Advanced N-Haloimide Halogenation Strategy for High-Purity Vinylene Carbonate Production
The global demand for high-performance lithium-ion batteries has intensified the search for robust electrolyte additives, with vinylene carbonate (VC) standing out as a critical component for stabilizing the solid electrolyte interphase (SEI) on graphite anodes. Patent CN115745943A introduces a transformative synthetic methodology that addresses the longstanding safety and environmental bottlenecks associated with traditional VC production. This innovation shifts the paradigm from hazardous gas-phase chlorination to a controlled, solution-phase halogenation using N-haloimides, offering a pathway to high-purity intermediates essential for next-generation energy storage systems. For R&D directors and procurement strategists in the new energy sector, this patent represents a significant leap forward in process safety and product quality, potentially redefining the supply chain dynamics for battery grade chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of vinylene carbonate has relied heavily on two primary routes, both of which carry substantial operational liabilities that hinder scalable and sustainable manufacturing. The first conventional route involves the direct chlorination of ethylene carbonate using elemental chlorine gas under ultraviolet irradiation. While chemically effective, this method necessitates the handling of highly toxic and corrosive chlorine gas, imposing rigorous safety protocols, specialized containment infrastructure, and significant environmental compliance costs. The second prevalent method utilizes sulfuryl chloride as the chlorinating agent; however, this approach requires elevated temperatures that promote the decomposition of the reagent and generates large volumes of hydrogen chloride gas as a by-product. Neutralizing this acidic waste stream consumes vast quantities of alkali and creates complex wastewater treatment challenges, drastically inflating the operational expenditure and ecological footprint of the manufacturing facility.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in the patent leverages N-haloimides, such as N-chlorosuccinimide (NCS) or N-bromosuccinimide (NBS), as solid-state halogenating agents. This strategic substitution eliminates the need for hazardous gas handling equipment and mitigates the risk of accidental toxic releases. The reaction proceeds smoothly in common organic solvents like tetrahydrofuran or toluene at moderate temperatures ranging from 50°C to 150°C. Furthermore, the by-product of the halogenation step is succinimide or its derivatives, which are solid compounds that can be efficiently removed via simple crystallization and filtration at low temperatures (0°C to 10°C). This "one-pot" philosophy not only streamlines the workflow but also ensures that the subsequent elimination step begins with a highly purified intermediate, setting the stage for superior final product quality without the burden of extensive downstream purification.
Mechanistic Insights into N-Haloimide Mediated Halogenation and Elimination
The core chemical transformation relies on a precise two-step sequence that maximizes atom economy while minimizing side reactions. In the initial halogenation phase, the N-haloimide acts as an electrophilic source of halogen, reacting with ethylene carbonate to form a halogenated ethylene carbonate intermediate. This step is kinetically controlled and benefits from the stability of the N-haloimide reagent, which prevents the runaway exotherms often seen with gaseous chlorine. Following the removal of the succinimide by-product through crystallization, the filtrate containing the halogenated intermediate undergoes a base-catalyzed elimination reaction. Bases such as triethylamine, sodium hydroxide, or sodium methoxide abstract a proton adjacent to the halogen substituent, triggering an E2 elimination mechanism that forms the carbon-carbon double bond characteristic of vinylene carbonate. This mechanistic pathway is highly selective, effectively suppressing polymerization or ring-opening side reactions that typically plague VC synthesis.
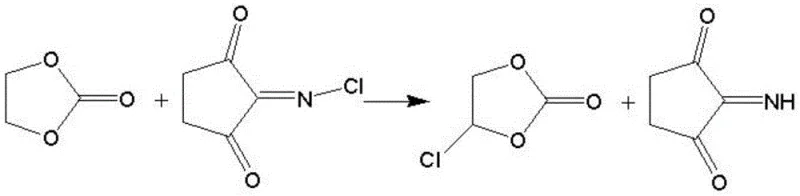
Impurity control is intrinsically built into this mechanism through the physical state of the by-products. Unlike the gaseous HCl generated in sulfuryl chloride routes, which remains dissolved and difficult to separate completely, the succinimide by-product precipitates out of the solution upon cooling. This phase separation allows for a mechanical removal of the bulk impurity before the elimination step even begins. Additionally, the use of mild bases like triethylamine generates amine hydrochloride salts that are also solid and easily filtered off. This multi-stage solid-liquid separation strategy ensures that the final distillation step deals with a much cleaner feedstock, directly contributing to the reported gas chromatography purity levels exceeding 98%. For technical teams, this implies a robust process window where minor deviations in reaction time do not catastrophically impact the final impurity profile.
How to Synthesize Vinylene Carbonate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale execution, emphasizing the importance of temperature control and sequential workup procedures. The process begins with the dissolution of ethylene carbonate and the N-haloimide in a suitable solvent, followed by heating to reflux to drive the halogenation to completion. Once the starting material is consumed, the solvent is concentrated, and the mixture is cooled to induce crystallization of the succinimide by-product. The resulting filtrate is then treated with a base to effect the elimination, followed by filtration of the salt waste. Finally, the crude product is isolated via vacuum distillation. For a comprehensive understanding of the specific molar ratios, solvent choices, and exact thermal profiles required for optimal yield, please refer to the standardized synthesis steps provided in the guide below.
- Halogenation: React ethylene carbonate with N-haloimide (e.g., NCS) in solvent at 50-150°C, then crystallize to remove succinimide by-product.
- Elimination: Treat the halogenated intermediate with a base (e.g., triethylamine) at 50-150°C to induce dehydrohalogenation.
- Purification: Filter solid salts and perform vacuum distillation (65-80°C) to isolate pure vinylene carbonate and recover solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this N-haloimide based synthesis route offers profound advantages that extend beyond mere chemical elegance, directly addressing the pain points of cost, safety, and logistics faced by procurement managers and supply chain heads. The transition from gaseous to solid reagents fundamentally alters the risk profile of the manufacturing site, reducing the need for expensive corrosion-resistant reactors and specialized gas scrubbing systems. This capital expenditure saving is complemented by a reduction in operational complexity, as solid reagents are easier to store, transport, and dose with high precision compared to pressurized gas cylinders. Consequently, the overall cost of goods sold (COGS) is optimized not just through raw material efficiency, but through significant reductions in safety compliance overhead and waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of toxic chlorine gas and sulfuryl chloride removes the necessity for heavy-duty safety infrastructure and continuous monitoring systems, leading to substantial capital and operational cost savings. Furthermore, the ability to recover and recycle solvents like tetrahydrofuran or toluene through simple distillation enhances the overall material efficiency of the process. The solid by-products, such as succinimide, are non-corrosive and easier to handle than acidic gas wastes, reducing the financial burden associated with hazardous waste treatment and neutralization chemicals.
- Enhanced Supply Chain Reliability: Relying on solid N-haloimides instead of regulated toxic gases significantly de-risks the raw material supply chain. Solid reagents have longer shelf lives, are less susceptible to transportation delays caused by hazardous material regulations, and can be sourced from a broader range of chemical suppliers. This stability ensures consistent production schedules and minimizes the risk of shutdowns due to raw material shortages or regulatory changes regarding the transport of compressed toxic gases, thereby guaranteeing a steady flow of high-purity electrolyte additives to battery manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for green chemistry principles, generating minimal hazardous waste and avoiding the release of noxious fumes into the environment. This aligns perfectly with increasingly stringent global environmental regulations, facilitating easier permitting for plant expansion and scale-up. The simplicity of the workup procedure, involving filtration and distillation rather than complex extraction or washing steps, makes the technology highly amenable to continuous processing and large-scale batch production, ensuring that supply can meet the exploding demand of the electric vehicle market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, derived directly from the patent specifications and comparative analysis. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for potential licensing or procurement partnerships. Understanding these nuances is crucial for making informed decisions about integrating this safer, high-efficiency method into existing production lines.
Q: Why is the N-haloimide route safer than traditional chlorine gas methods?
A: Traditional methods utilize toxic chlorine gas or sulfuryl chloride, which pose severe safety risks and generate corrosive HCl waste. The N-haloimide route uses stable solid reagents, eliminating gas handling hazards and simplifying waste management.
Q: What purity levels can be achieved with this synthesis method?
A: The patent demonstrates that this method consistently achieves product purity greater than 98% via GC analysis, meeting the stringent requirements for lithium-ion battery electrolyte additives.
Q: How does this process improve supply chain reliability for electrolyte manufacturers?
A: By replacing hazardous gaseous reagents with shelf-stable solids like N-chlorosuccinimide, the process reduces regulatory burdens and transportation risks, ensuring more consistent production scheduling and raw material availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinylene Carbonate Supplier
As the demand for high-performance battery materials continues to surge, partnering with a technically proficient CDMO is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications mean that we can deliver vinylene carbonate and other critical electrolyte additives that meet the exacting standards required by top-tier battery cell manufacturers, minimizing the risk of cell failure and maximizing performance.
We invite you to engage with our technical procurement team to discuss how this advanced N-haloimide synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this safer, greener process. We encourage you to reach out for specific COA data and route feasibility assessments to validate how our capabilities align with your supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
