Advanced Manufacturing of Key Antiviral Intermediates for Paxlovid and Boceprevir Supply Chains
Advanced Manufacturing of Key Antiviral Intermediates for Paxlovid and Boceprevir Supply Chains
The global pharmaceutical landscape has been profoundly reshaped by the urgent demand for effective antiviral therapeutics, particularly following the emergence of SARS-CoV-2. At the heart of this medical response lies the critical need for reliable supply chains of complex small-molecule intermediates. Patent CN115322136A discloses a groundbreaking methodology for the preparation of (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate hydrochloride, a pivotal building block known as INTA. This compound serves as the structural core for both Paxlovid (PF-07321332) and Boceprevir, two potent protease inhibitors that have demonstrated significant efficacy in treating viral infections. The structural complexity of these drugs, featuring rigid bicyclic frameworks, presents substantial synthetic challenges that often hinder rapid scale-up and cost-effective manufacturing.
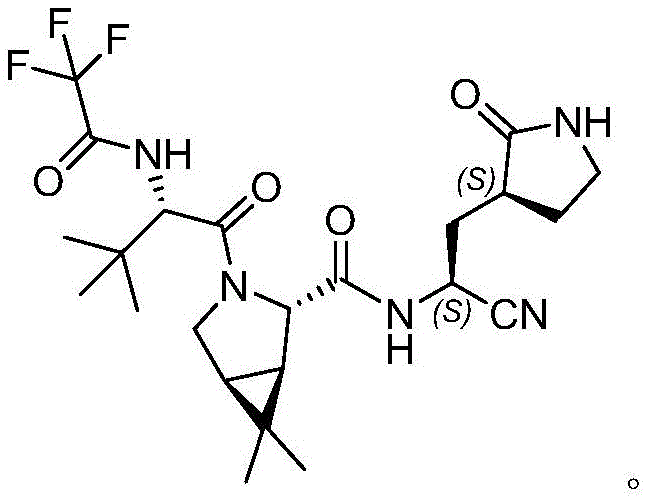
Traditional synthetic routes to access this specific azabicyclo scaffold have historically been plagued by inefficiencies, including multi-step sequences, harsh reaction conditions, and the utilization of hazardous reagents such as potassium cyanide. These legacy methods not only pose severe environmental and safety risks but also introduce variability in chiral purity, which is paramount for the biological activity of the final API. The innovation presented in this patent addresses these systemic issues by introducing a streamlined, three-step protocol that leverages advanced organometallic catalysis. By shifting the paradigm towards a more sustainable and operationally simple process, this technology offers a viable pathway for reliable pharmaceutical intermediate supplier networks to meet the escalating global demand for antiviral medications without compromising on quality or safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the 3-azabicyclo[3.1.0]hexane core has relied on methodologies that are inherently difficult to manage on an industrial scale. Conventional approaches often necessitate the use of highly toxic cyanide sources for ring closure, which imposes rigorous safety protocols and expensive waste treatment requirements that drastically inflate production costs. Furthermore, existing literature describes routes that involve numerous protection and deprotection steps, leading to cumulative yield losses and extended cycle times. The reliance on precious metal catalysts or stoichiometric amounts of expensive reagents further exacerbates the economic burden, making the final intermediate prohibitively expensive for widespread generic production. Additionally, controlling stereochemistry in these older methods is frequently problematic, often requiring tedious chiral chromatography to achieve the necessary optical purity, which is a significant bottleneck in cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
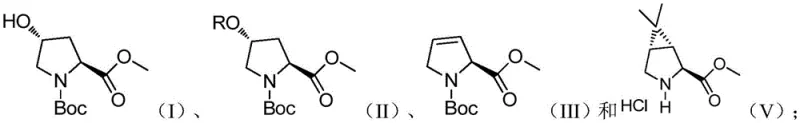
In stark contrast, the method disclosed in patent CN115322136A introduces a highly efficient strategy that bypasses these traditional pitfalls. The new route begins with a straightforward hydroxyl protection of N-Boc-trans-4-hydroxy-L-proline methyl ester, followed by a base-mediated elimination to generate a key alkene intermediate. The crown jewel of this synthesis is the subsequent cyclopropanation step, which utilizes a specialized cobalt catalyst system to construct the bicyclic framework with exceptional precision. This approach operates under mild thermal conditions, typically between 20°C and 50°C, eliminating the need for energy-intensive heating or cryogenic cooling. The result is a robust process that delivers the target compound with yields exceeding 66% and optical purity (ee) greater than 95%, directly addressing the critical quality attributes required for API synthesis.
Mechanistic Insights into Cobalt-Catalyzed Cyclopropanation
The success of this novel synthetic route hinges on the sophisticated application of a cobalt-based catalytic system, specifically [2-t-Bu PDI]CoBr2. This catalyst is not merely a reagent but a finely tuned molecular machine designed to facilitate the transfer of a carbene species to the alkene substrate with high stereoselectivity. The preparation of this catalyst involves the condensation of diacetylpyridine with 2,6-diisopropylaniline to form a tridentate ligand, which is then complexed with cobalt bromide. This specific ligand architecture creates a steric environment that favors the formation of the desired (1R,2S,5S) stereoisomer while suppressing the formation of unwanted diastereomers. The mechanism likely proceeds through the generation of a cobalt-carbene intermediate upon reaction with the dihaloalkane (e.g., 2,2-dichloropropane), which then undergoes a concerted addition to the double bond of the pyrroline intermediate.
![Synthesis of the Cobalt Catalyst [2-t-Bu PDI]CoBr2 from SM1 and SM2](/insights/img/paxlovid-intermediate-synthesis-cobalt-catalysis-supplier-20260307113824-07.webp)
Crucially, the inclusion of metal activators such as zinc powder and additives like zinc bromide plays a vital role in maintaining the active oxidation state of the cobalt center and enhancing the turnover number. This synergistic effect ensures that the reaction proceeds to completion with minimal catalyst loading, often as low as 0.001 to 0.1 mol%. From an impurity control perspective, this catalytic system is remarkably clean, producing very few side products compared to radical-based non-catalytic methods. The high chiral fidelity observed (ee >95%) suggests that the chiral information from the starting proline derivative is effectively transmitted and locked in during the cyclopropanation event, thereby minimizing the need for downstream purification and ensuring a consistent impurity profile suitable for regulatory submission.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylate Efficiently
The practical implementation of this synthesis is designed for scalability, utilizing common organic solvents and standard reactor configurations. The process initiates with the activation of the hydroxyl group, followed by elimination to set up the olefin for the final ring-closing step. Each stage has been optimized to balance reaction kinetics with ease of workup, ensuring that the intermediate can be isolated with high purity before proceeding to the next transformation. This modular approach allows for flexibility in manufacturing, enabling producers to stock stable intermediates if necessary. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the standardized guide below which outlines the critical process controls.
- Perform hydroxyl protection on N-Boc-trans-4-hydroxy-L-proline methyl ester using methanesulfonyl chloride and a base like triethylamine to form the mesylate intermediate.
- Conduct an elimination reaction on the protected intermediate using sodium methoxide followed by methylation to generate the alkene precursor.
- Execute the key cyclopropanation reaction using a specialized cobalt catalyst ([2-t-Bu PDI]CoBr2), zinc powder, and 2,2-dichloropropane to form the final bicyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to optimize the sourcing of critical antiviral ingredients. The shift away from hazardous cyanide chemistry not only aligns with increasingly stringent environmental regulations but also significantly lowers the barrier to entry for contract manufacturing organizations (CMOs). By simplifying the synthetic route, the overall production timeline is compressed, allowing for faster response to market fluctuations and emergency health demands. The use of earth-abundant cobalt instead of precious metals like palladium or rhodium further insulates the supply chain from volatile commodity pricing, ensuring more predictable budgeting and cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and toxic cyanide reagents leads to substantial savings in raw material costs. Furthermore, the high yield of the cyclopropanation step minimizes material loss, meaning less starting material is required to produce the same amount of final product. The simplified workup procedures reduce solvent consumption and waste disposal fees, contributing to a leaner and more economically efficient production model that enhances overall margin potential.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as N-Boc-trans-4-hydroxy-L-proline methyl ester, are commercially available in bulk quantities from multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions (20-50°C) mean that the process can be run in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment, thereby increasing the pool of qualified manufacturing partners capable of executing the synthesis reliably.
- Scalability and Environmental Compliance: This process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The avoidance of genotoxic reagents simplifies the regulatory clearance process and reduces the burden on environmental health and safety (EHS) teams. The robust nature of the cobalt catalyst system ensures consistent performance from kilogram to tonne scale, providing the stability needed for long-term supply agreements and ensuring continuity of care for patients relying on these life-saving medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their supply chains.
Q: What are the advantages of this new synthesis method over traditional routes?
A: This method eliminates the use of highly toxic potassium cyanide and expensive transition metal catalysts found in conventional routes. It offers a shorter synthetic sequence with milder reaction conditions (20-50°C), resulting in higher overall yields (>66%) and superior chiral purity (ee >95%).
Q: Is the cobalt catalyst used in this process commercially viable for large-scale production?
A: Yes, the catalyst [2-t-Bu PDI]CoBr2 is synthesized from readily available starting materials (diacetylpyridine and 2,6-diisopropylaniline) in high yield. The low loading required (0.001-0.1 mol%) and the ability to recycle or minimize metal waste make it highly suitable for industrial scale-up.
Q: How does this process impact the supply chain stability for Paxlovid manufacturers?
A: By utilizing common solvents like dichloromethane and THF, and avoiding specialized high-pressure equipment or cryogenic conditions, this process reduces lead times and operational risks. The robustness of the reaction ensures consistent supply of the critical INTA intermediate, mitigating bottlenecks in antiviral drug manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable INTA Supplier
As the demand for antiviral therapeutics continues to evolve, securing a stable supply of high-quality intermediates like INTA is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver solutions that bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We operate under stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for GMP API synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced cobalt-catalyzed route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer picture of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the rapid and efficient manufacturing of next-generation antiviral intermediates.
