Scalable Production of High-Purity S-Configuration Fluralaner via Mild Catalytic Routes
The pharmaceutical and veterinary industries are witnessing a pivotal shift towards enantiomerically pure active ingredients, driven by regulatory demands for higher efficacy and safety profiles. Patent CN115677605A introduces a groundbreaking synthetic methodology for S-configuration Fluralaner, a potent isoxazoline-class ectoparasiticide widely used in animal health. This innovation addresses critical bottlenecks in existing manufacturing processes by replacing wasteful chiral resolution techniques with a direct, catalytic asymmetric synthesis. By leveraging a chiral thiourea organocatalyst, the process establishes stereochemistry early in the sequence, ensuring that the biologically active S-enantiomer is produced with high fidelity. This technical advancement not only streamlines the production workflow but also aligns with modern green chemistry principles by minimizing waste generation and energy consumption.
For global procurement teams and R&D directors, the implications of this patent are profound. It offers a viable pathway to secure a reliable veterinary drug intermediate supplier capable of delivering high-quality materials without the volatility associated with complex resolution steps. The ability to produce S-Fluralaner with superior enantiomeric purity directly impacts the dosage efficiency of the final veterinary product, potentially lowering the required active ingredient load per treatment. As we delve into the technical specifics, it becomes clear that this methodology represents a significant leap forward in cost reduction in veterinary drug manufacturing, providing a competitive edge for manufacturers who adopt this streamlined approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of S-configuration Fluralaner has been plagued by inefficiencies inherent in chiral resolution strategies. Traditional Route 1, as cited in the background art, relies on separating racemic mixtures using chiral preparative columns or resolving agents. This approach is fundamentally flawed from an economic and sustainability perspective because it inherently caps the maximum theoretical yield at 50%, leaving the unwanted R-enantiomer as waste material that requires costly disposal or complex recycling. Furthermore, alternative synthetic pathways often employ expensive and hazardous reagents, such as hydroxytoluenesulfonyl iodobenzene, which drive up raw material costs significantly. These legacy methods also frequently necessitate harsh reaction conditions, including high-temperature reduction steps ranging from 110°C to 120°C, which impose severe stress on reactor equipment and increase the risk of thermal runaway incidents. The combination of low overall yields, often hovering around 20%, and the need for specialized separation infrastructure creates a fragile supply chain vulnerable to disruptions and price fluctuations.
The Novel Approach
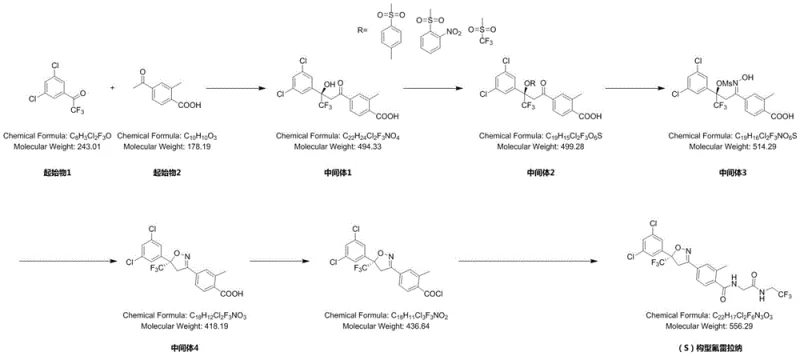
In stark contrast, the methodology disclosed in patent CN115677605A revolutionizes the synthesis landscape by implementing a direct five-step sequence that bypasses the need for late-stage resolution. The core innovation lies in the initial nucleophilic substitution step, which utilizes a chiral thiourea catalyst to induce asymmetry right from the starting materials, specifically 3,5-dichloro-2,2,2-trifluoroacetophenone and 4-acetyl-2-methylbenzoic acid. This strategic design ensures that the chiral center is established with high selectivity before the molecule undergoes further functionalization. The subsequent steps, including hydroxyl protection, oximation, Mitsunobu cyclization, and amide condensation, are all conducted under remarkably mild conditions, typically between 20°C and 30°C. This elimination of extreme thermal requirements not only enhances operational safety but also drastically simplifies the engineering controls needed for commercial scale-up of complex isoxazoline derivatives. By avoiding expensive iodine-based oxidants and utilizing common, cost-effective reagents like sulfonyl chlorides and triphenylphosphine, the new route achieves a total yield exceeding 50%, effectively doubling the output efficiency compared to traditional methods.
Mechanistic Insights into Chiral Thiourea-Catalyzed Nucleophilic Substitution
The heart of this synthetic breakthrough is the enantioselective nucleophilic substitution catalyzed by a chiral thiourea derivative, specifically 1-[3,5-bis(trifluoromethyl)phenyl]-3-[(1R,2R)-(-)-2-(dimethylamino)cyclohexyl]thiourea. This organocatalyst operates through a dual-activation mechanism where the thiourea moiety forms hydrogen bonds with the electrophilic carbonyl oxygen of the acetophenone derivative, thereby increasing its susceptibility to nucleophilic attack. Simultaneously, the tertiary amine group on the cyclohexyl ring interacts with the nucleophile, orienting it in a specific spatial arrangement that favors the formation of the S-configuration stereocenter. This precise molecular recognition is critical for achieving the high enantiomeric excess (ee) values observed, which range from 90% to 94.5% across different embodiments. The robustness of this catalytic system is evident in its tolerance to various solvents, including tetrahydrofuran, toluene, and dichloromethane, allowing process chemists to optimize solubility and reaction kinetics without compromising stereocontrol. Such mechanistic elegance ensures that impurities arising from the wrong enantiomer are minimized at the source, reducing the burden on downstream purification units.
Following the establishment of chirality, the synthesis proceeds through a carefully orchestrated sequence of functional group transformations designed to maintain integrity and purity. The hydroxyl protection step employs sulfonyl chlorides to mask reactive alcohol groups, preventing side reactions during the subsequent oximation with hydroxylamine hydrochloride. The pivotal Mitsunobu reaction then facilitates the intramolecular cyclization to form the isoxazoline ring, a structural motif essential for the biological activity of Fluralaner. This step utilizes triphenylphosphine and an azo compound, such as diethyl azodicarboxylate, to activate the hydroxyl group for nucleophilic displacement by the oxime nitrogen. Finally, the amide condensation couples the heterocyclic core with the trifluoroethylamine side chain using oxalyl chloride activation. Throughout this cascade, the process incorporates multiple crystallization events, which serve as powerful purification drivers to remove triphenylphosphine oxide and other byproducts, ensuring that the final high-purity S-configuration Fluralaner meets stringent HPLC specifications of over 99%.
How to Synthesize S-Configuration Fluralaner Efficiently
The implementation of this synthetic route requires precise control over stoichiometry and temperature to maximize the benefits of the chiral catalyst and minimize byproduct formation. The process begins with the careful addition of the chiral thiourea catalyst to a solution of the ketone and acid starting materials, maintaining the temperature strictly between 20°C and 30°C to preserve catalyst activity and selectivity. Subsequent steps involve standard workup procedures such as acid quenching and solvent exchange, which are well-suited for large-scale manufacturing equipment. The detailed standardized synthesis steps, including specific molar ratios and solvent volumes optimized for industrial throughput, are outlined in the guide below.
- Perform nucleophilic substitution using a chiral thiourea catalyst at 20-30°C to establish stereochemistry.
- Execute hydroxyl protection with sulfonyl chloride followed by hydroxylamine oximation to form the oxime intermediate.
- Conduct Mitsunobu reaction for cyclization and finalize with amide condensation using oxalyl chloride activation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers transformative benefits for supply chain stability and cost management. The shift away from chiral resolution means that manufacturers no longer have to account for the loss of half their raw material input, effectively doubling the mass efficiency of the process. This improvement in atom economy translates directly into substantial cost savings on starting materials, which are often fluorinated aromatics subject to market volatility. Furthermore, the elimination of high-temperature reduction steps removes the need for specialized high-pressure reactors and extensive cooling systems, allowing production to occur in standard glass-lined steel reactors found in most multipurpose chemical plants. This flexibility significantly enhances supply chain resilience by expanding the pool of qualified contract manufacturing organizations (CMOs) capable of producing the intermediate. The use of common, non-proprietary reagents like triphenylphosphine and oxalyl chloride further mitigates the risk of supply disruptions caused by reliance on single-source specialty chemicals.
- Cost Reduction in Manufacturing: The economic advantage of this route is primarily driven by the replacement of expensive hypervalent iodine reagents with inexpensive sulfonyl chlorides and the doubling of overall yield. By avoiding the 50% yield loss inherent in resolution processes, the effective cost per kilogram of the active S-enantiomer is drastically reduced. Additionally, the mild reaction conditions lower utility costs associated with heating and cooling, contributing to a leaner manufacturing budget. The simplified purification strategy, relying on crystallization rather than chromatography, reduces solvent consumption and waste disposal fees, creating a more sustainable and cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of the synthetic pathway ensures consistent quality and delivery timelines, which is critical for meeting the demands of the global veterinary market. Since the process does not depend on rare earth metals or unstable catalysts that require inert atmosphere handling beyond standard nitrogen purging, it is less susceptible to logistical delays. The ability to source raw materials from multiple suppliers due to their commodity status further de-risks the procurement strategy. This reliability allows pharmaceutical companies to maintain lower safety stock levels while ensuring continuous production of the final drug product, optimizing working capital.
- Scalability and Environmental Compliance: The mild operating conditions and absence of heavy metal catalysts make this process highly scalable and environmentally friendly. Scaling from pilot plant to commercial production is straightforward because the exothermic profiles of the reactions are manageable, reducing the risk of thermal hazards during batch enlargement. The reduction in hazardous waste generation, particularly the avoidance of iodine-containing byproducts and heavy metal residues, simplifies regulatory compliance and wastewater treatment. This alignment with green chemistry principles not only lowers environmental fees but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of S-configuration Fluralaner based on the insights from patent CN115677605A. These answers are derived from the specific experimental data and comparative examples provided in the documentation, offering clarity on yield expectations, purity standards, and process safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this new route into their existing manufacturing portfolios.
Q: What distinguishes this S-Fluralaner synthesis from conventional resolution methods?
A: Unlike conventional methods that rely on chiral separation wasting 50% of the R-enantiomer, this patent utilizes asymmetric organocatalysis to directly synthesize the S-configuration, significantly improving atom economy and overall yield.
Q: How does the new route improve safety and cost compared to prior art?
A: The process eliminates the need for expensive hypervalent iodine reagents and avoids high-temperature reduction steps (110-120°C), operating instead at mild ambient temperatures (20-30°C), which reduces energy consumption and equipment stress.
Q: What purity and enantiomeric excess levels are achievable?
A: The optimized protocol consistently delivers HPLC purity exceeding 99% and enantiomeric excess (ee) values ranging from 90% to 94.5%, surpassing the 81% ee typical of older synthetic pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Configuration Fluralaner Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value veterinary intermediates like S-configuration Fluralaner. Our team of expert process chemists has extensively analyzed the methodology described in CN115677605A and possesses the technical capability to implement this advanced catalytic protocol immediately. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including HPLC purity greater than 99% and enantiomeric excess values exceeding 90%, guaranteeing that every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this innovative synthesis technology for your veterinary drug pipeline. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new route can optimize your bottom line. We encourage you to contact us today to request specific COA data from our pilot batches and discuss route feasibility assessments for your project. Let us help you secure a stable, high-quality supply of S-Fluralaner that drives your product's success in the competitive global market.
