Advanced Synthesis of 4-Amino-2-Cyclopentene-1-Methanol Hydrochloride for Commercial API Production
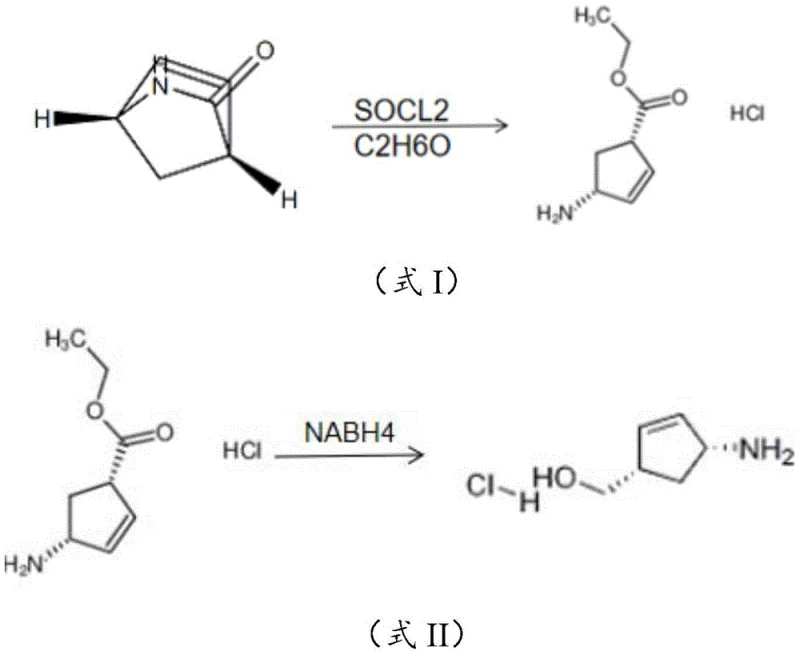
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiretroviral intermediates, and the preparation method disclosed in patent CN111484418B represents a significant leap forward in the synthesis of 4-amino-2-cyclopentene-1-methanol hydrochloride. This compound serves as a pivotal chiral building block for the manufacture of life-saving AIDS medications such as Abacavir and Carbovir, making its efficient production a matter of global health importance. The patented technology addresses long-standing challenges in yield optimization and impurity control by introducing a novel biphasic reduction strategy that fundamentally alters the reaction landscape. By leveraging a specific dichloromethane-water solvent system during the sodium borohydride reduction step, the process effectively suppresses the formation of stubborn amide by-products that have historically plagued single-phase reduction attempts. This technical breakthrough not only enhances the chemical purity of the final intermediate but also streamlines the overall workflow by removing the necessity for cumbersome amino protection and deprotection sequences. For R&D directors and process chemists, this methodology offers a compelling alternative to traditional routes that rely on expensive protecting groups like BOC anhydride or harsh acidic conditions. The ability to achieve high conversion rates while maintaining mild reaction conditions underscores the potential for this technology to become a new standard in the commercial manufacturing of cyclopentene-based pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-2-cyclopentene-1-methanol hydrochloride has been fraught with economic and environmental inefficiencies that hinder large-scale adoption. Traditional pathways often necessitate the use of BOC anhydride to protect the amino group prior to reduction, a step that introduces significant raw material costs and adds extra unit operations for subsequent deprotection. Furthermore, alternative methods attempting to bypass protection often rely on the direct reduction of carboxylic acid derivatives using substantial quantities of high-cost acid catalysts, which generate considerable hazardous waste and complicate downstream purification. In many prior art scenarios, the reduction of the ester functionality in the presence of a free or semi-protected amine leads to competitive side reactions, specifically the nucleophilic attack of the amine on the ester to form cyclic or linear amides. These side reactions not only consume valuable starting materials but also create impurities that are structurally similar to the target product, making them exceptionally difficult to remove via standard crystallization or chromatography. The reliance on solvents like methanol or ethanol in previous reduction attempts has proven insufficient to prevent these intermolecular interactions, resulting in product purities that frequently fall below the stringent requirements for API intermediates. Consequently, manufacturers have faced a dilemma between accepting lower yields with higher purification costs or investing in expensive protection strategies that elongate the supply chain.
The Novel Approach
The innovative methodology presented in the patent data circumvents these historical bottlenecks through a clever manipulation of solvent polarity and phase behavior during the critical reduction step. Instead of relying on homogeneous reaction conditions, the process utilizes a biphasic system comprising dichloromethane and water, which creates a unique microenvironment for the reacting species. In this system, the organic amine moiety of the intermediate preferentially partitions into the organic dichloromethane phase, while the ester group remains largely dispersed within the aqueous phase. This physical separation drastically reduces the probability of collision between the nucleophilic amine and the electrophilic ester carbonyl, thereby kinetically inhibiting the formation of amide by-products. The use of thionyl chloride for the initial ring-opening esterification ensures that the intermediate is obtained as a stable hydrochloride salt, which further modulates the reactivity of the amino group. By combining this stable salt formation with the biphasic reduction using sodium borohydride, the process achieves a remarkable balance between reactivity and selectivity. Experimental comparisons demonstrate that substituting dichloromethane with solvents like chloroform, tetrahydrofuran, or alcohols fails to provide the same degree of phase separation, leading to a marked decrease in both purity and yield. This specific solvent engineering allows the reaction to proceed with high efficiency without the need for additional protecting groups, representing a true process intensification.
Mechanistic Insights into Biphasic Sodium Borohydride Reduction
The core mechanistic advantage of this synthesis lies in the thermodynamic and kinetic control exerted by the dichloromethane-water interface during the hydride transfer. When sodium borohydride is introduced into the biphasic mixture, the hydride ions primarily interact with the ester functionality located in the aqueous or interfacial region, reducing it to the corresponding alcohol. Simultaneously, the protonated amine species, stabilized as a salt or free base depending on the local pH adjusted by sodium bicarbonate, exhibits higher solubility in the organic dichloromethane layer. This distribution coefficient is the key to suppressing the aminolysis side reaction; in a homogeneous polar solvent like methanol, both functional groups are fully solvated and mobile, facilitating the intramolecular or intermolecular attack that leads to lactam or amide formation. However, in the biphasic system, the effective concentration of the amine in the vicinity of the ester is kept artificially low. Furthermore, the use of the hydrochloride salt form of the ring-opened intermediate ensures that the amino group is initially less nucleophilic, requiring the pH adjustment step to liberate the free amine only when necessary for the final product stability, yet the phase separation maintains the spatial distance required to prevent side reactions. This delicate balance allows for the use of a relatively mild reducing agent like sodium borohydride, avoiding the safety hazards and strict anhydrous conditions associated with stronger reducers like lithium aluminum hydride. The result is a clean reaction profile where the primary pathway dominates, leading to the high purity observed in the experimental data.
Impurity control in this process is further enhanced by the specific choice of reagents and the order of addition, which minimizes the generation of over-reduced species or stereochemical impurities. The ring-opening step using thionyl chloride in ethanol proceeds with inversion of configuration, establishing the correct stereochemistry early in the sequence, which is preserved throughout the reduction due to the mild conditions employed. The absence of strong acids or bases during the reduction phase prevents racemization, a common issue in the synthesis of chiral cyclopentene derivatives. Additionally, the workup procedure involving extraction and pH-controlled crystallization leverages the solubility differences between the target hydrochloride salt and any remaining neutral organic impurities. By adjusting the pH of the organic layer to 1-2 with hydrochloric acid, the product precipitates as a pure salt, leaving non-basic impurities in the mother liquor. This crystallization-driven purification is highly effective and scalable, reducing the reliance on resource-intensive chromatographic separations. The combination of mechanistic suppression of by-products and physical separation during isolation ensures that the final material meets the rigorous quality standards required for downstream coupling reactions in the synthesis of complex antiretroviral agents.
How to Synthesize 4-Amino-2-Cyclopentene-1-Methanol Hydrochloride Efficiently
The execution of this synthesis requires precise control over temperature and phase ratios to maximize the benefits of the biphasic system described in the patent. The process begins with the careful addition of thionyl chloride to a cooled solution of the bicyclic ketone in ethanol, maintaining the temperature below 10°C to prevent exothermic runaway and ensure selective esterification. Following the formation of the ring-opened hydrochloride salt, the reaction mixture is concentrated and then redissolved in the critical dichloromethane-water mixture. The ratio of these solvents is paramount, with a near 1:1 volume ratio proving optimal for creating the necessary phase boundary. Before introducing the reducing agent, the pH of the aqueous phase is adjusted to 8-9 using sodium bicarbonate, which prepares the system for reduction while maintaining the biphasic integrity. The sodium borohydride is then added in portions at temperatures below 5°C to manage hydrogen evolution and heat generation. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are essential to maintain safety and consistency.
- Perform esterification ring-opening of 2-azabicyclo[2,2,1]hept-5-ene-3-one using thionyl chloride in ethanol to form the stable hydrochloride salt.
- Dissolve the ring-opened intermediate in a dichloromethane-water biphasic system and adjust pH to 8-9 using sodium bicarbonate.
- Conduct reduction with sodium borohydride at low temperature (<5°C) to selectively reduce the ester without forming amide by-products.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages by fundamentally simplifying the bill of materials and reducing the environmental footprint of the manufacturing process. The elimination of BOC anhydride, a reagent known for its volatility in price and sensitivity to moisture, removes a significant cost driver and supply risk from the production line. Furthermore, the avoidance of heavy acid catalysts reduces the burden on waste treatment facilities, lowering the operational expenditures associated with environmental compliance and hazardous waste disposal. The use of commodity chemicals such as thionyl chloride, ethanol, and sodium borohydride ensures that raw material sourcing remains stable and resilient against market fluctuations, providing a secure supply chain for long-term production contracts. The shortened synthetic route, which combines ring-opening and reduction without intermediate isolation of protected species, translates directly into reduced cycle times and higher throughput capacity for existing manufacturing assets. This efficiency gain allows suppliers to respond more rapidly to demand spikes for antiretroviral therapies, enhancing the overall reliability of the pharmaceutical supply network. Additionally, the high purity achieved directly from crystallization minimizes the need for reprocessing or extensive refining, further driving down the cost of goods sold and improving margin potential for manufacturers adopting this technology.
- Cost Reduction in Manufacturing: The removal of expensive protecting group chemistry and the substitution of costly acid catalysts with inexpensive inorganic salts lead to a drastic reduction in raw material expenses. By shortening the synthetic sequence, manufacturers save on labor, energy, and solvent consumption across multiple unit operations, resulting in a leaner and more cost-effective production model that enhances competitiveness in the generic API market.
- Enhanced Supply Chain Reliability: Relying on widely available bulk solvents like dichloromethane and ethanol mitigates the risk of supply disruptions often associated with specialized reagents. The robustness of the biphasic system tolerates minor variations in reagent quality, ensuring consistent output even when sourcing from multiple vendors, which is critical for maintaining continuous production schedules and meeting just-in-time delivery commitments to downstream API formulators.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous workup facilitate easy scale-up from kilogram to tonne scales without requiring exotic high-pressure or cryogenic equipment. The reduction in hazardous waste generation aligns with green chemistry principles, simplifying regulatory approvals and reducing the carbon footprint of the manufacturing site, which is increasingly important for multinational pharmaceutical companies aiming to meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, drawing directly from the experimental data and mechanistic insights provided in the patent documentation. Understanding these nuances is crucial for process engineers and procurement specialists evaluating the feasibility of adopting this technology for commercial production. The answers highlight the specific advantages of the biphasic system and the quality benchmarks achievable through this method.
Q: Why is the dichloromethane-water biphasic system critical for this reduction?
A: The biphasic system physically separates the organic amine (in DCM) from the ester group (in water), preventing the formation of unwanted amide by-products that typically plague single-phase reductions.
Q: How does this method improve cost efficiency compared to traditional routes?
A: This route eliminates the need for expensive BOC anhydride protection groups and avoids the use of high-cost, polluting acid catalysts, significantly lowering raw material and waste treatment costs.
Q: What purity levels can be achieved with this patented process?
A: Experimental data indicates that optimizing the solvent system to dichloromethane/water yields product purity exceeding 99%, far superior to methanol or chloroform systems which drop below 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-2-Cyclopentene-1-Methanol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against HIV/AIDS, and we are committed to delivering 4-amino-2-cyclopentene-1-methanol hydrochloride with unmatched consistency and purity. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate biphasic reduction chemistry is executed with precision at every batch size. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to verify that every shipment meets the exacting standards required for GMP API synthesis. Our commitment to quality assurance means that clients can rely on us not just as a vendor, but as a strategic partner capable of navigating the complexities of chiral intermediate manufacturing.
We invite pharmaceutical companies and contract manufacturers to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless integration of our high-performance intermediates into your drug development pipeline.
