Revolutionizing Sucralose-6-Ester Purification: A Technical Deep Dive into Yield Optimization and Cost Reduction
Revolutionizing Sucralose-6-Ester Purification: A Technical Deep Dive into Yield Optimization and Cost Reduction
The global demand for high-intensity sweeteners continues to drive innovation in the manufacturing of sucralose, a cornerstone ingredient in the food and beverage industry. At the heart of this production lies the critical intermediate, sucralose-6-ester, where purity and yield directly dictate the economic viability of the final API. Recent advancements detailed in patent CN113939524A introduce a transformative approach to purifying this intermediate, specifically targeting the notoriously difficult repeated mother liquors that have long plagued manufacturers. This technology shifts the paradigm from simple physical separation to active chemical conversion, offering a robust solution for recovering valuable product from waste streams. By integrating selective oxidation with controlled alkaline hydrolysis, this method addresses the persistent challenges of low separation efficiency and high energy consumption associated with conventional purification techniques. For R&D leaders and supply chain strategists, understanding this mechanistic breakthrough is essential for optimizing production lines and securing a competitive edge in the sweetener intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of sucralose-6-ester from repeated crystallization mother liquors has been a bottleneck characterized by diminishing returns and escalating operational costs. The traditional industry standard involves multiple cycles of water boiling extraction, followed by organic solvent extraction and recrystallization. This physical separation approach suffers from inherent thermodynamic limitations; as the concentration of the target ester decreases in the mother liquor, the solubility dynamics favor the retention of the product in the aqueous waste phase rather than the organic extractant. Consequently, manufacturers face significant product loss, with substantial quantities of sucralose-6-ester ending up in final waste liquids. Furthermore, the process is energy-intensive, requiring repeated heating and concentration steps that strain utility budgets. The accumulation of polychlorinated byproducts, such as sucralose diester and tetrachlorosucrose-6-ester, further complicates the matrix, leading to darker colors, higher viscosity, and difficult downstream processing that increases environmental compliance burdens.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN113939524A employs a proactive chemical strategy that converts these stubborn impurities directly into the desired sucralose-6-ester. Instead of attempting to physically separate the target molecule from structurally similar byproducts, this novel route utilizes hydrogen peroxide as a selective oxidant in conjunction with an alkaline hydrolysis agent. This dual-action system effectively cleaves specific functional groups on the impurity molecules—such as extra ester groups or chlorine atoms—transforming them into the target mono-ester structure. This chemical conversion fundamentally alters the mass balance of the process, turning what was previously considered waste into valuable inventory. The result is a streamlined workflow that not only simplifies the purification sequence but also dramatically enhances the overall yield of the production campaign. By mitigating the need for excessive boiling and multiple extraction cycles, this approach offers a pathway to significantly lower operating expenditures and a reduced environmental footprint.
Mechanistic Insights into Oxidative Alkaline Hydrolysis
The core of this technological advancement lies in the precise orchestration of oxidation and hydrolysis reactions within the complex matrix of the mother liquor. The process initiates with the addition of hydrogen peroxide under controlled low-temperature conditions, typically ranging from -10°C to 20°C. During this phase, the oxidant selectively targets the susceptible bonds within the polychlorinated impurities, such as sucralose diester, forming reactive composite intermediates. This step is critical; the temperature must be maintained rigorously to prevent the over-oxidation of the already formed sucralose-6-ester, ensuring that the reaction remains selective for the higher-order impurities. Following the oxidation period, an alkaline hydrolysis agent, preferably dimethylamine or ammonia, is introduced to the mixture. This base serves a dual purpose: it neutralizes the acidic environment generated by the oxidation and facilitates the hydrolytic cleavage of the intermediate complexes. The pH is carefully adjusted to a range of 8.5 to 10.5, creating the optimal conditions for the removal of extraneous ester or chlorine groups, thereby reconstructing the target sucralose-6-ester molecular architecture.
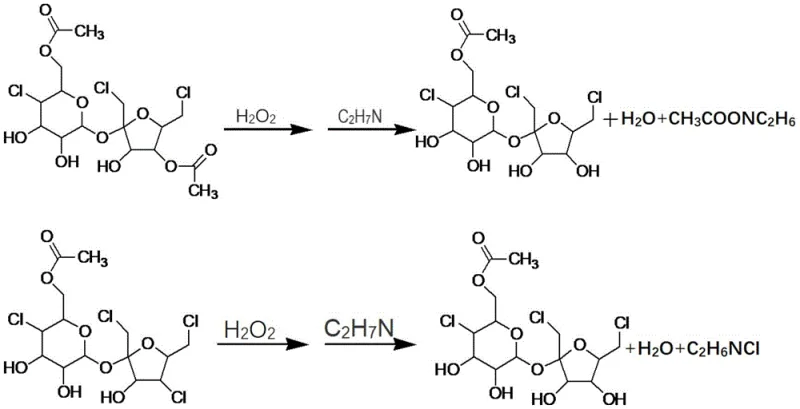
Following the conversion phase, the reaction mixture undergoes a critical workup sequence designed to isolate the newly formed product while removing insoluble polymeric byproducts. The pH of the solution is adjusted to neutrality, specifically between 6 and 8, using mineral acids like hydrochloric acid, which can often be sourced from upstream chlorination recycling streams. This neutralization step stabilizes the product and prepares the matrix for phase separation. Upon heating and filtration, insoluble colored impurities—often resulting from sucrose polymerization or coking during earlier chlorination stages—are effectively removed. The subsequent liquid-liquid extraction using ethyl acetate ensures that the regenerated sucralose-6-ester is efficiently transferred from the aqueous phase into the organic phase. This mechanistic understanding underscores the robustness of the process, demonstrating how careful control of reaction parameters can unlock value from waste streams that were previously deemed unrecoverable, providing a clear advantage for manufacturers seeking to maximize atom economy.
How to Synthesize Sucralose-6-Ester Efficiently
The implementation of this purification protocol requires strict adherence to the specified reaction conditions to ensure safety and maximum conversion efficiency. The process begins with the conditioning of the repeated mother liquor, often involving a pre-cooling step to manage the exothermic nature of the subsequent oxidation. Operators must monitor the addition rates of hydrogen peroxide and the alkaline agent closely, as deviations can lead to incomplete conversion or product degradation. The detailed standardized synthetic steps, including specific molar ratios, stirring speeds, and crystallization parameters, are outlined in the technical guide below to assist process engineers in replicating these results at scale.
- Impurity Conversion: Add hydrogen peroxide oxidant to the repeated mother liquor at low temperature (-10 to 20°C), followed by the addition of an alkaline hydrolysis agent like dimethylamine to convert sucralose diesters into the target ester.
- pH Adjustment and Filtration: Neutralize the reaction mixture to pH 6-8 using acid, maintain stability, heat to dissolve solubles, and filter out insoluble polymeric impurities.
- Extraction and Crystallization: Extract the aqueous phase with ethyl acetate, combine organic phases, concentrate under vacuum, and crystallize at 0-5°C to obtain high-purity sucralose-6-ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this oxidative purification technology represents a strategic opportunity to enhance margin stability and supply security. The primary economic driver is the substantial recovery of product from mother liquors that would otherwise be discarded or subjected to costly, low-yield reprocessing. By chemically converting impurities back into the saleable intermediate, manufacturers can effectively increase the output of their existing reactors without expanding capital infrastructure. This intensification of production capacity leads to a direct reduction in the cost of goods sold (COGS), as the fixed costs of production are amortized over a larger volume of high-purity product. Furthermore, the elimination of energy-intensive water boiling steps translates into lower utility consumption, aligning production costs with more sustainable operational models. These efficiencies create a buffer against raw material price volatility, offering a more resilient cost structure for long-term supply agreements.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of energy-intensive boiling steps significantly lower the operational expenditure profile of the facility. By recovering valuable intermediates from waste streams, the effective yield per batch is increased, which dilutes the fixed overhead costs across a larger production volume. This process optimization removes the need for expensive downstream polishing steps often required to meet purity specifications when using conventional extraction methods. Additionally, the ability to recycle upstream byproducts like dimethylamine further contributes to raw material cost savings, creating a closed-loop system that minimizes waste disposal fees and maximizes resource utilization.
- Enhanced Supply Chain Reliability: Implementing this high-yield purification method mitigates the risk of supply shortages caused by low batch yields or failed crystallizations common in traditional processes. The robustness of the chemical conversion ensures consistent output quality, reducing the variability that often disrupts just-in-time delivery schedules. By maximizing the recovery from every batch of chlorination mother liquor, manufacturers can maintain higher inventory levels of critical intermediates without needing to increase the frequency of hazardous chlorination runs. This stability allows for more predictable lead times and strengthens the reliability of the supply chain, ensuring that downstream API production is never halted due to intermediate shortages.
- Scalability and Environmental Compliance: The process is designed for seamless scale-up, utilizing standard unit operations such as stirred tank reactors and filtration systems that are commonplace in fine chemical manufacturing. The reduction in wastewater volume and the lowering of organic load in effluent streams significantly ease the burden on environmental treatment facilities. By converting chlorinated organic impurities into the target product rather than discharging them, the facility reduces its environmental liability and compliance risks. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational food and pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this oxidative purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route. Understanding these details is crucial for technical teams assessing the integration of this method into existing production workflows.
Q: How does this oxidative method improve yield compared to traditional water boiling?
A: Traditional methods rely on physical extraction which leaves significant product in the waste stream. This patented chemical conversion actively transforms impurities like sucralose diester back into the target sucralose-6-ester, drastically increasing total recovery from mother liquor.
Q: What are the critical reaction conditions for the oxidation step?
A: The oxidation reaction requires strict temperature control between -10°C and 20°C, preferably 0-10°C, using 15-40% hydrogen peroxide. Reaction times typically range from 1 to 12 hours to ensure complete conversion without over-oxidizing the target product.
Q: Why is dimethylamine preferred as the alkaline hydrolysis agent?
A: Dimethylamine is often available as a byproduct from the upstream DMF solvent recovery in sucralose synthesis. Using it here optimizes material utilization, reduces raw material costs, and effectively adjusts the pH to the required 8.5-10.5 range for de-esterification of impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sucralose-6-Ester Supplier
The technical potential of this oxidative purification route underscores the importance of partnering with a CDMO that possesses deep expertise in complex intermediate synthesis. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of sucralose-6-ester meets the exacting standards required for food-grade applications. We understand that the transition from laboratory innovation to industrial reality requires precise engineering and unwavering attention to detail, capabilities that are embedded in our operational DNA.
We invite you to engage with our technical procurement team to discuss how this advanced purification technology can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits this process offers your organization. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing excellence can drive value and efficiency in your sweetener production pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
