Revolutionizing Bentazon Production: A Zero-Wastewater Technical Breakthrough for Global Agrochemical Supply Chains
Revolutionizing Bentazon Production: A Zero-Wastewater Technical Breakthrough for Global Agrochemical Supply Chains
The global agrochemical industry is currently facing immense pressure to adopt greener manufacturing processes that minimize environmental impact while maintaining economic viability. A pivotal advancement in this domain is detailed in patent CN102617511A, which discloses a novel, wastewater-free preparation method for bentazon, a widely used herbicide known for its efficacy against broad-leaved weeds and sedges in crops like rice and wheat. This technology represents a significant departure from traditional synthetic routes that have long been plagued by excessive water consumption and the generation of hazardous organic effluents. By re-engineering the reaction medium and optimizing the stoichiometry of the cyclization steps, this innovation offers a pathway to high-purity bentazon sodium salt with an overall yield exceeding 85%. For R&D directors and supply chain leaders, understanding the mechanistic nuances of this patent is crucial, as it provides a blueprint for sustainable agrochemical intermediate manufacturing that aligns with increasingly stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of bentazon has relied primarily on two pathways: the methyl anthranilate route and the isatoic anhydride route. While the methyl anthranilate route was utilized in early production phases, it suffers from significant drawbacks, including the necessity of using isopropylsulfamoyl chloride, a raw material that is difficult to source and complicates operational safety. Consequently, the industry largely shifted to the isatoic anhydride route; however, this method is not without its own severe environmental liabilities. In the conventional isatoic anhydride process, the aminolysis step generating the amide intermediate typically requires extensive aqueous washing to remove impurities, resulting in substantial volumes of wastewater contaminated with organic pollutants. Furthermore, the subsequent cyclization step involving phosphorus oxychloride and the final purification stages contribute to a massive water footprint, with legacy data indicating that producing just one ton of bentazon can generate between 20 to 30 tons of wastewater. This excessive discharge creates a heavy burden on wastewater treatment facilities and poses a significant compliance risk for manufacturers operating under modern environmental standards.
The Novel Approach
The methodology outlined in patent CN102617511A fundamentally重构 s the synthesis logic to eliminate these inefficiencies at the source. Instead of relying on aqueous workups, the new process utilizes aprotic polar solvents such as N,N-dimethylformamide (DMF), N,N-dimethylacetamide, or dimethyl sulfoxide during the initial aminolysis of isatoic anhydride with isopropylamine. This strategic choice of solvent suppresses side reactions and yields a transparent, homogeneous solution where the resulting amide possesses sufficient purity (>97%) to be used directly in the next step without any water washing. As illustrated in the reaction scheme below, the process flows seamlessly from aminolysis to sulfonation and cyclization, maintaining a closed-loop system for solvent and catalyst management.
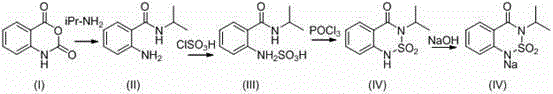
Furthermore, the innovation extends to the cyclization and hydrolysis phases. By carefully controlling the amount of water added during the hydrolysis of the phosphorus oxychloride intermediate—using only 2 to 2.4 equivalents relative to the stoichiometric requirement—the process achieves a theoretical water balance. The byproduct pyridinium salts, which would traditionally constitute waste, are instead converted back into valuable 2-picoline through a high-temperature distillation with lime and caustic soda. This holistic approach not only eradicates wastewater discharge but also enhances the atom economy of the entire synthesis, making it a superior choice for cost reduction in agrochemical manufacturing.
Mechanistic Insights into Isatoic Anhydride Aminolysis and Cyclization
To fully appreciate the technical robustness of this method, one must examine the specific chemical mechanisms that enable such high selectivity and yield. The initial aminolysis of isatoic anhydride is a nucleophilic acyl substitution reaction. In traditional aqueous or protic environments, the competitive hydrolysis of the anhydride can lead to the formation of anthranilic acid, a persistent impurity that complicates downstream purification. By employing aprotic polar solvents, the nucleophilicity of the isopropylamine is preserved while the hydrolytic degradation of the starting material is effectively suppressed. The reaction is conducted at a moderate temperature range of 50-60°C, which provides sufficient kinetic energy to drive the ring-opening of the anhydride without promoting thermal decomposition. The result is a crude amide intermediate with a yield greater than 99% and purity exceeding 97%, eliminating the need for the water-intensive washing steps that characterize older protocols.
The subsequent sulfonation and cyclization sequence is equally critical for impurity control. The formation of the benzothiadiazinone ring system requires precise activation of the aromatic ring followed by intramolecular closure. In this patent, chlorosulfonic acid is complexed with 2-picoline in dichloroethane to form a reactive double salt, which acts as a controlled sulfonating agent. This prevents the harsh, uncontrolled sulfonation that can lead to polysulfonated byproducts. Following sulfonation, phosphorus oxychloride (POCl3) serves as the dehydrating agent to facilitate ring closure. Crucially, the hydrolysis of the excess POCl3 and the intermediate imidoyl chloride is managed by adding a strictly limited amount of water (2-2.4 times the stoichiometric ratio) at 50°C. This precise dosing ensures that the water is consumed entirely by the hydrolysis reaction to form phosphoric and hydrochloric acids, which then react with the 2-picoline to form salts, leaving no free water to generate wastewater. This mechanistic precision is what allows the process to claim zero wastewater discharge while maintaining a high overall yield of over 85%.
How to Synthesize Bentazon Efficiently
Implementing this wastewater-free synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the dissolution of isatoic anhydride in a selected aprotic solvent, followed by the controlled addition of isopropylamine to generate the key amide intermediate. Subsequent steps involve the low-temperature formation of the sulfonating complex and the careful thermal management of the cyclization reflux. The final isolation of the bentazon sodium salt is achieved through direct alkaline extraction, bypassing the need for crystallization and drying of the acid form if the aqueous formulation is the desired end product. For process engineers looking to adopt this technology, the detailed standardized synthesis steps are provided in the guide below, which outlines the specific molar ratios and temperature profiles required for successful scale-up.
- Dissolve isatoic anhydride and isopropylamine in an aprotic polar solvent (e.g., DMF) at 50-60°C to form the amide intermediate without aqueous washing.
- React the amide with a chlorosulfonic acid/2-picoline complex in dichloroethane, followed by cyclization with phosphorus oxychloride.
- Perform controlled hydrolysis with minimal water, separate the pyridinium salt layer for 2-picoline recovery, and extract the product with NaOH to obtain the aqueous solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling strategic advantages that extend beyond mere regulatory compliance. The most significant benefit lies in the drastic simplification of the waste management infrastructure required for production. By eliminating the generation of 20 to 30 tons of wastewater per ton of product, manufacturers can significantly reduce the capital expenditure associated with effluent treatment plants and lower the ongoing operational costs related to wastewater disposal fees and environmental monitoring. This reduction in environmental liability translates directly into a more resilient supply chain, as facilities are less likely to face production halts due to environmental violations or capacity constraints in local waste treatment facilities. Moreover, the ability to recover and reuse 2-picoline with a recovery rate greater than 95% reduces the dependency on external raw material suppliers, thereby insulating the production process from market volatility in reagent pricing.
- Cost Reduction in Manufacturing: The elimination of aqueous washing steps and the recovery of expensive catalysts like 2-picoline lead to substantial savings in raw material consumption and utility usage. By avoiding the energy-intensive processes required to treat large volumes of organic-laden wastewater, the overall cost of goods sold (COGS) is optimized. Additionally, the high purity of the intermediate amide allows for a telescoped process where isolation steps are minimized, further reducing labor and solvent costs associated with multiple unit operations.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as isatoic anhydride and isopropylamine, combined with common industrial solvents like dichloroethane and DMF, ensures a stable supply base. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without significant yield loss, enhances the reliability of batch-to-batch production. This stability is critical for meeting the rigorous delivery schedules demanded by global agrochemical formulators, ensuring a continuous flow of high-purity agrochemical intermediates to the market.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reactor configurations and avoiding exotic catalysts or extreme pressure conditions. The "zero wastewater" claim is not merely a marketing slogan but a chemically engineered reality that positions manufacturers favorably in regions with strict environmental enforcement. This compliance advantage facilitates smoother permitting processes for capacity expansion and reduces the risk of regulatory shutdowns, securing long-term supply continuity for partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this wastewater-free bentazon synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN102617511A, providing a reliable foundation for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this greener manufacturing route.
Q: How does this patent address the wastewater issue in traditional bentazon production?
A: The patent eliminates the aqueous washing step after aminolysis by using aprotic polar solvents and achieves water balance during the hydrolysis/cyclization phase, preventing the generation of 20-30 tons of wastewater per ton of product typical in older methods.
Q: What is the overall yield and purity achievable with this new method?
A: According to the experimental data in patent CN102617511A, the process achieves an overall yield of over 85% with product purity reaching 98% (HPLC) after acidification, demonstrating high efficiency suitable for industrial scale-up.
Q: Can the 2-picoline catalyst be recovered in this process?
A: Yes, the pyridinium salt layer formed during the reaction is treated with a mixture of calcium oxide and sodium hydroxide (2:1 molar ratio) and distilled at 230°C, allowing for the recovery of 2-picoline with a recovery rate greater than 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bentazon Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic methodologies like the one described in CN102617511A requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO specializing in complex organic synthesis, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this innovative bentazon process to the global market. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this zero-wastewater protocol, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We are committed to delivering not just a chemical product, but a sustainable supply solution that aligns with your corporate responsibility goals.
We invite you to collaborate with us to leverage this breakthrough technology for your agrochemical portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can improve your margins. Please contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a reliable, cost-effective, and environmentally responsible supply of bentazon for the seasons ahead.
