Advanced Asymmetric Difluoromethylation for High-Value Pharmaceutical Intermediates
Advanced Asymmetric Difluoromethylation for High-Value Pharmaceutical Intermediates
The integration of fluorine atoms into organic molecules has revolutionized modern medicinal chemistry, significantly enhancing metabolic stability and bioavailability. Patent CN113072456A introduces a groundbreaking methodology for the synthesis of chiral alpha-difluoromethyl amino acid compounds, addressing a critical gap in the production of high-value pharmaceutical intermediates. This technology leverages a sophisticated copper-catalyzed asymmetric difluoromethylation reaction to construct quaternary carbon centers with exceptional stereocontrol. Unlike traditional approaches that often yield racemic mixtures requiring costly resolution, this novel route delivers optically active compounds directly. The structural versatility of the resulting products, encompassing formulas I, II, and III as depicted below, underscores their potential utility in developing next-generation enzyme inhibitors and therapeutic agents.
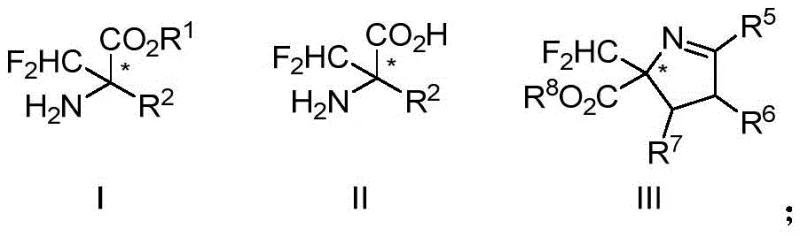
For R&D directors seeking reliable pharmaceutical intermediate suppliers, this patent represents a paradigm shift in accessing complex fluorinated building blocks. The ability to precisely install a difluoromethyl group at the alpha-position of amino acids opens new avenues for drug discovery, particularly for targets like Ornithine Decarboxylase (ODC). The method's robustness across diverse substrates ensures that research teams can rapidly generate libraries of chiral fluorinated analogs for structure-activity relationship studies without being bottlenecked by synthetic complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-difluoromethyl amino acids has been plagued by significant chemical challenges that hinder efficient production. Conventional routes frequently rely on harsh reaction conditions, including the use of strong bases and multistep sequences that degrade sensitive functional groups. Furthermore, many existing methods fail to control stereochemistry effectively, resulting in racemic outputs that necessitate additional downstream purification steps such as chiral chromatography or enzymatic resolution. These extra steps not only drastically increase manufacturing costs but also reduce overall yield and extend lead times. The reliance on unstable difluoromethylating reagents in older protocols further complicates process safety and scalability, making it difficult for procurement managers to secure consistent supplies of high-purity materials for clinical trials.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles through a streamlined, one-pot asymmetric catalytic process. By utilizing a chiral phosphine ligand complexed with a copper catalyst, the reaction achieves direct difluoromethylation of aldimine esters with remarkable precision. As illustrated in the reaction scheme below, the process converts simple starting materials into complex chiral intermediates in a single operational step. This approach eliminates the need for pre-functionalized chiral auxiliaries or post-synthetic resolution, thereby simplifying the workflow significantly. The reaction proceeds under mild conditions, typically between 0°C and 50°C, which enhances operational safety and reduces energy consumption compared to cryogenic alternatives. This efficiency translates directly into cost reduction in API manufacturing, allowing producers to deliver high-enantiomeric excess products with superior economic viability.
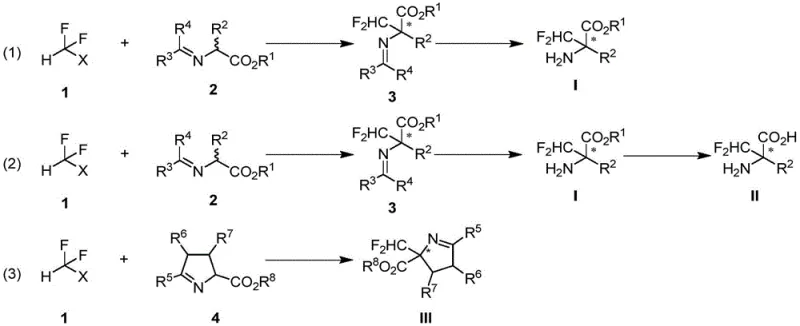
Mechanistic Insights into Copper-Catalyzed Asymmetric Difluoromethylation
The core of this technological breakthrough lies in the unique interaction between the copper catalyst and the specialized chiral phosphine ligands. The catalyst system, formed by reacting a metal precursor such as tetrakis(acetonitrile)copper(I) tetrafluoroborate with a ferrocenyl-based chiral ligand, creates a highly defined chiral environment around the metal center. This environment dictates the facial selectivity of the nucleophilic attack by the difluoromethyl species onto the imine substrate. The ligand structure, featuring bulky substituents on the oxazoline ring and the ferrocene backbone, provides the necessary steric hindrance to block one face of the substrate while directing the reagent to the other. This precise spatial arrangement is critical for achieving the high enantiomeric excess values reported, often exceeding 90% ee across a wide range of substrates.
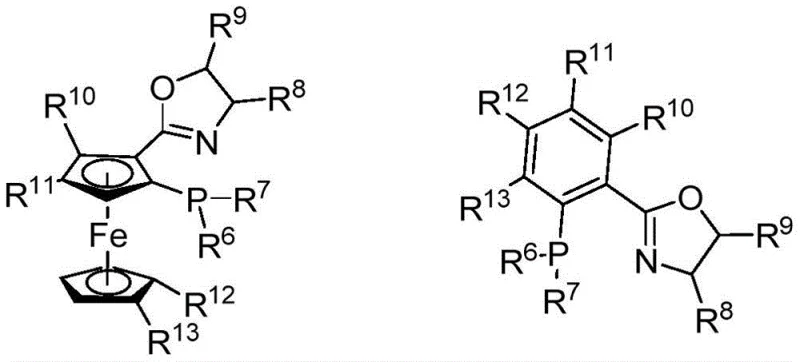
From an impurity control perspective, the mechanism offers distinct advantages by minimizing side reactions. The mild basicity of the system, often employing cesium carbonate, prevents the epimerization of the sensitive alpha-carbon center, a common issue in amino acid chemistry. Additionally, the high turnover frequency of the copper catalyst ensures that the reaction reaches completion within a reasonable timeframe, typically 24 to 96 hours, reducing the exposure of the product to potentially degrading conditions. For quality assurance teams, this means a cleaner crude reaction profile with fewer byproducts, simplifying the final purification via column chromatography or crystallization. The result is a high-purity pharmaceutical intermediate that meets stringent regulatory specifications for chiral drugs.
How to Synthesize Chiral Alpha-Difluoromethyl Amino Acids Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. The process begins with the in situ generation of the active catalyst species under an inert argon atmosphere to prevent oxidation of the copper center. Subsequently, the aldimine ester substrate and the difluoromethyl source are introduced alongside a stoichiometric amount of base. The reaction mixture is then stirred at controlled temperatures until thin-layer chromatography confirms full conversion of the starting material. Following the reaction, a straightforward acidic workup hydrolyzes the intermediate imine to release the free amine, which is then isolated through standard extraction and purification techniques. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during implementation.
- Prepare the catalyst by mixing a metal precursor like Cu(MeCN)4BF4 with a chiral ferrocenyl phosphine ligand in an organic solvent under inert atmosphere.
- Add the aldimine ester substrate and a base such as cesium carbonate to the reaction mixture along with the difluoromethylating agent.
- Stir the reaction at ambient temperature until completion, followed by acid hydrolysis and standard extraction and purification protocols to isolate the target amino acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers transformative benefits regarding cost structure and supply reliability. The elimination of chiral resolution steps represents a major reduction in processing time and solvent usage, directly lowering the cost of goods sold. By avoiding the need for expensive chiral resolving agents or preparative HPLC columns, manufacturers can achieve substantial cost savings while maintaining high throughput. Furthermore, the use of commercially available copper salts and ligands ensures that the supply chain is not dependent on proprietary or scarce reagents, mitigating the risk of raw material shortages. This accessibility allows for more flexible sourcing strategies and better negotiation leverage with upstream chemical suppliers.
- Cost Reduction in Manufacturing: The streamlined one-step nature of this synthesis significantly reduces operational expenditures by cutting down on unit operations. Traditional multi-step routes often involve isolation and purification of intermediates, each adding labor, equipment time, and material costs. In contrast, this telescoped process minimizes handling and maximizes atom economy. The high yields and enantioselectivity mean less waste generation and lower disposal costs, contributing to a leaner and more profitable manufacturing model. Additionally, the mild reaction conditions reduce energy demands for heating or cooling, further enhancing the economic efficiency of large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is crucial for maintaining uninterrupted supply to downstream API manufacturers. Since the process does not require extreme temperatures or pressures, it can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure reduces the need for capital investment in specialized equipment. Moreover, the broad substrate scope means that the same catalytic platform can be adapted to produce various derivatives, allowing suppliers to respond quickly to changing market demands for different fluorinated amino acid variants without retooling entire production lines.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous reagents. The use of common organic solvents like THF or acetonitrile simplifies solvent recovery and recycling programs, aligning with green chemistry principles. Reduced waste generation and lower energy consumption contribute to a smaller carbon footprint, helping companies meet increasingly strict environmental regulations. The ability to produce high-purity chiral intermediates efficiently supports the rapid scale-up required for late-stage clinical development and commercial launch, ensuring that supply can keep pace with market growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric difluoromethylation technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating the process for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this method into existing production workflows.
Q: What is the primary advantage of this copper-catalyzed method over traditional synthesis?
A: The primary advantage is the ability to directly introduce a chiral difluoromethyl group with high enantioselectivity (up to 98% ee) under mild conditions, eliminating the need for difficult resolution of racemic mixtures.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the process utilizes readily available reagents and operates at temperatures ranging from 0°C to 50°C, making it highly suitable for commercial scale-up without requiring extreme cryogenic conditions.
Q: What types of substrates are compatible with this asymmetric difluoromethylation strategy?
A: The method demonstrates broad substrate scope, successfully converting various aldimine esters including those with alkyl, aryl, benzyl, and heterocyclic side chains into the corresponding chiral alpha-difluoromethyl amino acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alpha-Difluoromethyl Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination technologies in the development of modern therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We are committed to delivering high-purity chiral alpha-difluoromethyl amino acids that meet rigorous QC labs standards and stringent purity specifications. Our state-of-the-art facilities are equipped to handle the specific requirements of copper-catalyzed reactions, guaranteeing consistent quality and reliability for your critical projects.
We invite you to collaborate with us to leverage this innovative synthesis route for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timelines and optimize your supply chain economics.
