Advanced Manufacturing of Ramelteon: A Cost-Effective Three-Step Synthetic Route
Advanced Manufacturing of Ramelteon: A Cost-Effective Three-Step Synthetic Route
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of active pharmaceutical ingredients (APIs) and their key intermediates. Patent CN112500380A, published in March 2021, introduces a transformative preparation method for Ramelteon, a potent melatonin receptor agonist used for treating insomnia. This intellectual property outlines a concise three-step synthesis starting from a nitrile precursor (Compound IV), utilizing a chemoselective reduction, a highly efficient chiral resolution, and a final acylation. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this technology represents a significant leap forward in process efficiency. By replacing hazardous high-pressure hydrogenation and expensive chiral catalysts with a mild nickel-borohydride system and cost-effective S-Mandelic acid, the patent addresses critical pain points in modern API manufacturing. The following analysis dissects the technical merits and commercial implications of this novel route for stakeholders in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ramelteon has been plagued by operational complexities and economic inefficiencies that hinder large-scale commercialization. Prior art, such as the processes described in WO2008150953 and JP11080106, often relies on multi-step sequences exceeding five or six distinct operations. A major bottleneck in these traditional routes is the reliance on asymmetric hydrogenation using homogeneous Ruthenium catalysts, which are not only prohibitively expensive but also necessitate high-pressure reaction vessels. Furthermore, conventional chiral resolution strategies frequently employ resolving agents like L-(-)-dibenzoyltartaric acid (L-DBTA) or S-Ibuprofen, which typically suffer from low resolution yields, often capping at approximately 30%. These inefficiencies result in a cumulative total yield of merely 10-20%, forcing manufacturers to process vast quantities of raw materials to obtain modest amounts of the final API. Additionally, the use of hazardous reagents like triphosgene and Grignard reagents in older pathways introduces severe safety liabilities and complicates waste management, making these routes unattractive for cost reduction in pharmaceutical manufacturing.
The Novel Approach
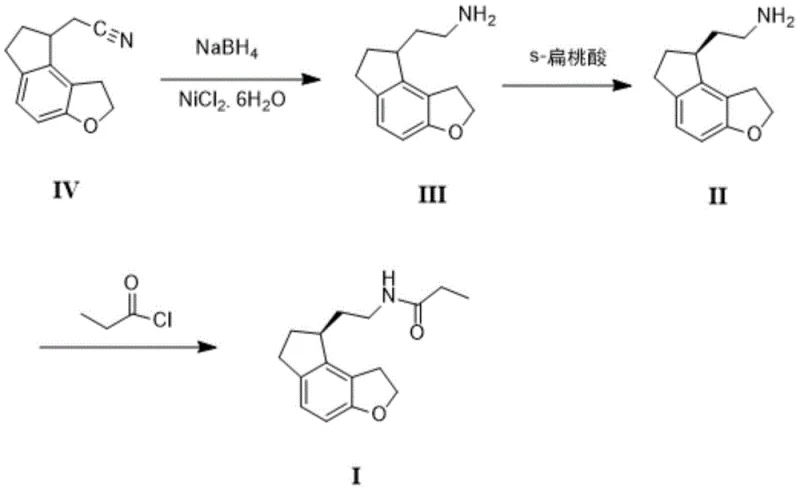
In stark contrast, the methodology disclosed in CN112500380A streamlines the production of Ramelteon into a highly efficient three-step sequence that maximizes atom economy and operational safety. As illustrated in the reaction scheme below, the process initiates with the reduction of the nitrile group in Compound IV to a primary amine (Compound III) using a Nickel Chloride and Sodium Borohydride system. This is followed by a breakthrough chiral resolution step using S-Mandelic acid, which isolates the desired enantiomer (Compound II) with exceptional efficiency. Finally, a straightforward acylation yields the target molecule. This approach eliminates the need for high-pressure equipment and precious metal catalysts, fundamentally altering the cost structure of production. The simplicity of the operation allows for easier commercial scale-up of complex pharmaceutical intermediates, ensuring that the supply chain remains resilient against technical disruptions. By integrating the resolution and subsequent acylation without intermediate solvent swaps or extensive recrystallization, the process significantly reduces solvent consumption and processing time.
Mechanistic Insights into NiCl2-Catalyzed Reduction and Chiral Resolution
The core innovation of this patent lies in the mechanistic elegance of the reduction and resolution steps, which are tailored for high selectivity and purity. The first step employs an in situ generated nickel boride catalyst from NiCl2·6H2O and NaBH4 in an ethanol-water mixture. Unlike catalytic hydrogenation which requires specialized high-pressure reactors, this chemical reduction proceeds smoothly at mild temperatures between 40°C and 50°C. The mechanism involves the chemoselective reduction of the nitrile functionality to a primary amine while leaving the sensitive fused ring system intact. This chemoselectivity is crucial for maintaining the structural integrity of the indeno-furan scaffold, preventing side reactions that could generate difficult-to-remove impurities. The use of water as a co-solvent further enhances the green chemistry profile of the reaction, facilitating easy workup and reducing the environmental footprint associated with organic solvent disposal.
Following the reduction, the chiral resolution step utilizes S-Mandelic acid to separate the enantiomers of the racemic amine. The interaction between the chiral acid and the amine forms diastereomeric salts with significantly different solubility profiles in methyl tert-butyl ether. The patent data indicates that this specific pairing achieves a resolution yield of over 45%, a substantial improvement over the 30% ceiling observed with traditional resolving agents. Mechanistically, the steric and electronic complementarity between S-Mandelic acid and the (S)-enantiomer of the amine favors the crystallization of the desired salt, leaving the unwanted enantiomer in the mother liquor. Crucially, the resulting chiral amine possesses an optical purity exceeding 99% without the need for subsequent recrystallization. This high level of stereochemical control ensures that the final Ramelteon product meets stringent regulatory standards for chiral drugs, minimizing the risk of toxicological issues associated with the wrong enantiomer.
How to Synthesize Ramelteon Efficiently
The synthesis of Ramelteon via this patented route offers a practical blueprint for laboratories and pilot plants aiming to establish a robust supply of this sleep aid intermediate. The protocol is designed to be operationally simple, requiring standard glassware and avoiding extreme conditions. The process begins with the preparation of the racemic amine, followed by the critical resolution step where temperature control and solvent ratios are key to maximizing yield. The final acylation is a standard Schotten-Baumann type reaction that proceeds rapidly at room temperature. For detailed procedural specifics regarding molar ratios, temperature gradients, and workup procedures, operators should refer to the standardized synthesis guidelines provided below. Adhering to these parameters ensures reproducibility and consistent quality across batches, which is essential for maintaining a reliable supply chain for downstream API manufacturers.
- Perform chemoselective reduction of the nitrile precursor (Compound IV) using Sodium Borohydride and Nickel Chloride Hexahydrate in an ethanol-water system to obtain the racemic amine (Compound III).
- Execute chiral resolution of the racemic amine using S-Mandelic acid in methyl tert-butyl ether, followed by basification and extraction to isolate the optically pure amine (Compound II).
- Conduct the final acylation reaction by reacting the chiral amine with propionyl chloride in dichloromethane using an aqueous sodium hydroxide acid-binding agent to yield Ramelteon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible strategic benefits beyond mere technical feasibility. The elimination of high-pressure hydrogenation and expensive homogeneous catalysts removes significant capital expenditure barriers and operational risks. By shifting to a ambient pressure chemical reduction, facilities can utilize existing reactor infrastructure without needing specialized high-pressure autoclaves, thereby accelerating the timeline for technology transfer and production startup. Furthermore, the drastic reduction in synthesis steps—from six or more in prior art to just three—directly correlates with reduced labor costs, lower utility consumption, and decreased waste generation. This streamlined workflow enhances the overall agility of the manufacturing process, allowing suppliers to respond more quickly to market demand fluctuations for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The substitution of costly chiral Ruthenium catalysts and complex resolving agents with inexpensive S-Mandelic acid and Nickel salts drives down the raw material cost per kilogram significantly. Additionally, the high resolution yield (>45%) means that less starting material is wasted compared to traditional methods where nearly 70% of the racemate might be discarded or require recycling. The ability to proceed to the final acylation step directly from the resolution filtrate without isolating and drying the intermediate amine further saves on solvent usage and energy costs associated with evaporation and drying. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like Sodium Borohydride, Nickel Chloride, and S-Mandelic acid ensures a stable and secure supply of raw materials, unlike specialized catalysts which may have long lead times or single-source dependencies. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing, reduces the likelihood of batch failures and production delays. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing stockouts that could disrupt the production of finished dosage forms. Consequently, partners adopting this technology can offer more dependable delivery schedules and stronger service level agreements to their pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-hundred gram scales in the patent examples, with clear pathways to ton-scale production. The avoidance of hazardous reagents like triphosgene and the reduction in solvent volume due to fewer steps align with increasingly strict environmental regulations and green chemistry initiatives. The simplified waste stream, characterized primarily by aqueous saline solutions and common organic solvents, is easier and cheaper to treat than the heavy metal-contaminated waste from Ruthenium-catalyzed processes. This environmental compliance reduces the regulatory burden and potential liability for manufacturing sites, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ramelteon synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in patent CN112500380A. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific advantages in terms of yield, purity, and operational safety that distinguish this method from legacy processes.
Q: What are the advantages of using S-Mandelic acid for Ramelteon resolution compared to traditional agents?
A: According to patent CN112500380A, using S-Mandelic acid achieves a resolution yield exceeding 45% with over 99% chiral purity without recrystallization. This significantly outperforms prior art agents like L-(-)-DBTA or S-Ibuprofen, which typically yield around 30% and require complex purification steps.
Q: How does the Nickel-Borohydride reduction method improve safety over hydrogenation?
A: The patented method utilizes NaBH4 and NiCl2·6H2O at mild temperatures (40-50°C), eliminating the need for high-pressure hydrogen gas and expensive homogeneous Ruthenium catalysts. This drastically reduces equipment requirements and safety risks associated with high-pressure hydrogenation reactors.
Q: What is the overall yield and purity profile of this synthetic route?
A: The process delivers a total yield of over 42% from the initial raw materials, which is substantially higher than the 10-20% typical of literature methods. The final Ramelteon product achieves HPLC purity of 99.5% to 99.7% directly after drying, avoiding the need for multiple recrystallizations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ramelteon Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant manufacturing pathways for high-value pharmaceutical intermediates like Ramelteon. Our technical team has thoroughly analyzed the innovations within CN112500380A and possesses the expertise to implement this advanced three-step synthesis at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral purity >99% and HPLC purity >99.5%, guaranteeing that every batch meets the exacting standards required for FDA-regulated markets.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this cost-effective technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your Ramelteon supply chain with a solution that balances technical excellence with commercial viability.
