a phase-transfer-like medium in conjunction with water creates a unique reaction environment that facilitates the interaction between the solid zinc surface and the organic substrate, leading to high conversion efficiencies. As illustrated in the reaction scheme below, this transformation is broadly applicable to various monosaccharides and disaccharides, providing a unified platform for glycal synthesis.
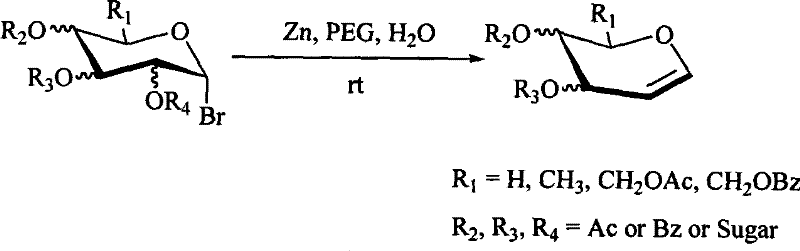
The operational simplicity of adding zinc powder to a PEG-water slurry means that specialized equipment is not required, making this technology immediately transferable to existing manufacturing facilities. For supply chain managers, this translates to a robust process that is less susceptible to disruptions caused by the scarcity of exotic reagents, ensuring a steady flow of critical intermediates for downstream API synthesis.
Mechanistic Insights into Zinc-Mediated Reductive Elimination
The core of this technological advancement lies in the zinc-mediated reductive elimination mechanism, which effectively removes the anomeric bromine and the adjacent acetoxy group to form the characteristic C1-C2 double bond of the vinyl sugar. In this system, zinc acts as a single-electron donor, initiating a radical or organozinc intermediate at the anomeric center which subsequently undergoes elimination. The presence of PEG likely stabilizes the transition state and enhances the solubility of the organic substrate within the aqueous medium, thereby accelerating the reaction kinetics without the need for external heating. This mechanistic pathway is distinct from base-mediated eliminations that often lead to side reactions or degradation of sensitive protecting groups, thus preserving the integrity of the sugar scaffold. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters for specific substrates, as the electronic nature of the protecting groups (acetyl vs. benzoyl) can influence the rate of elimination.
From an impurity control perspective, this mechanism offers significant advantages by minimizing the formation of byproducts commonly associated with harsh acidic or basic conditions. The mildness of the zinc reduction ensures that other labile functional groups on the sugar ring remain intact, resulting in a cleaner crude reaction profile that simplifies purification. This high level of chemoselectivity is paramount for the production of pharmaceutical intermediates where impurity profiles are strictly regulated. By avoiding the use of strong bases, the risk of epimerization or hydrolysis of the ester protecting groups is substantially reduced, leading to higher overall yields of the desired stereoisomer. This precision in synthesis directly supports the R&D goal of achieving high-purity standards required for clinical grade materials.
How to Synthesize Acetylated Vinyl Sugars Efficiently
The practical implementation of this synthesis route is designed to be accessible and straightforward, requiring minimal specialized training or equipment. The protocol involves dissolving the acetylated bromo-sugar substrate in a mixture of PEG and water, followed by the addition of zinc powder and stirring at ambient temperature. Reaction monitoring is easily achieved via thin-layer chromatography (TLC), allowing for precise determination of the endpoint to prevent over-reaction. Detailed standardized synthetic steps for specific substrates like acetylated glucal or galactal are outlined in the structured guide below, ensuring reproducibility across different batches and scales.
- Dissolve the acetylated bromo-sugar substrate (e.g., acetylated bromoglucose) in a mixture of Polyethylene Glycol (PEG-400, 600, or 2000) and water within a reaction vessel.
- Add activated Zinc powder to the reaction mixture at room temperature and stir continuously to initiate the reductive elimination process.
- Monitor reaction progress via TLC until completion (typically 20 minutes to 6 hours), then purify the resulting vinyl sugar product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Zn/PEG/H2O methodology presents a compelling value proposition centered on cost efficiency and operational resilience. The shift away from expensive noble metal catalysts and volatile organic solvents to commodity chemicals like zinc and PEG fundamentally alters the cost structure of vinyl sugar production. This reduction in raw material complexity directly contributes to a more stable pricing model for these critical intermediates, shielding buyers from the volatility often seen in the specialty chemical market. Furthermore, the simplified workflow reduces the labor hours required for reaction setup and monitoring, allowing manufacturing teams to allocate resources more effectively across other projects.
- Cost Reduction in Manufacturing: The elimination of costly transition metal catalysts and the replacement of expensive organic solvents with a PEG-water system leads to substantial cost savings in raw material procurement. Additionally, the ability to run reactions at room temperature removes the energy costs associated with heating or cooling large reactors, further driving down the operational expenditure. The simplified purification process, resulting from a cleaner reaction profile, also reduces the consumption of silica gel and eluents, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: By relying on widely available and inexpensive reagents such as zinc powder and polyethylene glycol, the supply chain becomes significantly more robust against disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the inputs for this process are commodity chemicals with stable global availability. This reliability ensures consistent production schedules and reduces the risk of stockouts for downstream API manufacturers who depend on a steady supply of high-quality glycal intermediates.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium drastically simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The absence of halogenated solvents and heavy metals means that wastewater treatment is less complex and costly, facilitating easier permitting for scale-up operations. This green chemistry profile not only enhances the corporate sustainability image but also future-proofs the manufacturing process against tightening environmental legislation, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this zinc-mediated synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows.
Q: What types of sugar substrates are compatible with this Zn/PEG reduction method?
A: The method is highly versatile, successfully converting various acetylated and benzoylated bromo-sugars including glucose, mannose, galactose, arabinose, and disaccharides like maltose, lactose, and cellobiose into their corresponding vinyl sugar derivatives with high efficiency.
Q: How does this process improve environmental compliance compared to traditional methods?
A: By utilizing a PEG-water solvent system instead of volatile organic compounds (VOCs) and employing non-toxic zinc powder, the process significantly reduces hazardous waste generation and eliminates the need for complex solvent recovery systems, aligning with strict green chemistry standards.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the procedure operates at room temperature with simple stirring and inexpensive reagents, removing the need for cryogenic conditions or expensive transition metal catalysts, which makes it inherently scalable for industrial manufacturing of glycal intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinyl Sugar Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient glycal synthesis in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative Zn/PEG protocol can be fully realized at an industrial level. We are committed to delivering high-purity vinyl sugar intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to green chemistry principles allows us to offer sustainable solutions that do not compromise on quality or performance.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this greener synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both cost and reliability in the competitive pharmaceutical landscape.
