Revolutionizing Imidazophenanthroline Production: A Solvent-Free Commercial Scale-Up Strategy
The chemical landscape for functional heterocyclic compounds is undergoing a significant transformation, driven by the urgent need for sustainable manufacturing practices that do not compromise on yield or purity. Patent CN102180877A introduces a groundbreaking methodology for the synthesis of imidazophenanthroline compounds, specifically addressing the limitations associated with traditional solution-phase chemistry. This technology leverages a solvent-free mechanochemical approach, utilizing the condensation of phenanthroline dione with various aldehydes and ammonium salts under grinding conditions. For R&D directors and procurement specialists alike, this represents a pivotal shift towards greener, more cost-effective production of high-value intermediates used in luminescent materials and potential pharmaceutical applications. The elimination of harsh acidic catalysts and bulk organic solvents not only streamlines the operational workflow but also significantly mitigates the environmental hazards typically associated with the production of these complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl imidazophenanthrolines has relied heavily on condensation reactions conducted in strong acidic media, such as concentrated sulfuric acid or glacial acetic acid. These conventional protocols, while effective in driving the reaction to completion, present substantial drawbacks for modern industrial manufacturing, particularly regarding safety and waste management. The use of large volumes of corrosive acids necessitates specialized corrosion-resistant equipment and rigorous safety protocols to protect personnel from chemical burns and inhalation hazards. Furthermore, the post-reaction workup involves complex neutralization steps that generate massive quantities of saline wastewater, creating a significant burden on effluent treatment facilities and increasing the overall E-factor of the process. The presence of residual acid can also complicate downstream purification, potentially leading to product degradation or the formation of unwanted byproducts that are difficult to separate, thereby impacting the final purity profile required for high-performance electronic or biological applications.
The Novel Approach
In stark contrast to the hazardous traditional routes, the novel solvent-free grinding method described in the patent data offers a streamlined, environmentally benign alternative that maintains high reaction efficiency. By mechanically mixing phenanthroline dione, aldehydes, and ammonium salts, the reactants are brought into intimate contact without the need for a liquid medium, facilitating the condensation reaction through mechanochemical activation. This approach operates at moderate temperatures, typically ranging from 60°C to 90°C, which significantly reduces energy consumption compared to high-temperature reflux conditions often required in solvent-based systems. The absence of solvent not only simplifies the reaction setup but also eliminates the need for solvent recovery systems, thereby reducing capital expenditure and operational complexity. This method demonstrates excellent selectivity and yield across a broad substrate scope, including various aromatic and aliphatic aldehydes, proving its robustness as a versatile platform for producing diverse imidazophenanthroline derivatives suitable for commercial scale-up.
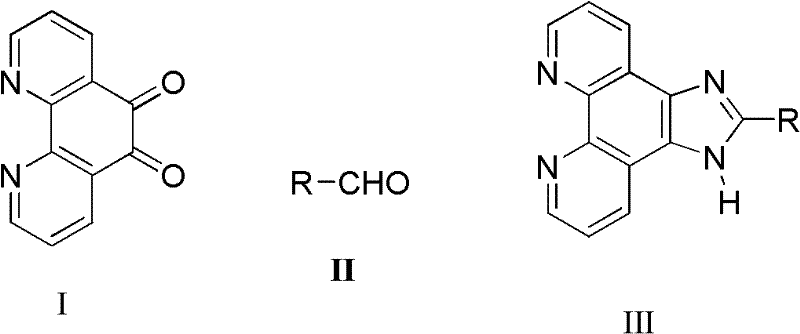
Mechanistic Insights into Solvent-Free Condensation Catalysis
The mechanistic underpinning of this solvent-free synthesis relies on the unique properties of solid-state reactivity, where the mechanical energy input from grinding serves to overcome the activation energy barrier for the condensation reaction. In the absence of a solvent shell, the reactant molecules are forced into close proximity, enhancing the frequency of effective collisions and promoting the nucleophilic attack of the amine species on the carbonyl carbon of the dione. The ammonium salt acts as both a nitrogen source and a mild catalyst, facilitating the dehydration step required to close the imidazole ring without the need for strong mineral acids. This solid-state environment often leads to different selectivity profiles compared to solution chemistry, potentially suppressing side reactions that are solvent-mediated, such as hydrolysis or polymerization. The result is a cleaner reaction profile with fewer impurities, which is critical for applications in organic light-emitting diodes (OLEDs) or as ligands in catalytic systems where trace metal or organic contaminants can quench performance.
Impurity control in this mechanochemical process is inherently superior due to the simplified reaction matrix and the absence of solvent-derived contaminants. In traditional acid-catalyzed routes, sulfonation or acetylation of the aromatic rings can occur as side reactions, introducing structural analogs that are challenging to remove via standard recrystallization. The neutral to mildly acidic conditions provided by the ammonium salt in the solid state minimize these electrophilic aromatic substitution side reactions, preserving the integrity of the sensitive phenanthroline backbone. Furthermore, the post-reaction workup involves a simple washing procedure with water and mild base adjustment, which effectively removes unreacted ammonium salts and polar byproducts without dissolving the target product. This high separation efficiency ensures that the crude product obtained after filtration is already of high purity, reducing the number of recrystallization cycles needed and minimizing product loss during purification, which directly translates to improved overall process mass intensity.
How to Synthesize Imidazophenanthroline Efficiently
To implement this synthesis route effectively, operators must focus on the precise control of stoichiometry and mechanical mixing parameters to ensure uniform reaction progression throughout the bulk material. The patent specifies a molar ratio of phenanthroline dione to aldehyde to ammonium salt, with a slight excess of aldehyde and a significant excess of ammonium salt recommended to drive the equilibrium towards the product. The grinding step is crucial; insufficient mixing can lead to hot spots or incomplete conversion, while excessive grinding might introduce mechanical impurities or cause localized overheating. Once the mixture is homogeneous, maintaining the temperature within the optimal window of 60°C to 90°C is essential to balance reaction rate with thermal stability, as monitored by thin-layer chromatography (TLC) to determine the precise endpoint. Detailed standardized synthesis steps follow below to guide the technical team in replicating these results.
- Mix phenanthroline dione, aldehyde, and ammonium salt in a reactor according to optimized molar ratios.
- Grind the mixture thoroughly to ensure homogeneity before initiating the thermal condensation reaction.
- Maintain reaction temperature between 60°C and 90°C, monitor via TLC, and purify the crude product through recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this solvent-free technology offers tangible strategic advantages that extend beyond mere technical feasibility into the realm of cost optimization and risk mitigation. The elimination of volatile organic solvents and corrosive acids fundamentally alters the cost structure of the manufacturing process by removing the expenses associated with solvent purchase, storage, recovery, and hazardous waste disposal. This reduction in material overhead directly contributes to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates. Additionally, the simplified operational workflow reduces the dependency on specialized infrastructure, such as acid-resistant reactors or explosion-proof solvent handling systems, thereby lowering capital investment requirements for new production lines and enhancing the agility of the supply chain to respond to market demand fluctuations.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the complete removal of bulk solvents and strong acid catalysts, which traditionally account for a significant portion of raw material and waste treatment costs. By operating under solvent-free conditions, the facility eliminates the need for expensive solvent recovery distillation columns and the associated energy consumption for heating and cooling large liquid volumes. Furthermore, the simplified workup procedure, which avoids complex neutralization and extraction steps, reduces labor hours and consumable usage, such as filter aids and drying agents. This lean manufacturing approach ensures that resources are focused solely on value-added transformation rather than waste management, resulting in substantial economic efficiency gains without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of readily available, commodity-grade raw materials such as ammonium acetate and common aldehydes, which are less susceptible to geopolitical supply disruptions compared to specialized catalysts or anhydrous solvents. The robustness of the solid-state reaction conditions means that production is less vulnerable to variations in raw material quality, as the grinding process can accommodate minor fluctuations in particle size or moisture content without catastrophic failure. This reliability ensures consistent delivery schedules for downstream customers, reducing the risk of production stoppages due to raw material shortages or quality rejections. Moreover, the reduced hazard profile of the process simplifies logistics and storage requirements, allowing for safer and more flexible transportation of inputs and outputs.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the inherent safety of solvent-free operations, which removes the risks of solvent fires, explosions, and toxic vapor emissions. This aligns perfectly with increasingly stringent global environmental regulations regarding VOC emissions and hazardous waste generation, future-proofing the manufacturing asset against regulatory tightening. The ability to operate at moderate temperatures also reduces the thermal load on plant utilities, making it easier to integrate into existing facilities without major retrofitting. Consequently, the process supports sustainable growth strategies, enabling the company to expand capacity to meet rising demand for high-purity intermediates while maintaining a minimal environmental footprint and adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solvent-free synthesis technology, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational benefits and feasibility for potential partners looking to integrate this method into their supply chains. Understanding these nuances is critical for making informed decisions about process adoption and long-term sourcing strategies.
Q: How does this solvent-free method improve environmental compliance compared to traditional acid-catalyzed routes?
A: Traditional methods often utilize large volumes of sulfuric acid or glacial acetic acid, generating significant hazardous waste streams. This novel solvent-free grinding technique eliminates the need for corrosive liquid acids, drastically reducing waste treatment costs and environmental footprint while simplifying regulatory compliance.
Q: What are the purity implications of eliminating solvent residues in the final product?
A: By avoiding bulk solvents, the risk of solvent entrapment within the crystal lattice is minimized. This leads to a cleaner crude profile, reducing the burden on downstream purification steps like recrystallization and ensuring higher final purity specifications suitable for sensitive electronic or pharmaceutical applications.
Q: Is this mechanochemical process scalable for industrial tonnage production?
A: Yes, the process relies on solid-state grinding and moderate thermal conditions (60-90°C), which are highly adaptable to industrial mixing and heating equipment. The absence of volatile organic compounds (VOCs) further enhances safety and scalability for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazophenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into robust, commercial-grade manufacturing processes that meet the rigorous demands of the global pharmaceutical and electronic materials industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity imidazophenanthroline compounds that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging our expertise in green chemistry and process optimization, we help our clients achieve their sustainability goals while securing a stable supply of high-performance chemical intermediates.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements and drive value across your organization. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic benefits of switching to this solvent-free methodology. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to validate the quality and compatibility of our materials with your downstream applications. Together, we can build a more sustainable and efficient supply chain for the next generation of functional materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
