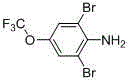
Advanced Water-Phase Synthesis of 2,6-Dibromo-4-(trifluoromethoxy)aniline for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance high purity with environmental sustainability. A pivotal advancement in this domain is detailed in patent CN102491910B, which discloses a novel water-phase method for synthesizing 2,6-dibromo-4-(trifluoromethoxy)aniline. This compound serves as a critical building block for high-performance agricultural bactericides, such as thiophene fluorine bacterium amine, and various pesticide herbicides. Unlike traditional organic solvent-based approaches that struggle with waste management and impurity profiles, this innovation leverages water as the primary medium, coupled with a unique mechanical dispersion strategy using inert grinding media. The technical breakthrough lies not only in the substitution of hazardous solvents but in the sophisticated management of reaction kinetics through oxidative bromine regeneration. For R&D directors and process chemists, this represents a paradigm shift towards greener chemistry without compromising the stringent quality standards required for downstream API synthesis. The method achieves exceptional yields ranging from 97.5% to 99.1% and maintains product purity between 98.4% and 99.4%, demonstrating its viability for rigorous commercial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of poly-brominated anilines has been plagued by significant technical and environmental hurdles. Prior art, such as US5045554, relied heavily on acetic acid as a solvent, necessitating a twofold molar excess of bromine relative to the substrate. This stoichiometric imbalance leads to severe wastage of bromine resources and generates substantial quantities of hydrogen bromide-acetic acid solutions that are notoriously difficult to treat. Furthermore, alternative aqueous methods described in patents like US6114584, while avoiding organic solvents, often suffer from poor mass transfer characteristics. When bromine feed rates are suboptimal, the reaction tends to produce a mixture of mono- and di-bromo derivatives that coalesce into a viscous, jelly-like mass. This physical phenomenon creates severe agitation difficulties, leading to hot spots, incomplete reactions, and challenging downstream filtration processes. The presence of persistent mono-bromo impurities is particularly detrimental, as these structural analogs possess properties closely resembling the target molecule, making purification via crystallization or distillation energetically expensive and technically demanding.
The Novel Approach
The methodology outlined in CN102491910B fundamentally re-engineers the reaction environment to overcome these legacy issues. By introducing inert grinding media—such as zirconia beads, granulated glass spheres, or stainless steel shots—into the aqueous reaction matrix, the process effectively disrupts the formation of viscous agglomerates. These media act as physical dispersants, ensuring that the intermediate species remain suspended and accessible for further bromination, thereby driving the reaction to completion with minimal mono-bromo residue. Additionally, the strategic use of hydrogen peroxide allows for the catalytic regeneration of bromine from the hydrogen bromide byproduct, significantly lowering the net consumption of elemental bromine. This approach not only simplifies the stoichiometry but also mitigates the accumulation of acidic waste. The result is a streamlined process where the reaction mother liquor can be directly recycled for subsequent batches, creating a semi-continuous loop that enhances overall process efficiency and reduces the environmental footprint associated with solvent disposal.
Mechanistic Insights into Oxidative Bromination with Mechanical Dispersion
The core chemical transformation involves an electrophilic aromatic substitution where the amino group directs bromination to the ortho positions. However, the novelty of this patent lies in the synergistic effect between the chemical oxidant and the mechanical additives. In a standard aqueous bromination, the generation of HBr lowers the pH, potentially protonating the aniline nitrogen and deactivating the ring towards further electrophilic attack. The addition of hydrogen peroxide (30% concentration) serves a dual purpose: it oxidizes the generated HBr back into molecular bromine (Br2), maintaining the concentration of the active electrophile, and it helps buffer the acidity to some extent by consuming the bromide ions. The molar ratio is carefully optimized at 1:1.0-1.1 for the substrate to bromine, and 1:1.3-1.5 for hydrogen peroxide, ensuring that there is sufficient oxidizing power to drive the cycle without causing over-oxidation of the sensitive aniline moiety. This redox cycle is crucial for maintaining a steady state of reactive bromine species throughout the 6 to 8-hour reaction window.
From a physical-organic perspective, the inclusion of inert grinding media addresses the heterogeneity of the reaction mixture. As the bromination proceeds, the solubility of the intermediates changes, often leading to precipitation. Without mechanical intervention, these precipitates can encapsulate unreacted starting material or trap byproducts, leading to the aforementioned "jelly" effect. The grinding media, added at 3% to 5% of the substrate mass, provides a high-surface-area interface that prevents particle aggregation. Under vigorous stirring conditions of 300 to 500 r/min, these beads collide with the forming solids, breaking up clusters and ensuring uniform exposure to the brominating agents. This mechanical action is vital for suppressing the formation of the 2-mono-bromo derivative, keeping its content below 0.1%. Such precise impurity control is essential for downstream coupling reactions in pesticide synthesis, where even trace impurities can catalyze decomposition or reduce the biological efficacy of the final agrochemical product.
How to Synthesize 2,6-Dibromo-4-(trifluoromethoxy)aniline Efficiently
The operational protocol for this synthesis is designed for scalability and reproducibility, leveraging standard chemical engineering equipment with minor modifications for the inclusion of grinding media. The process begins with the charging of the reactor with water, the aniline substrate, and the selected inert beads, followed by controlled temperature management between 20°C and 30°C. The sequential addition of reagents is critical; bromine is introduced first to initiate the substitution, followed by hydrogen peroxide to sustain the oxidative cycle. Detailed parameters regarding stirring speeds, drop rates, and filtration techniques are essential for replicating the high yields reported in the patent data. For process engineers looking to implement this technology, adherence to the specific sequence of operations ensures the prevention of exothermic runaways and maximizes the efficiency of the bromine regeneration loop.
- Prepare the reaction system by adding 4-(trifluoromethoxy)aniline, water solvent, and inert grinding media (such as zirconia beads) into a reactor equipped with agitation.
- Sequentially add bromine and 30% hydrogen peroxide dropwise while maintaining temperature between 20-30°C and stirring at 300-500 r/min to facilitate oxidative regeneration of bromine.
- Post-reaction, separate the inert grinding media, filter the mixture to recycle the filtrate for subsequent batches, and dry the filter cake to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this water-phase technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of acetic acid and other organic solvents removes the need for complex solvent recovery distillation columns and reduces the fire hazard classification of the production facility. This simplification of the infrastructure directly translates to lower capital expenditure (CAPEX) for new plants and reduced operating expenditure (OPEX) for existing ones. Furthermore, the ability to recycle the reaction filtrate means that water consumption is drastically minimized, and the volume of wastewater requiring treatment is significantly reduced. This aligns perfectly with increasingly stringent global environmental regulations, reducing the risk of production shutdowns due to compliance issues. The supply chain becomes more resilient as the dependency on volatile organic solvents, which are subject to price fluctuations and transport restrictions, is removed from the bill of materials.
- Cost Reduction in Manufacturing: The most significant driver for cost optimization in this process is the efficient utilization of bromine. By employing hydrogen peroxide to regenerate bromine from the HBr byproduct, the net consumption of elemental bromine is substantially lowered compared to traditional methods that require a large excess. This stoichiometric efficiency means that for every ton of product produced, the raw material cost associated with halogenation is markedly decreased. Additionally, the removal of the neutralization step typically required to handle large volumes of hydrobromic acid waste eliminates the cost of base reagents and the disposal fees associated with salt byproducts. The process essentially turns a waste liability into a reusable asset, creating a leaner manufacturing model that improves gross margins without sacrificing quality.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the complexity of purification. Because this method inherently suppresses the formation of difficult-to-remove mono-bromo impurities through mechanical dispersion, the reliance on extensive recrystallization or chromatographic purification is diminished. This streamlining of the downstream processing shortens the overall production cycle time, allowing for faster turnover of batches and more responsive fulfillment of customer orders. Moreover, the use of water as a solvent mitigates the risks associated with the transportation and storage of flammable organic liquids, simplifying logistics and warehousing requirements. Suppliers adopting this method can offer more consistent lead times, as the process is less susceptible to the variability introduced by solvent quality or recovery inefficiencies.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces heat and mass transfer challenges that are magnified in viscous systems. The incorporation of inert grinding media ensures that the reaction mixture remains fluid and manageable even at larger scales, preventing the formation of dead zones where side reactions could occur. This robustness facilitates a smoother scale-up from pilot plant to commercial production, reducing the time and cost associated with process validation. From an environmental standpoint, the drastic reduction in three wastes (wastewater, waste gas, and solid waste) positions this manufacturing route as a sustainable choice. The recyclable nature of both the solvent and the grinding media means that the process generates minimal solid waste, supporting corporate sustainability goals and easing the burden on waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this water-phase synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this intermediate into their broader supply chains.
Q: How does the water-phase method improve impurity control compared to acetic acid solvents?
A: The water-phase method utilizes inert grinding media to prevent the agglomeration of intermediate mono-bromo derivatives, which often form viscous jelly-like masses in other solvents. This mechanical dispersion ensures uniform reaction kinetics, reducing mono-bromo impurities to less than 0.1% and achieving product purity of 98.4-99.4%.
Q: What is the role of hydrogen peroxide in this bromination process?
A: Hydrogen peroxide acts as an oxidizing agent that regenerates elemental bromine from the hydrogen bromide byproduct formed during the reaction. This in-situ regeneration significantly reduces the total consumption of elemental bromine and minimizes the generation of acidic waste streams.
Q: Can the reaction mother liquor be recycled for industrial production?
A: Yes, the patent data confirms that the filtrate can be directly recycled as the solvent for the next batch without significant loss in yield or purity. This closed-loop solvent system drastically reduces wastewater discharge and lowers the overall E-factor of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dibromo-4-(trifluoromethoxy)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced agrochemicals depends on the reliability and quality of the underlying intermediates. Our technical team has extensively analyzed the water-phase oxidative bromination pathway and possesses the expertise to scale this diverse synthesis route from laboratory benchmarks to full commercial production. We are equipped to handle volumes ranging from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities operate under stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the 98.4% to 99.4% purity threshold required for high-performance pesticide formulations. We understand that impurity profiles can make or break a downstream synthesis, which is why our focus on mechanical dispersion and oxidative regeneration is central to our manufacturing philosophy.
We invite you to collaborate with us to optimize your supply chain for 2,6-dibromo-4-(trifluoromethoxy)aniline. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive down costs and enhance the sustainability of your agrochemical portfolio through the adoption of this superior water-phase technology.
