Advanced Dinaphthoazepinium Salts for Scalable Enantioselective Epoxidation Manufacturing
Advanced Dinaphthoazepinium Salts for Scalable Enantioselective Epoxidation Manufacturing
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the urgent need for cleaner, more sustainable catalytic processes that eliminate heavy metal residues. Patent CN1198157A introduces a groundbreaking class of dinaphthoazepinium salts, specifically designed to serve as highly efficient organocatalysts for the enantioselective epoxidation of prochiral olefins. Unlike traditional transition-metal complexes that often require rigorous purification steps to meet pharmaceutical standards, these novel organic salts operate through a distinct oxaziridinium mechanism that ensures exceptional stereocontrol without metal contamination. The core innovation lies in the unique structural rigidity provided by the binaphthyl backbone, which creates a well-defined chiral environment essential for inducing high enantiomeric excess in the resulting epoxide products. 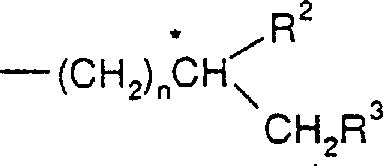 This structural motif allows for precise tuning of steric and electronic properties by modifying substituents A, B, and R1, thereby optimizing the catalyst for a wide range of substrate classes including styrenes and cyclic alkenes. For R&D directors focused on impurity profiles, this metal-free approach represents a paradigm shift that simplifies regulatory filings and reduces the burden of validation for residual metal limits.
This structural motif allows for precise tuning of steric and electronic properties by modifying substituents A, B, and R1, thereby optimizing the catalyst for a wide range of substrate classes including styrenes and cyclic alkenes. For R&D directors focused on impurity profiles, this metal-free approach represents a paradigm shift that simplifies regulatory filings and reduces the burden of validation for residual metal limits.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on metalloporphyrin and Salen-manganese complexes, such as the famous Jacobsen catalyst, to achieve asymmetric epoxidation. While effective, these transition-metal-based systems suffer from inherent drawbacks that complicate large-scale manufacturing and increase overall production costs. The primary concern is the potential leaching of toxic heavy metals like manganese or chromium into the final product, necessitating expensive and time-consuming scavenging procedures to meet stringent ICH Q3D guidelines for elemental impurities. Furthermore, these metal catalysts often operate via radical intermediates, which can lead to non-stereospecific side reactions and lower selectivity when dealing with sensitive substrates that lack pi-stabilizing groups. The requirement for specific co-oxidants and the sensitivity of metal centers to moisture or air also impose strict operational constraints that limit process flexibility and robustness in a commercial plant setting.
The Novel Approach
The technology disclosed in CN1198157A offers a compelling alternative by utilizing purely organic dinaphthoazepinium salts that function as precursors to active oxaziridinium species. This approach completely bypasses the need for transition metals, thereby eliminating the risk of metal contamination and the associated downstream purification costs. The reaction mechanism is stereospecific, meaning cis-alkenes yield cis-epoxides and trans-alkenes yield trans-epoxides with high fidelity, a feature that is critical for maintaining the structural integrity of complex pharmaceutical intermediates. Moreover, the catalytic cycle utilizes inexpensive and environmentally benign oxidants such as potassium hydrogen persulfate (KHSO5) in combination with sodium bicarbonate, which are readily available on a global scale. This shift to organocatalysis not only enhances the safety profile of the manufacturing process but also aligns perfectly with green chemistry principles by reducing the generation of hazardous heavy metal waste streams.
Mechanistic Insights into Oxaziridinium-Mediated Oxygen Transfer
The efficacy of these dinaphthoazepinium salts stems from their ability to undergo reversible oxidation to form highly reactive oxaziridinium ions, which act as the actual oxygen transfer agents. In the presence of a nucleophilic oxidant like KHSO5, the iminium salt catalyst is converted into the oxaziridinium species, which then transfers an oxygen atom to the olefinic substrate in a concerted manner. This non-radical pathway is crucial because it prevents the scrambling of stereochemistry that often plagues radical-based oxidation methods, ensuring that the chiral information encoded in the catalyst backbone is faithfully transferred to the product. The rigid binaphthyl structure locks the catalyst into a specific conformation that shields one face of the reactive oxaziridinium ring, forcing the substrate to approach from the less hindered side and thus dictating the enantioselectivity of the reaction. 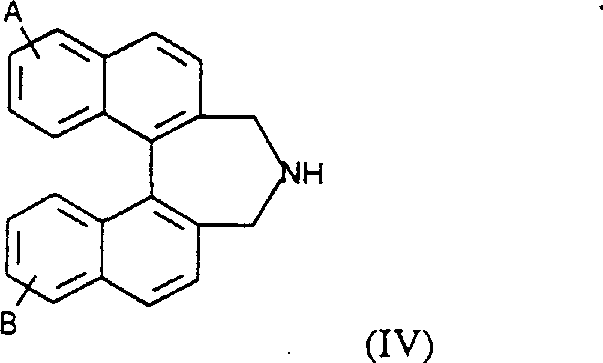 Understanding this mechanistic nuance is vital for process chemists, as it highlights the importance of maintaining anhydrous conditions during catalyst preparation to prevent hydrolysis of the active iminium species before the catalytic cycle begins. The stability of the oxaziridinium intermediate is finely balanced, allowing for rapid turnover while minimizing decomposition pathways that could lead to catalyst deactivation or the formation of achiral byproducts.
Understanding this mechanistic nuance is vital for process chemists, as it highlights the importance of maintaining anhydrous conditions during catalyst preparation to prevent hydrolysis of the active iminium species before the catalytic cycle begins. The stability of the oxaziridinium intermediate is finely balanced, allowing for rapid turnover while minimizing decomposition pathways that could lead to catalyst deactivation or the formation of achiral byproducts.
From an impurity control perspective, the absence of metal ions means that the primary impurities are likely to be organic in nature, such as over-oxidized catalyst species or unreacted starting materials, which are generally easier to separate via standard chromatographic or crystallization techniques. The patent data indicates that the reaction proceeds efficiently at neutral to alkaline pH levels, typically between pH 8 and 10, which helps to stabilize the oxaziridinium intermediate against acid-catalyzed degradation. This pH tolerance provides a wider operating window for manufacturing, reducing the risk of batch failures due to minor fluctuations in reaction conditions. Additionally, the use of aqueous acetonitrile as a solvent system facilitates the dissolution of both the organic substrate and the inorganic oxidant, creating a homogeneous reaction environment that maximizes mass transfer and reaction rates without the need for phase transfer catalysts.
How to Synthesize Dinaphthoazepinium Salts Efficiently
The synthesis of these high-value chiral catalysts involves a logical sequence of transformations starting from readily available chiral amines, leveraging robust organic reactions that are well-suited for scale-up. The process begins with the oxidation of a chiral amine precursor to the corresponding imine, followed by quaternization to install the necessary cationic charge that defines the azepinium salt structure. Each step is optimized to maximize yield and optical purity, ensuring that the final catalyst performs consistently in demanding asymmetric transformations. The detailed synthetic pathway outlined in the patent demonstrates the feasibility of constructing the complex binaphthyl framework through coupling reactions and subsequent functional group manipulations. 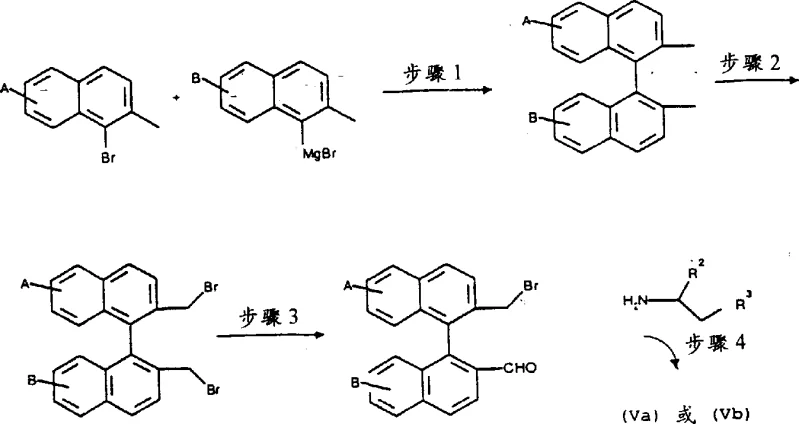 This comprehensive route provides a clear blueprint for manufacturing teams to establish a reliable supply of the catalyst, minimizing dependency on external sources and securing the intellectual property around the production process. By controlling the stereochemistry at the early stages of the synthesis, manufacturers can ensure that the final catalyst possesses the requisite chirality to induce the desired enantioselectivity in the epoxidation reaction.
This comprehensive route provides a clear blueprint for manufacturing teams to establish a reliable supply of the catalyst, minimizing dependency on external sources and securing the intellectual property around the production process. By controlling the stereochemistry at the early stages of the synthesis, manufacturers can ensure that the final catalyst possesses the requisite chirality to induce the desired enantioselectivity in the epoxidation reaction.
- Oxidize the chiral amine precursor (Formula IV) using potassium permanganate in THF to generate the corresponding imine intermediate.
- React the resulting imine with an alkylating agent such as trimethyloxonium tetrafluoroborate in anhydrous dichloromethane under inert atmosphere.
- Precipitate the final dinaphthoazepinium salt by adding diethyl ether to the reaction mixture and dry under high vacuum to obtain the pure catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic technology presents a strategic opportunity to optimize manufacturing costs and mitigate supply risks associated with critical raw materials. The elimination of precious or toxic metals from the process removes a significant cost center related to metal scavenging resins, specialized waste disposal, and extensive analytical testing for residual metals. Furthermore, the reagents required for this process, such as potassium persulfate and simple alkylating agents, are commodity chemicals with stable global supply chains, reducing the vulnerability to price volatility often seen with specialized ligands or rare earth metals. The ability to conduct reactions at ambient temperatures also translates to substantial energy savings, as there is no need for energy-intensive heating or cryogenic cooling systems that are typical for many metal-catalyzed asymmetric reactions.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost structure of asymmetric epoxidation by removing the need for expensive transition metal salts and complex chiral ligands that are often difficult to synthesize. By utilizing simple organic salts that can be prepared in high yields from abundant starting materials, the overall cost of goods sold for the chiral intermediate is significantly reduced. Additionally, the simplified workup procedure, which avoids metal scavenging steps, shortens the production cycle time and reduces the consumption of auxiliary materials like filtration aids and specialized resins. This streamlined process flow enhances overall plant throughput and allows for better utilization of existing manufacturing assets without the need for major capital investment in new equipment.
- Enhanced Supply Chain Reliability: Relying on organocatalysts derived from common organic building blocks insulates the supply chain from the geopolitical and logistical uncertainties that frequently affect the availability of rare metals. The synthetic route described in the patent utilizes standard chemical transformations that can be easily replicated by multiple contract manufacturing organizations, fostering a competitive supplier base and preventing single-source bottlenecks. The robustness of the catalyst under various storage and handling conditions further ensures that inventory can be maintained with minimal degradation, providing a buffer against unexpected demand surges. This reliability is crucial for long-term project planning and ensures continuity of supply for critical drug substances that depend on these chiral building blocks.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on mild reaction conditions and common solvents like acetonitrile and water, which are easily managed in large-scale reactors. The absence of heavy metals simplifies environmental compliance and waste treatment, as the effluent streams do not require specialized processing to remove toxic metal ions before discharge. This alignment with green chemistry principles not only reduces the environmental footprint of the manufacturing operation but also enhances the corporate sustainability profile, which is increasingly important for meeting the ESG goals of major pharmaceutical clients. The ease of scale-up minimizes the technical risks associated with technology transfer from lab to pilot to commercial production, accelerating the time to market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of dinaphthoazepinium salts in industrial processes, based on the specific capabilities and limitations detailed in the patent literature. These insights are intended to clarify the operational parameters and benefits for stakeholders evaluating this technology for integration into their existing manufacturing platforms. Understanding these nuances is essential for making informed decisions about process development and resource allocation.
Q: How do these organocatalysts compare to traditional metal-based systems?
A: Unlike Jacobsen-type manganese complexes, these dinaphthoazepinium salts operate via a non-radical oxaziridinium mechanism, eliminating the risk of heavy metal contamination in the final API and simplifying downstream purification processes significantly.
Q: What represents the primary cost driver in this catalytic system?
A: The primary cost efficiency comes from the use of inexpensive oxidants like potassium hydrogen persulfate and the ability to run reactions at ambient temperatures, which drastically reduces energy consumption compared to cryogenic metal-catalyzed processes.
Q: Is this technology scalable for industrial production?
A: Yes, the synthesis utilizes robust reaction conditions such as room temperature alkylation and standard solvent systems like acetonitrile and water, making the commercial scale-up of complex chiral intermediates highly feasible without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinaphthoazepinium Salts Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern pharmaceutical manufacturing and are committed to delivering high-purity dinaphthoazepinium salts that meet the rigorous demands of the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee the enantiomeric excess and chemical purity of every batch we produce. Our dedication to quality assurance means that you can rely on our materials to deliver consistent performance in your critical asymmetric epoxidation reactions, minimizing the risk of batch variability.
We invite you to engage with our technical procurement team to discuss how our customized solutions can drive value for your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our metal-free catalytic systems. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, allowing you to make data-driven decisions that optimize both your scientific outcomes and your bottom line. Let us be your partner in advancing the frontiers of asymmetric synthesis.
