Industrial Scale Production of Quinazolinedione Phenylalanine Derivatives for Anti-Inflammatory APIs
The pharmaceutical industry continuously seeks robust synthetic pathways for complex anti-inflammatory agents, particularly those targeting alpha4 integrin inhibition. Patent CN101589028A presents a significant technological advancement in the preparation of phenylalanine derivatives possessing quinazolinedione skeletons, which are critical intermediates for treating conditions such as rheumatoid arthritis and asthma. This innovation addresses longstanding challenges in yield optimization and process scalability by shifting from traditional solid-phase or ester-protected routes to a more efficient liquid-phase synthesis utilizing free carboxylic acid precursors. The core breakthrough lies in the strategic use of specific anthranilic acid derivatives that allow for higher convergence and purity in the final active pharmaceutical ingredient (API) intermediates. 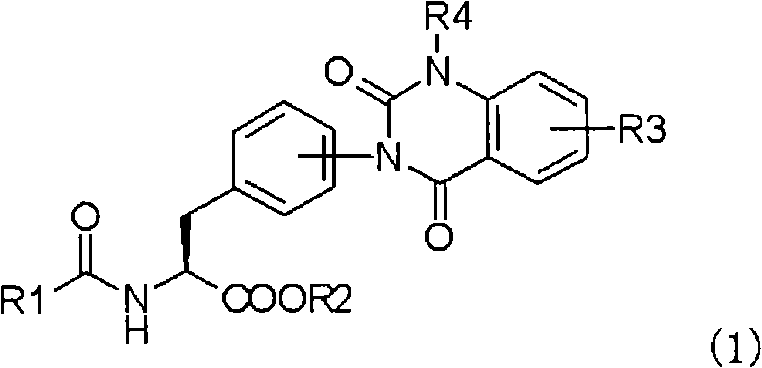
For procurement and supply chain leaders, understanding the underlying chemistry of this patent is essential for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality. The described methodology not only enhances the chemical efficiency but also simplifies the purification landscape, reducing the burden on downstream processing units. By leveraging this specific synthetic route, manufacturers can achieve substantial cost savings in API manufacturing through reduced solvent usage and higher throughput per batch. The transition to this novel approach represents a pivotal shift towards more sustainable and economically viable production models for high-value therapeutic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinedione scaffolds relied heavily on solid-phase resin loading or the use of ester-protected anthranilic acid derivatives, as documented in prior art such as WO02/16329 and WO2004/74264. These conventional methods often suffered from inherent inefficiencies, including lower overall yields and the generation of difficult-to-remove impurities that compromised the purity profile of the final product. For instance, comparative data within the patent highlights that using ester-protected starting materials resulted in yields as low as 55%, necessitating extensive recycling or disposal of valuable reaction mass. Furthermore, solid-phase techniques, while useful for library synthesis, are notoriously difficult to scale for commercial tonnage due to resin costs and filtration bottlenecks. 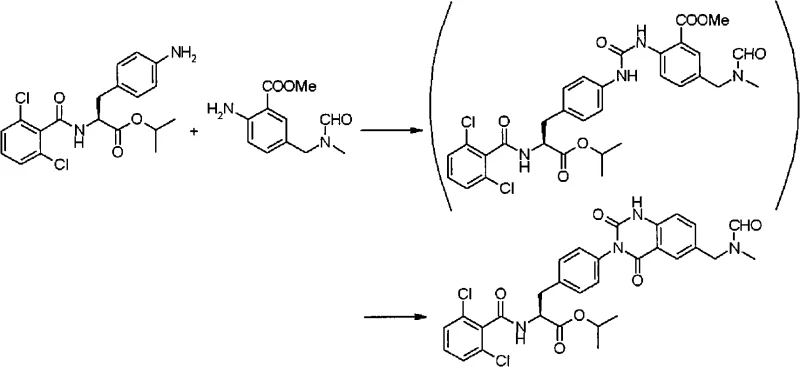
The reliance on protected groups also introduces additional synthetic steps, such as protection and deprotection sequences, which elongate the production timeline and increase the consumption of reagents and solvents. This linear expansion of the synthetic route directly impacts the cost of goods sold (COGS) and extends the lead time for high-purity API intermediates. In an industrial setting, the accumulation of by-products from these less efficient routes requires rigorous chromatographic purification, which is often impractical at multi-kilogram scales. Consequently, there was a critical need for a convergent synthesis strategy that could bypass these limitations while maintaining strict control over stereochemistry and impurity profiles.
The Novel Approach
The novel approach disclosed in CN101589028A fundamentally alters the reaction landscape by employing anthranilic acid derivatives with unprotected carboxylic acid groups as key building blocks. This strategic modification allows for a direct condensation with acylphenylalanine derivatives in the presence of a carbonyl-introducing agent, leading to the formation of asymmetric urea intermediates with significantly improved efficiency. The elimination of the ester protection group reduces the step count and avoids the hydrolysis steps typically required to reveal the reactive carboxylic acid functionality later in the sequence. This streamlined pathway not only accelerates the reaction kinetics but also enhances the solubility characteristics of the intermediates, facilitating better mixing and heat transfer in large reactors.
Moreover, the new method demonstrates exceptional scalability, as evidenced by successful kilogram-scale runs where yields exceeded 88% for the key cyclization step. The process utilizes robust crystallization techniques for isolation, replacing complex chromatographic separations with simple filtration and washing operations. This shift to crystallization-driven purification is a hallmark of industrial viability, ensuring that the commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal equipment modification. The ability to produce high-purity quinazolinedione derivatives consistently makes this approach highly attractive for contract development and manufacturing organizations (CDMOs) aiming to optimize their production portfolios for anti-inflammatory drugs.
Mechanistic Insights into CDI-Mediated Cyclization
The core of this synthetic innovation revolves around the dual role of 1,1'-carbonyldiimidazole (CDI) as both a coupling agent and a cyclization promoter. In the initial step, CDI activates the amino group of the acylphenylalanine derivative, forming a reactive imidazolide intermediate that readily reacts with the anthranilic acid derivative to form the asymmetric urea bond. This activation mode is superior to traditional phosgene-based methods due to the milder reaction conditions and the non-toxic nature of the imidazole by-product. The reaction proceeds efficiently in polar aprotic solvents like DMF, where the solubility of both the peptide-like substrate and the aromatic acid is maximized, ensuring homogeneous reaction conditions that are critical for reproducibility.
Following urea formation, the same activating agent or a subsequent addition facilitates the intramolecular cyclization to close the quinazolinedione ring. The presence of the free carboxylic acid on the anthranilic moiety is crucial here, as it allows for direct nucleophilic attack on the activated carbonyl center without the need for prior ester hydrolysis. 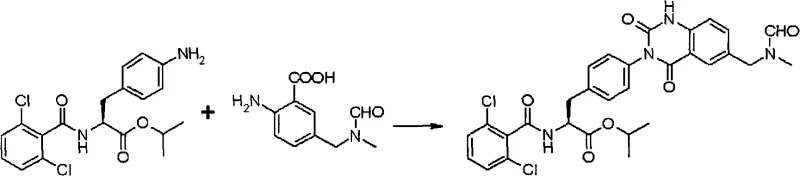
This mechanistic pathway minimizes the formation of oligomeric by-products that often plague peptide couplings, resulting in a cleaner crude reaction profile. The subsequent N-alkylation step, typically performed using methyl p-toluenesulfonate under basic conditions, further functionalizes the scaffold with high regioselectivity. The robustness of this mechanism ensures that even at elevated temperatures required for cyclization, the chiral integrity of the phenylalanine backbone is preserved, which is a non-negotiable requirement for biological activity. Understanding these mechanistic nuances allows process chemists to fine-tune parameters such as temperature and stoichiometry to maximize yield and minimize waste.
How to Synthesize Quinazolinedione Phenylalanine Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure optimal yield and purity. The process generally begins with the activation of the acylphenylalanine derivative followed by coupling with the specific anthranilic acid component. Detailed operational procedures involve careful temperature management during the exothermic activation phase and controlled addition of reagents to prevent localized overheating. The subsequent cyclization and alkylation steps demand strict monitoring of pH and reaction progress via HPLC to determine the exact endpoint, ensuring that over-reaction or degradation does not occur.
- React acylphenylalanine derivative with carbonyl-introducing agent (CDI) and anthranilic acid derivative to form asymmetric urea.
- Convert the urea derivative into quinazolinedione ring using a carboxyl activator in the presence of base.
- Perform N-alkylation using methyl p-toluenesulfonate followed by deprotection to yield the final phenylalanine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers transformative benefits for procurement strategies and supply chain resilience in the pharmaceutical sector. The primary advantage lies in the drastic simplification of the manufacturing workflow, which translates directly into reduced operational expenditures and enhanced throughput. By eliminating unnecessary protection and deprotection steps, manufacturers can significantly reduce the consumption of raw materials and solvents, leading to substantial cost savings in API manufacturing. Furthermore, the high yield associated with the free acid route means that less starting material is required to produce the same amount of final product, optimizing the utilization of expensive chiral building blocks.
- Cost Reduction in Manufacturing: The elimination of ester protection groups removes the need for additional reagents and reaction steps, thereby lowering the overall cost of goods. The high yield observed in the novel process reduces the amount of waste generated, which in turn lowers waste disposal costs and environmental compliance burdens. Additionally, the use of crystallization for purification instead of chromatography significantly reduces solvent consumption and processing time, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent production output, minimizing the risk of batch failures that can disrupt supply chains. The use of commercially available and stable reagents like CDI and methyl p-toluenesulfonate reduces dependency on specialized or hazardous chemicals, enhancing the reliability of the raw material supply. This stability allows for better forecasting and inventory management, ensuring that critical intermediates are available when needed for downstream API synthesis.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from gram to kilogram quantities without loss of efficiency, making it ideal for commercial production. The reduced solvent usage and waste generation align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. The ability to handle the reaction in standard stainless steel reactors without specialized equipment further supports rapid scale-up and technology transfer to manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of quinazolinedione phenylalanine derivatives. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They provide clarity on the specific advantages of this method over traditional approaches and its applicability to large-scale manufacturing environments.
Q: Why is the free carboxylic acid route preferred over the ester route?
A: Using anthranilic acid derivatives with a free carboxylic acid group significantly improves reaction yield compared to ester-protected counterparts, as demonstrated by an increase from 55% to 88% in pilot examples.
Q: What is the key reagent for ring closure in this process?
A: 1,1'-Carbonyldiimidazole (CDI) is the preferred carbonyl-introducing agent and carboxyl activator, facilitating both urea bond formation and subsequent cyclization with minimal by-products.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the patent describes kilogram-scale experiments (e.g., 4.00 kg starting material) with robust crystallization steps, indicating high suitability for commercial scale-up and industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinedione Phenylalanine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate complex synthetic routes like CN101589028A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinazolinedione phenylalanine derivatives meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced technology for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and reduce overall development costs.
