Advanced Photo-Induced Synthesis of Aromatic Vinyl Bromide Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign pathways for constructing complex molecular scaffolds. Patent CN111675650A introduces a groundbreaking preparation method for aromatic vinyl bromide derivatives, addressing critical bottlenecks in existing synthetic methodologies. This innovation leverages a photo-induced atom transfer radical addition (ATRA) strategy, utilizing visible blue light as a clean energy source to drive the bifunctionalization of alkynes. By replacing harsh chemical initiators with renewable light energy, this technology not only enhances the sustainability of the manufacturing process but also significantly broadens the scope of compatible substrates. For R&D directors and procurement specialists, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity, offering a robust solution for the production of high-value intermediates used in drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the bifunctionalization of alkynes to introduce bromine atoms has relied heavily on traditional Atom Transfer Radical Addition (ATRA) strategies that are fraught with significant operational and environmental drawbacks. Conventional processes typically necessitate the use of stoichiometric amounts of toxic and hazardous initiators, such as organic peroxides, organotin reagents, or trialkylboranes, to generate the required free radicals. Furthermore, these methods often demand high-intensity ultraviolet (UV) light or elevated temperatures, creating harsh reaction conditions that can degrade sensitive functional groups and limit substrate tolerance. The reliance on such aggressive reagents not only poses severe safety risks to personnel but also generates substantial toxic waste, complicating downstream purification and increasing the overall cost of waste disposal. Consequently, the economic benefits of these traditional routes are frequently eroded by the need for specialized containment equipment and extensive environmental remediation efforts.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111675650A offers a sophisticated and eco-friendly alternative that operates under remarkably mild conditions. This novel approach employs a catalytic system driven by visible blue light emitted from standard LEDs, eliminating the need for dangerous chemical initiators entirely. The reaction proceeds at room temperature under a nitrogen atmosphere, utilizing dimethyl sulfoxide (DMSO) as a solvent, which ensures excellent solubility for a wide range of organic substrates. By shifting the energy input from thermal or high-energy UV sources to visible light, the process minimizes side reactions and decomposition, thereby preserving the integrity of complex molecular architectures. This transition to a photo-catalytic regime not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
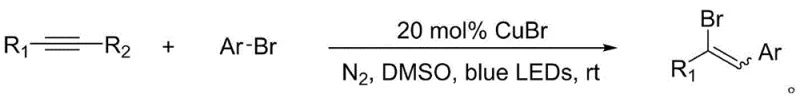
Mechanistically, this transformation is driven by the unique ability of the copper(I) bromide (CuBr) catalyst to mediate radical generation under visible light irradiation. The process initiates when the CuBr catalyst absorbs photons from the blue LED source, facilitating the homolytic cleavage of the carbon-bromine bond in the aryl bromide substrate. This generates a reactive aryl radical species which subsequently adds across the triple bond of the alkyne derivative. The resulting vinyl radical then abstracts a bromine atom from the copper-bromide complex, regenerating the catalyst and yielding the final aromatic vinyl bromide product. This catalytic cycle is highly efficient, requiring only 20 mol% of the copper catalyst to drive the reaction to completion. The mild nature of this radical generation mechanism is crucial for impurity control; by avoiding high-energy UV photons and thermal stress, the formation of polymeric byproducts and degradation fragments is drastically reduced. This results in a cleaner crude reaction mixture, which simplifies the subsequent purification steps and ensures the delivery of high-purity aromatic vinyl bromide derivatives essential for sensitive downstream applications.
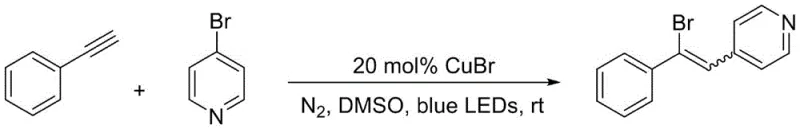
How to Synthesize Aromatic Vinyl Bromide Derivatives Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves dissolving the alkyne derivative and the brominated aromatic compound in DMSO, followed by the addition of the CuBr catalyst. The mixture is then sealed under nitrogen and exposed to 36W blue LEDs for a duration of 24 to 36 hours, with reaction progress monitored via thin-layer chromatography (TLC). Upon completion, the solvent is removed via rotary evaporation, and the product is isolated through standard silica gel column chromatography. For detailed operational parameters and specific stoichiometric ratios tailored to your production needs, please refer to the standardized synthesis guide below.
- Dissolve alkyne derivatives, brominated aromatic compounds, and 20 mol% CuBr catalyst in DMSO solvent under a nitrogen atmosphere at room temperature.
- Expose the reaction mixture to 36W blue LEDs light for 24 to 36 hours while monitoring progress via TLC.
- Upon completion, rotary evaporate the solvent and purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photo-induced synthesis technology offers tangible strategic advantages that extend beyond mere technical feasibility. The elimination of stoichiometric toxic initiators translates directly into a simplified supply chain, as there is no longer a need to source, store, and handle hazardous peroxide or organotin compounds. This reduction in hazardous material handling significantly lowers insurance premiums and regulatory compliance costs associated with dangerous goods. Furthermore, the use of ambient temperature and visible light reduces the energy footprint of the manufacturing process, contributing to long-term operational cost savings and supporting corporate sustainability goals. The robustness of the method ensures consistent batch-to-batch quality, which is critical for maintaining reliable supply lines to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the replacement of expensive and hazardous chemical initiators with inexpensive and abundant visible light energy. By utilizing a catalytic amount of CuBr rather than stoichiometric reagents, the raw material costs are significantly optimized. Additionally, the mild reaction conditions minimize equipment wear and tear and reduce the complexity of the reactor setup, as there is no need for high-pressure or high-temperature vessels. The simplified workup procedure, involving straightforward solvent evaporation and chromatography, further reduces labor hours and solvent consumption, leading to substantial cost savings in the overall manufacturing budget without compromising on output quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as phenylacetylene derivatives and bromopyridines, are commercially available commodity chemicals with stable global supply chains. The method's high substrate compatibility means that suppliers are not locked into sourcing exotic or custom-synthesized precursors, thereby reducing lead times and mitigating the risk of supply disruptions. The ability to run the reaction at room temperature also means that production is less susceptible to utility failures or heating system malfunctions, ensuring a more resilient manufacturing operation capable of meeting tight delivery schedules for reliable pharmaceutical intermediate supplier commitments.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the availability of industrial-grade LED arrays and flow chemistry reactors, which allow for uniform light penetration even at larger volumes. The absence of toxic byproducts and the use of a recoverable copper catalyst simplify waste treatment protocols, ensuring strict adherence to increasingly stringent environmental regulations. This eco-friendly profile not only avoids potential fines and shutdowns but also enhances the brand reputation of the manufacturer as a leader in sustainable chemistry, making it easier to secure contracts with environmentally conscious multinational corporations seeking cost reduction in pharmaceutical intermediate manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in patent CN111675650A, providing a clear understanding of the technology's capabilities and limitations for potential partners and stakeholders evaluating this route for their supply chain.
Q: What are the advantages of this photo-induced method over traditional ATRA processes?
A: Unlike traditional Atom Transfer Radical Addition (ATRA) which requires stoichiometric toxic initiators like peroxides or organotin reagents, this method utilizes environmentally friendly visible blue light and a catalytic amount of CuBr, significantly reducing environmental pollution and operational hazards.
Q: What is the substrate compatibility of this synthesis route?
A: The method demonstrates excellent substrate compatibility, successfully accommodating phenylacetylene compounds, aliphatic alkynes, terminal and non-terminal alkynes, as well as propargylic acid compounds, allowing for the synthesis of diverse aromatic vinyl bromide structures.
Q: What are the typical reaction conditions and yields reported?
A: Reactions are conducted at room temperature under nitrogen using DMSO as the solvent. Reported yields vary by substrate, with examples showing yields ranging from 18% to 78% and product purity reaching up to 99% after purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Vinyl Bromide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the photo-induced synthesis technology described in patent CN111675650A for the production of high-value aromatic vinyl bromide derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated at the bench level are successfully translated to industrial manufacturing. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific project requirements. By leveraging our expertise, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener methodology. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring a seamless transition to a more efficient and sustainable supply chain for your critical intermediates.
