Scalable Synthesis of 2-(3,3,3-Trifluoropropylthio)Adenosine for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly for antithrombotic agents like Cangrelor. A pivotal advancement in this domain is documented in patent CN108033983B, which details a highly efficient method for synthesizing 2-(3,3,3-trifluoropropylthio)adenosine. This compound serves as a vital building block in the production of P2Y12 receptor antagonists, necessitating a supply chain that guarantees both chemical integrity and commercial viability. The disclosed technology overcomes historical bottlenecks associated with nucleoside modification, offering a pathway that aligns with modern green chemistry principles while ensuring the rigorous quality standards demanded by global regulatory bodies. By leveraging a unique combination of inorganic bases and mixed solvent systems, this approach transforms a traditionally hazardous process into a manageable, high-yielding operation suitable for industrial application.
For R&D directors and procurement specialists evaluating potential partners, understanding the technical nuances of this synthesis is paramount. The shift from legacy methods to this novel protocol represents not just a chemical improvement but a strategic supply chain advantage. As a reliable pharmaceutical intermediates supplier, analyzing such patents allows us to anticipate production challenges and optimize cost structures before they impact the final API. The ability to produce high-purity pharmaceutical intermediates with reduced environmental footprint is no longer optional but a prerequisite for sustaining long-term partnerships in the competitive landscape of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoropropylthioadenosine has been plagued by severe operational constraints that hinder industrial scalability. Prior art, such as the methodology reported by Anthony H et al., relies heavily on the utilization of sodium hydride (NaH) as a strong base in anhydrous dimethylformamide (DMF) environments. This conventional route, depicted in the reaction scheme below, necessitates stringent moisture exclusion to prevent reagent decomposition and safety incidents. The reliance on NaH introduces significant pyrophoric risks, requiring specialized handling equipment and inert atmosphere protocols that drastically inflate capital expenditure and operational overheads. Furthermore, the use of high-boiling solvents like DMF complicates downstream processing, often requiring energy-intensive distillation or extensive aqueous workups to remove residual solvent traces that could contaminate the final product.
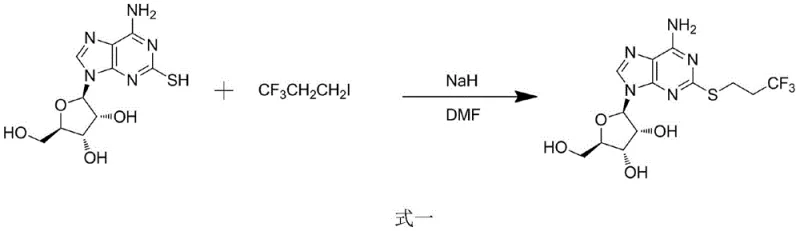
Beyond safety concerns, the conventional anhydrous approach frequently suffers from inconsistent yield profiles and poor atom economy. The harsh reaction conditions can promote side reactions, leading to complex impurity spectra that are difficult to purge during purification. For a procurement manager, this translates to higher rejection rates, increased waste disposal costs, and unpredictable batch-to-batch variability. The necessity for dry solvents and specialized reactors limits the number of qualified manufacturers capable of executing this chemistry, creating supply chain bottlenecks. Consequently, the traditional thought that trifluoropropylthioadenosine must be synthesized under prolonged anhydrous conditions has become a barrier to cost reduction in API manufacturing, forcing companies to accept higher prices for intermediates produced via these inefficient legacy pathways.
The Novel Approach
In stark contrast, the innovative method described in patent CN108033983B revolutionizes the production landscape by introducing a mixed solvent system comprising water and methanol alongside inexpensive inorganic bases. This novel approach, illustrated in the following reaction diagram, eliminates the need for hazardous sodium hydride and strictly anhydrous conditions, thereby democratizing the manufacturing process. By utilizing bases such as sodium hydroxide or potassium carbonate, the reaction proceeds smoothly at moderate temperatures between 70°C and 100°C, significantly reducing energy consumption and thermal stress on the equipment. The introduction of water into the solvent matrix not only acts as a co-solvent to enhance substrate solubility but also facilitates the ionization of the thiol group, promoting a more efficient nucleophilic attack on the trifluoropropyl chloride electrophile.
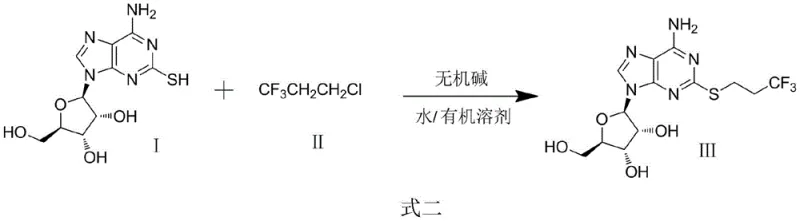
The operational simplicity of this new route offers profound commercial advantages for supply chain heads seeking reliability. The post-treatment process is remarkably straightforward; the product precipitates directly upon cooling the reaction mixture in an ice-water bath, allowing for simple filtration to isolate the crude material. This crystallization-driven isolation minimizes the need for chromatographic purification, which is often a major cost driver in nucleoside chemistry. Furthermore, the subsequent washing with cyclohexane effectively removes organic impurities without generating large volumes of hazardous waste. This streamlined workflow ensures that the yield of the obtained target product can reach impressive levels, up to 97.5%, while maintaining a purity profile that meets stringent specifications. The transition to this aqueous-compatible chemistry represents a paradigm shift, enabling the commercial scale-up of complex nucleoside analogs with unprecedented efficiency and safety.
Mechanistic Insights into Inorganic Base-Catalyzed Thioalkylation
To fully appreciate the technical superiority of this synthesis, one must delve into the mechanistic underpinnings of the inorganic base-catalyzed thioalkylation. In this system, the inorganic base plays a dual role: it acts as a proton scavenger to deprotonate the sulfhydryl group of adenosine-2-thione, generating a highly reactive thiolate anion, and it stabilizes the transition state within the polar protic solvent environment. Unlike strong, non-nucleophilic bases used in anhydrous conditions, inorganic hydroxides or carbonates in a methanol-water mixture create a buffered environment that suppresses competing elimination reactions of the alkyl halide. The methanol component enhances the solubility of the organic substrate, while water facilitates the dissociation of the inorganic salt, ensuring a high concentration of active nucleophiles is available throughout the reaction duration. This synergistic solvent effect promotes the formation of the active intermediate, driving the condensation reaction forward with high selectivity for the S-alkylation product over N-alkylation byproducts.
Impurity control is another critical aspect where this mechanism excels, providing R&D teams with confidence in the product's quality. The mild basicity of the inorganic reagents prevents the degradation of the sensitive ribose moiety, which is prone to epimerization or hydrolysis under harsher alkaline conditions. Additionally, the specific temperature window of 70-100°C is optimized to balance reaction kinetics with thermal stability, minimizing the formation of bis-alkylated side products or decomposition of the trifluoropropyl group. The precipitation step upon cooling leverages the differential solubility of the product versus the starting materials and inorganic salts in the cold mixed solvent, acting as an inherent purification stage. This mechanistic robustness ensures that reducing lead time for high-purity intermediates does not come at the expense of chemical integrity, making the process ideal for GMP-compliant manufacturing environments where trace impurities must be rigorously controlled.
How to Synthesize 2-(3,3,3-Trifluoropropylthio)Adenosine Efficiently
Implementing this synthesis at a production scale requires precise adherence to the optimized parameters defined in the patent to maximize yield and safety. The process begins with the preparation of a homogeneous reaction mixture, where the stoichiometry of the inorganic base is carefully calibrated to ensure complete deprotonation without excess alkalinity that could harm the product. Operators must monitor the exotherm during the addition of the alkyl halide to maintain the temperature within the specified 0-5°C range initially, preventing runaway reactions. Following the addition, the system is heated to the optimal reaction temperature, typically around 90°C, and maintained for a duration of approximately 10 hours to ensure full conversion as monitored by TLC. The detailed standardized synthesis steps, including specific mixing rates, cooling profiles, and filtration protocols, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- Dissolve adenosine-2-thione and inorganic base in a methanol-water mixture at 55°C.
- Cool to 0-5°C and add 3,3,3-trifluoropropyl chloride dropwise, then heat to 70-100°C for 10 hours.
- Cool the reaction, filter the precipitate, and purify via cyclohexane reflux to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of hazardous reagents like sodium hydride and high-boiling solvents like DMF fundamentally alters the cost structure of the intermediate. By removing the requirement for specialized anhydrous infrastructure and expensive waste treatment protocols associated with halogenated or amide solvents, the overall manufacturing overhead is significantly reduced. This cost reduction in pharmaceutical intermediates manufacturing allows for more competitive pricing models without compromising on quality, providing a buffer against raw material price fluctuations. Furthermore, the use of widely available inorganic bases and common solvents like methanol and water enhances supply chain resilience, reducing the risk of disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The transition to an aqueous-organic solvent system eliminates the need for costly solvent recovery units required for DMF, leading to substantial operational savings. The simplified workup procedure, which relies on precipitation and filtration rather than complex extractions or chromatography, reduces labor hours and consumable usage per batch. Additionally, the high yield of up to 97.5% minimizes raw material waste, ensuring that every kilogram of starting adenosine-2-thione is converted efficiently into valuable product. These factors combine to lower the cost of goods sold (COGS), enabling more aggressive pricing strategies in the global market while maintaining healthy margins for sustained investment in quality control.
- Enhanced Supply Chain Reliability: Utilizing common inorganic bases and standard solvents mitigates the risk of supply bottlenecks often associated with specialty chemicals. The robustness of the reaction conditions means that production can be scaled across multiple facilities without requiring extensive requalification of equipment, ensuring continuity of supply even during regional disruptions. The simplified safety profile reduces insurance premiums and regulatory compliance burdens, further stabilizing the supply chain. This reliability is crucial for downstream API manufacturers who depend on just-in-time delivery of critical intermediates to maintain their own production schedules, making this method a preferred choice for long-term contractual agreements.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction parameters that translate linearly from laboratory to pilot to commercial plant scales. The use of water as a primary solvent component aligns with green chemistry initiatives, reducing the environmental footprint of the manufacturing process and simplifying wastewater treatment. The absence of heavy metals or persistent organic pollutants in the waste stream facilitates easier regulatory approval and disposal. This environmental compliance not only protects the manufacturer from potential fines but also enhances the brand reputation of the supply chain partners, appealing to end-clients who prioritize sustainability in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-(3,3,3-trifluoropropylthio)adenosine, derived directly from the patent data and practical manufacturing experience. These insights are intended to clarify the feasibility of the process for potential partners and highlight the specific advantages over traditional methods. Understanding these details is essential for making informed decisions about sourcing strategies and technology transfer.
Q: Why is the inorganic base method superior to sodium hydride for this synthesis?
A: The inorganic base method eliminates the need for hazardous anhydrous conditions and expensive DMF solvents, significantly improving safety and reducing waste treatment costs while maintaining high yields up to 97.5%.
Q: What is the expected purity profile for this intermediate?
A: The novel aqueous-organic solvent system facilitates effective crystallization, resulting in high-purity products with minimal impurity profiles suitable for downstream API synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of simple equipment, non-hazardous reagents, and straightforward workup procedures makes this method highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(3,3,3-Trifluoropropylthio)Adenosine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antithrombotic therapies depends on a secure and high-quality supply of key intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are committed to delivering 2-(3,3,3-trifluoropropylthio)adenosine with stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to accommodate the specific requirements of nucleoside chemistry, providing a safe and compliant environment for the manufacture of complex pharmaceutical intermediates.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this optimized route for your specific volume requirements. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project needs. Together, we can drive down costs and accelerate the availability of life-saving medications through superior chemical manufacturing.
