Advanced Ruthenium-Catalyzed Condensation for High-Purity Stilbene Derivatives Manufacturing
The chemical industry is constantly seeking more robust and efficient catalytic systems to streamline the production of high-value intermediates. A significant breakthrough in this domain is documented in patent CN111961087A, which introduces a novel semi-sandwich ruthenium complex containing an ortho-position carborane-based benzothiazole ligand. This innovation addresses critical stability issues found in earlier generations of organometallic catalysts, specifically targeting the synthesis of stilbene derivatives which are fundamental structural motifs in numerous pharmaceutical and optical molecules. The disclosed technology represents a paradigm shift from harsh, sensitive catalytic processes to a more forgiving, green chemistry approach that maintains high efficiency while drastically simplifying operational requirements for industrial scale-up.
Traditional methods for constructing the stilbene backbone, such as the Wittig reaction and Heck coupling, have long been staples in organic synthesis but suffer from inherent drawbacks that complicate large-scale manufacturing. The Wittig reaction, for instance, necessitates the use of toxic phosphorus ylides and generates stoichiometric amounts of phosphine oxide waste, creating significant disposal challenges and environmental liabilities. Similarly, while the Heck reaction is powerful, it often produces substantial halide by-products and requires expensive palladium catalysts that can be difficult to remove from the final product to meet stringent pharmaceutical purity standards. Furthermore, previous attempts at direct C-H activation for this transformation, such as those reported by Matsuzaka, utilized ruthenium catalysts that were highly sensitive to air and moisture, demanding rigorous inert atmosphere techniques and elevated temperatures around 150°C for extended periods of up to 24 hours.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on legacy synthetic routes imposes severe constraints on supply chain agility and cost structures for fine chemical manufacturers. The necessity for strictly anhydrous and anaerobic conditions in previous ruthenium-catalyzed protocols increases capital expenditure on specialized equipment and raises the barrier for entry for contract manufacturing organizations. Additionally, the thermal instability of older catalyst formulations often leads to decomposition during the reaction course, resulting in inconsistent batch-to-batch reproducibility and lower overall atom economy. The generation of hazardous waste streams from phosphorus or halide by-products further complicates regulatory compliance, forcing procurement teams to manage complex waste treatment logistics that erode profit margins and delay time-to-market for critical drug intermediates.
The Novel Approach
The methodology outlined in the patent data offers a transformative solution by leveraging a uniquely designed ligand system that imparts exceptional stability to the ruthenium center. By incorporating an ortho-carborane cage coupled with a benzothiazole moiety, the new complex achieves a semi-sandwich geometry that protects the metal from deactivation by atmospheric oxygen or moisture. This structural innovation allows the catalytic condensation of toluene and aromatic aldehydes to proceed under remarkably mild conditions, typically between 50°C and 80°C, with reaction times reduced to just 6 to 10 hours. The process is not only operationally simpler but also exhibits superior atom economy, as the only by-product generated is water, aligning perfectly with modern green chemistry principles and reducing the environmental footprint of the manufacturing process.
Mechanistic Insights into Carborane-Stabilized Ruthenium Catalysis
The exceptional performance of this catalyst stems from the electronic and steric properties of the carborane-based ligand, which creates a highly stabilized coordination environment around the ruthenium atom. The three-dimensional aromaticity of the carborane cage provides strong sigma-donation capabilities that reinforce the metal-ligand bond, preventing the dissociation that typically leads to catalyst death in harsh environments. Mechanistically, the reaction likely proceeds through a C-H activation pathway where the ruthenium center activates the benzylic position of toluene, followed by coordination and insertion of the aromatic aldehyde. The robust nature of the complex ensures that the active catalytic species remains intact throughout the cycle, facilitating multiple turnover numbers without significant degradation, which is crucial for maintaining high turnover frequencies in industrial reactors.
From an impurity control perspective, the stability of the catalyst minimizes the formation of metal-leached by-products that often plague transition metal-catalyzed reactions. In traditional systems, ligand dissociation can lead to the precipitation of ruthenium black or the formation of unreactive clusters that contaminate the product stream. However, the tight binding of the carborane-benzothiazole ligand ensures that the metal remains sequestered within the coordination sphere, simplifying downstream purification processes. This characteristic is particularly valuable for pharmaceutical applications where residual metal limits are strictly regulated, allowing manufacturers to achieve high-purity stilbene derivatives with fewer purification steps and reduced solvent consumption during workup.
How to Synthesize Semi-Sandwich Ruthenium Complex Efficiently
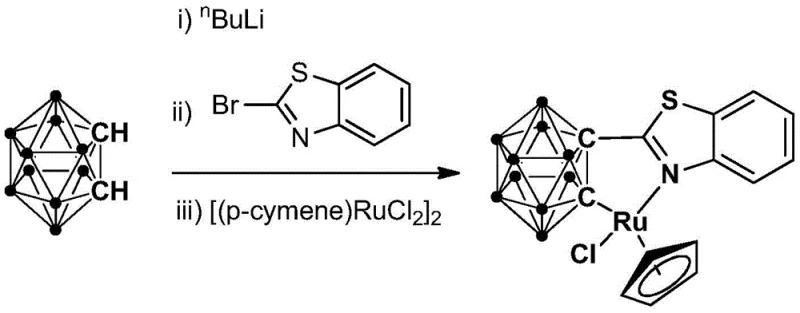
The preparation of this advanced catalyst follows a streamlined, one-pot strategy that avoids the need for isolating unstable intermediates, thereby enhancing safety and throughput. The process begins with the controlled lithiation of ortho-carborane at cryogenic temperatures, followed by sequential addition of the heterocyclic bromide and the ruthenium dimer. This telescoped approach minimizes material handling and exposure to potential contaminants, ensuring high fidelity in the final catalyst structure. For detailed operational parameters and safety protocols regarding reagent addition rates and quenching procedures, please refer to the standardized synthesis guide below.
- Perform lithiation of ortho-carborane using n-butyllithium at cryogenic temperatures (-70 to -80°C) followed by warming to room temperature.
- React the lithiated intermediate with bromobenzothiazole at room temperature for 6 to 8 hours to form the ligand precursor.
- Add the binuclear ruthenium compound [(p-cymene)RuCl2]2 and react at room temperature for 3 to 5 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers profound strategic benefits for organizations focused on optimizing their supply chain resilience and reducing total cost of ownership. The shift towards a water-generating, atom-economical process eliminates the need for costly waste management associated with phosphorus or halide disposal, directly impacting the bottom line through reduced environmental compliance fees. Furthermore, the ambient stability of the catalyst removes the requirement for expensive glovebox operations or specialized inert gas lines, allowing production to occur in standard multipurpose reactors that are readily available in most chemical manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of toxic reagents and the ability to operate at significantly lower temperatures translates to substantial energy savings and reduced raw material costs. By avoiding the use of stoichiometric phosphorus ylides or expensive palladium sources, the variable cost per kilogram of the final stilbene derivative is drastically lowered. Additionally, the simplified workup procedure, which often requires only concentration and column chromatography, reduces solvent usage and labor hours, contributing to a leaner and more cost-effective production model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: The robustness of the catalyst against air and moisture ensures consistent performance regardless of minor fluctuations in plant conditions, leading to reliable batch outcomes and predictable delivery schedules. Since the starting materials such as toluene and various substituted benzaldehydes are commodity chemicals with established global supply networks, the risk of raw material shortages is minimized. This reliability allows procurement managers to negotiate better long-term contracts and maintain lower safety stock levels, freeing up working capital for other strategic investments within the organization.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by water as the sole by-product, aligns seamlessly with increasingly stringent environmental regulations worldwide. Scaling this process from laboratory to commercial production does not require complex engineering controls for hazardous gas scrubbing or heavy metal waste treatment, facilitating faster regulatory approvals. The high selectivity of the reaction also means fewer side products are formed, reducing the load on purification units and enabling the facility to handle higher throughput volumes without expanding waste treatment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic system, derived directly from the experimental data and comparative analysis provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this technology into existing production lines for fine chemical intermediates.
Q: Is the semi-sandwich ruthenium catalyst sensitive to air and moisture?
A: Unlike previous ruthenium catalysts reported by Matsuzaka which required strict inert conditions, this novel complex containing ortho-carborane benzothiazole is insensitive to both air and water, allowing for stable storage and handling in ambient environments.
Q: What are the typical reaction conditions for the catalytic condensation?
A: The catalytic process operates under mild conditions, typically requiring temperatures between 50°C and 80°C for 6 to 10 hours, which is significantly lower than the 150°C required by prior art methods.
Q: What is the substrate scope for this catalytic system?
A: The system demonstrates excellent universality, successfully catalyzing the condensation of toluene with various aromatic aldehydes including those with electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like nitro, chloro, and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semi-Sandwich Ruthenium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this carborane-based ruthenium catalysis for the efficient production of high-value stilbene derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate delivered meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this innovative route can optimize your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener methodology. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your project timelines and volume requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
