Advanced Synthesis of High-Purity Sodium Picosulfate Intermediates for Commercial Scale-Up
Advanced Synthesis of High-Purity Sodium Picosulfate Intermediates for Commercial Scale-Up
The pharmaceutical landscape for gastrointestinal therapeutics demands increasingly rigorous standards for intermediate purity, particularly for prodrugs like sodium picosulfate which rely on precise metabolic activation. Patent CN112851574A introduces a transformative methodology for preparing high-purity sodium picosulfate and its critical intermediate, 4,4'-(2-pyridylmethylene)diphenol, addressing long-standing challenges in isomer control and process safety. This innovation shifts the paradigm from hazardous, multi-step halogenated routes to a streamlined, low-temperature homogeneous condensation process utilizing acetonitrile as a key solvent. By strictly controlling reaction thermodynamics between -15°C and 25°C, the method effectively suppresses the formation of structural isomers that compromise drug stability and patient safety. For R&D directors and procurement specialists, this represents a significant opportunity to enhance the quality profile of laxative formulations while simultaneously simplifying the manufacturing supply chain.
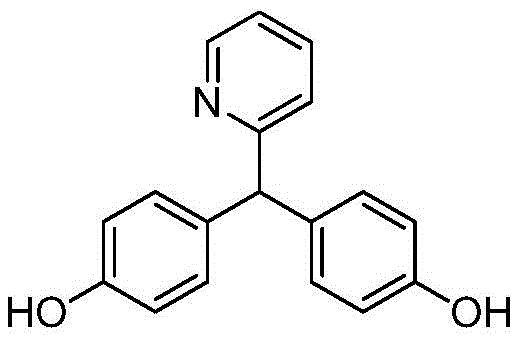
The core breakthrough lies in the ability to produce the intermediate with exceptional purity levels, often exceeding 99.8%, through a sophisticated crystallization refinement protocol. Traditional methods often struggle with the separation of ortho-substituted byproducts which possess similar physicochemical properties to the target para-substituted molecule. The disclosed process utilizes a specific sequence of alkali dissolution, pH adjustment, and alcohol-based dispersion crystallization to selectively precipitate the desired isomer. This level of control is not merely a technical achievement but a commercial imperative, as high-purity intermediates directly correlate to reduced degradation in gastric environments, thereby minimizing adverse gastrointestinal reactions such as nausea and abdominal distension in the final drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sodium picosulfate intermediates has been plagued by complex, hazardous, and inefficient pathways that pose significant risks to both operational safety and product quality. Early methodologies, such as those described in U.S. Patents 3528986 and 3558643, relied heavily on the use of halogenated starting materials like 2-chlorophenol or 2,6-dichlorophenol. These routes necessitated a subsequent reduction step using nickel-aluminum alloys in strong alkaline solutions to remove the halogen atoms, introducing severe safety hazards associated with hydrogen gas evolution and pyrophoric materials. Furthermore, the steric hindrance caused by ortho-halogen substituents often required elevated reaction temperatures during sulfation, leading to poor yields typically around 40% and complicating the purification process.
Alternative solvent-free or chloroform-based condensation methods, while avoiding halogens, introduced their own set of critical deficiencies regarding selectivity and environmental impact. In the absence of a polar coordinating solvent like acetonitrile, the reaction between phenol and 2-pyridinecarboxaldehyde tends to proceed heterogeneously, creating localized hot spots that promote the formation of difficult-to-remove isomer byproducts. These impurities are structurally similar to the target molecule, making them exceptionally challenging to separate via standard crystallization techniques. Additionally, some prior art attempts to mitigate these issues involved the addition of metal salts like cuprous chloride or ferrous ammonium sulfate, which inadvertently introduced new heavy metal impurities that require costly and time-consuming removal steps to meet stringent pharmacopoeial standards.
The Novel Approach
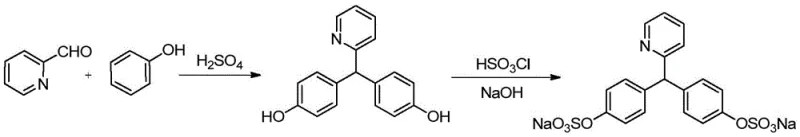
The novel approach detailed in the patent data fundamentally reengineers the synthesis by leveraging acetonitrile to create a stable, homogeneous reaction environment at controlled low temperatures. By maintaining the reaction mixture between -15°C and 25°C during the dropwise addition of concentrated sulfuric acid and 2-pyridylaldehyde, the process kinetically favors the formation of the desired 4,4'-isomer while suppressing ortho-substitution. This homogeneous system ensures uniform heat dissipation, preventing the thermal runaway that typically drives byproduct formation in solvent-free conditions. The result is a crude product with a significantly cleaner impurity profile, which drastically reduces the burden on downstream purification units and increases the overall throughput of the manufacturing line.
Furthermore, the purification strategy employs a clever sequence of pH-controlled crystallization and hot pulping with alcohols such as isopropanol or ethanol. Instead of relying on dangerous reduction steps or toxic metal complexing agents, the method utilizes simple acid-base chemistry and solubility differences to isolate the high-purity intermediate. The intermediate is first dissolved as a salt in an alkaline solution to remove insoluble inorganic impurities, then reprecipitated by adjusting the pH to a narrow range of 7.5 to 8.5. This gentle yet effective refinement ensures that the final intermediate meets the rigorous purity specifications required for pharmaceutical grade sodium picosulfate, setting a new benchmark for efficiency in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Catalyzed Condensation and Isomer Control
The mechanistic success of this process hinges on the precise modulation of electrophilic aromatic substitution dynamics within a polar aprotic solvent medium. In the presence of concentrated sulfuric acid, 2-pyridinecarboxaldehyde is activated to form a highly reactive electrophilic species, likely a carbocation or a protonated iminium ion equivalent. In traditional solvent-free systems, the high local concentration of reactants and lack of thermal buffering leads to uncontrolled attack at both the ortho and para positions of the phenol ring. However, the use of acetonitrile solvates the ionic intermediates and stabilizes the transition state, while the low temperature (-15°C to 5°C) provides the necessary thermodynamic bias towards the more stable para-substituted product. This kinetic control is essential for minimizing the generation of the 2,4'-isomer, which is the primary impurity of concern.
Following the condensation, the quenching and neutralization phases are critical for locking in the purity gains achieved during the reaction. The addition of ice water quenches the exothermic reaction instantly, preventing further rearrangement or degradation of the sensitive diphenol structure. Subsequent treatment with sodium hydroxide converts the phenolic hydroxyl groups into water-soluble phenolate salts, allowing for the filtration of insoluble tarry byproducts and inorganic salts. The careful readjustment of pH using hydrochloric acid in the presence of alcohol induces a controlled precipitation of the free phenol. This step acts as a secondary purification barrier, as the solubility of the target isomer differs subtly from its impurities in the alcohol-water mixture, enabling the isolation of a white solid with purity levels approaching 99.9% before the final sulfation step even begins.
How to Synthesize 4,4'-(2-pyridylmethylene)diphenol Efficiently
The synthesis protocol outlined in the patent offers a robust and scalable pathway for producing the critical sodium picosulfate intermediate with high consistency. The process begins with the preparation of a phenol-acetonitrile solution, which is cooled to sub-ambient temperatures to prepare for the exothermic addition of reagents. Concentrated sulfuric acid and 2-pyridylaldehyde are added sequentially under strict temperature control to ensure the reaction proceeds within the optimal kinetic window. Following the reaction period, the mixture is quenched, neutralized, and subjected to a multi-stage crystallization process involving isopropanol or ethanol to refine the crude material. The detailed standardized synthetic steps for this high-efficiency route are provided in the technical guide below.
- Condense phenol and 2-pyridinecarboxaldehyde in acetonitrile with concentrated sulfuric acid at -15 to 25°C.
- Quench the reaction, neutralize with alkali, and perform dispersion crystallization using organic alcohol to refine the crude intermediate.
- Sulfonate the refined intermediate with chlorosulfonic acid, convert to sodium salt, and purify via hot methanol extraction and ethyl acetate crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers profound strategic advantages centered around cost optimization and risk mitigation. By eliminating the need for halogenated raw materials and the subsequent hazardous reduction steps involving nickel-aluminum alloys, the process removes significant safety liabilities and regulatory burdens associated with heavy metal handling and disposal. The simplified workflow, which relies on commodity chemicals like phenol, acetonitrile, and sulfuric acid, ensures a more resilient supply chain that is less susceptible to the volatility of specialized reagent markets. This transition from a complex, multi-step hazardous process to a streamlined condensation-crystallization sequence translates directly into lower operational expenditures and enhanced production reliability.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reduction agents, such as nickel-aluminum alloys, coupled with the removal of heavy metal scavenging steps, results in substantial cost savings in raw material consumption and waste treatment. The high selectivity of the acetonitrile-based reaction minimizes the loss of valuable starting materials to byproduct formation, thereby improving the overall mass balance and yield efficiency of the plant. Furthermore, the use of common solvents like acetonitrile and isopropanol, which are easily recoverable and recyclable, reduces the recurring cost of solvent procurement and lowers the environmental compliance costs associated with volatile organic compound emissions.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as phenol and 2-pyridinecarboxaldehyde ensures a stable and continuous supply of raw materials, mitigating the risk of production stoppages due to specialty chemical shortages. The robustness of the low-temperature condensation process allows for consistent batch-to-batch quality, reducing the frequency of failed batches and the need for reprocessing. This predictability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by global pharmaceutical clients who require uninterrupted access to high-quality pharmaceutical intermediates.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction in acetonitrile facilitates excellent heat transfer and mixing, making the process highly amenable to scale-up from pilot plants to multi-ton commercial reactors without the hot-spot issues prevalent in solvent-free methods. The absence of halogenated waste streams and heavy metal residues simplifies the effluent treatment process, ensuring easier compliance with increasingly stringent environmental regulations. This green chemistry approach not only future-proofs the manufacturing facility against regulatory changes but also aligns with the sustainability goals of major multinational pharmaceutical corporations seeking eco-friendly suppliers.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial aspects of the sodium picosulfate synthesis process, derived directly from the patent specifications and comparative analysis of prior art. These insights are designed to clarify the operational benefits and quality assurances provided by the new methodology for stakeholders evaluating this technology for potential licensing or procurement partnerships. Understanding these nuances is key to appreciating the value proposition of this advanced manufacturing route.
Q: How does the new acetonitrile-based method improve purity compared to solvent-free processes?
A: The use of acetonitrile creates a homogeneous reaction system at low temperatures (-15 to 25°C), which significantly suppresses the formation of isomer byproducts that are common in heterogeneous, solvent-free conditions. This kinetic control allows for easier removal of impurities during the subsequent crystallization steps.
Q: Why is the removal of isomer impurities critical for sodium picosulfate safety?
A: Sodium picosulfate is a prodrug that releases active diphenols in the colon. Impurities or premature release due to instability can cause severe gastrointestinal irritation, nausea, and abdominal pain. High purity ensures the drug remains stable in the stomach and only activates in the large intestine.
Q: Does this process eliminate the need for hazardous hydrogenation steps?
A: Yes, unlike prior art methods that utilize halogenated phenols requiring dangerous nickel-aluminum alloy reduction, this novel route uses direct condensation of phenol, completely eliminating the need for high-risk hydrogenation and heavy metal removal steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Picosulfate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to high-purity synthesis routes requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the precise low-temperature conditions and specialized crystallization protocols required for this process, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch of high-purity sodium picosulfate intermediate we produce.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their laxative portfolios. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this safer, more efficient route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and discover how we can support your goal of delivering safer, higher-quality gastrointestinal therapeutics to the global market.
