Scalable Albendazole Manufacturing via Zinc Chloride Complexation and Hydrazine Reduction
Scalable Albendazole Manufacturing via Zinc Chloride Complexation and Hydrazine Reduction
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high yield with environmental sustainability, particularly for broad-spectrum antiparasitic agents like albendazole. Patent CN101270091A introduces a transformative preparation method that addresses critical bottlenecks in traditional manufacturing, specifically targeting the purification of key intermediates and the elimination of hazardous reagents. This technical insight report analyzes the novel integration of polyethylene glycol (PEG) phase transfer catalysis and zinc chloride complexation separation, offering a viable alternative to the historically dominant but environmentally taxing o-nitroaniline routes. By replacing toxic sodium cyanide and sodium sulfide with safer alkaline and hydrazine-based systems, this methodology not only enhances operator safety but also significantly streamlines the downstream purification processes required for high-purity active pharmaceutical ingredients (APIs).
For R&D directors and process chemists, the strategic value of this patent lies in its ability to produce intermediates with purity levels exceeding 99% without relying on energy-intensive ultra-low temperature crystallization. The implementation of a zinc chloride clathrate separation technique allows for the isolation of 2-nitro-4-propylthioaniline directly from the reaction mixture, bypassing the complex distillation or freezing steps often required in conventional syntheses. Furthermore, the substitution of sodium sulfide with hydrazine hydrate in the reduction step fundamentally alters the waste profile of the facility, removing the burden of treating large volumes of sulfur-laden effluent. This report serves as a comprehensive guide for procurement and supply chain leaders evaluating reliable albendazole supplier options that prioritize both cost efficiency and regulatory compliance in veterinary and human pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of albendazole has relied heavily on routes derived from U.S. Patent US 4,152,522, which utilizes o-nitroaniline as the starting material. While this pathway offers reasonable yields, it is plagued by severe safety and operational deficiencies that hinder modern scalable production. A primary concern is the utilization of sodium cyanide during the thiocyanation and subsequent alkylation steps, introducing extreme toxicity risks that require specialized containment infrastructure and rigorous waste management protocols. Additionally, the isolation of the critical intermediate, 2-nitro-4-propylthioaniline, traditionally necessitates cooling the reaction mixture to ultra-low temperatures ranging from -50°C to -70°C to induce crystallization. This requirement imposes a massive energy penalty on the manufacturing process and limits the batch size due to the cooling capacity of standard industrial reactors, creating a significant bottleneck for cost reduction in pharmaceutical intermediates manufacturing.
Furthermore, the reduction of the nitro group to the corresponding diamine in conventional processes typically employs sodium sulfide, which generates substantial quantities of sulfur-containing wastewater. This effluent is not only environmentally hazardous but also costly to treat to meet increasingly stringent global discharge standards. The reliance on methyl cyanocarbamate for the final cyclization step further exacerbates the water usage issue, as this reagent often contains 80% to 90% water by weight, leading to dilute reaction conditions and difficult product isolation. Consequently, the crude albendazole produced via these legacy methods often exhibits deep coloration and high levels of individual impurities, necessitating multiple recrystallization steps that erode overall yield and extend production lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN101270091A presents a sophisticated solution to these entrenched problems by re-engineering the reaction conditions and separation techniques. Instead of relying on toxic cyanides, the new process employs pure sodium hydroxide or potassium hydroxide solutions in conjunction with polyethylene glycol (PEG 200-20000) as a phase transfer catalyst. This modification accelerates the condensation reaction between 2-nitro-4-thiocyanatoaniline and bromopropane, allowing it to proceed efficiently at moderate temperatures between 50°C and 120°C. More critically, the introduction of a zinc chloride complexation strategy revolutionizes the purification of the intermediate. By adding a zinc chloride solution to the organic layer, the target compound forms a solid complex that can be easily filtered off, effectively separating it from inorganic salts and unreacted starting materials without the need for energy-intensive cryogenic cooling.
In the reduction phase, the process substitutes sodium sulfide with hydrazine hydrate, catalyzed by ferric chloride and activated carbon. This shift eliminates the generation of sulfur waste entirely and achieves reduction yields exceeding 90% with product purity surpassing 99%. The final cyclization utilizes O-methyl isourea formate in an organic solvent system, which avoids the excessive water introduction associated with methyl cyanocarbamate. This allows for the recovery of organic solvents via distillation, significantly reducing the volume of acidic wastewater generated. Collectively, these innovations create a cleaner, safer, and more economically viable pathway for the commercial scale-up of complex pharmaceutical additives, ensuring a consistent supply of high-quality albendazole suitable for both veterinary and human therapeutic applications.
Mechanistic Insights into ZnCl2 Complexation and FeCl3-Catalyzed Reduction
The core innovation of this synthesis route lies in the selective separation mechanism facilitated by zinc chloride. In the condensation step, the reaction mixture contains the desired 2-nitro-4-propylthioaniline alongside various organic byproducts and inorganic salts such as sodium bromide. Traditional physical separation methods struggle to isolate the product due to similar solubility profiles. However, the addition of a 20% to 80% weight concentration zinc chloride solution induces the formation of a specific coordination complex between the zinc ion and the nitrogen/sulfur atoms of the aniline derivative. This 2-nitro-4-propylthioaniline zinc chloride solid complex precipitates out of the solution, allowing for simple filtration. Subsequent treatment with an alkaline solution destroys the complex, releasing the pure free base of the intermediate while keeping the zinc in the aqueous phase. This chemical separation technique ensures that the intermediate entering the reduction step is of exceptionally high purity, which is crucial for minimizing side reactions in downstream processing.
Following isolation, the nitro group reduction is achieved through a catalytic transfer hydrogenation mechanism using hydrazine hydrate. The presence of ferric chloride (FeCl3) and activated carbon creates a highly active catalytic surface that facilitates the decomposition of hydrazine into reactive nitrogen species and hydrogen equivalents. These species selectively reduce the nitro group (-NO2) to the amino group (-NH2) without affecting the sensitive propylthio ether linkage, a common failure point in less selective reduction methods. The use of activated carbon not only acts as a support for the iron catalyst but also adsorbs colored impurities, contributing to the pale color of the final product. This mechanistic precision ensures that the resulting 4-propylthio-o-phenylenediamine maintains a purity profile that supports the production of albendazole meeting strict pharmacopoeial standards.
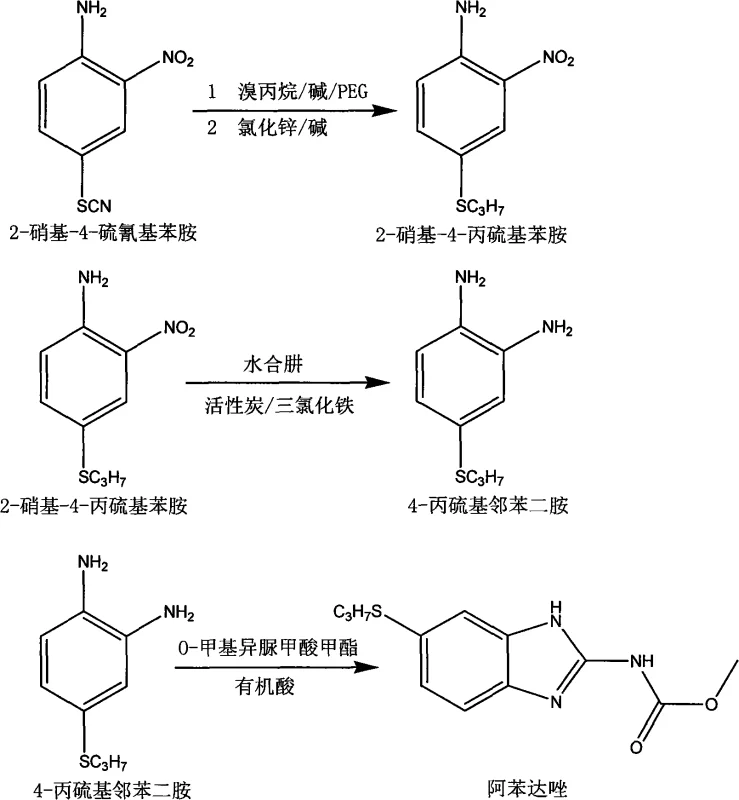
The final cyclization step involves the reaction of the diamine with O-methyl isourea formate in the presence of an organic acid such as acetic or formic acid. This reaction proceeds through a nucleophilic attack of the amine nitrogen on the imino carbon of the isourea, followed by intramolecular cyclization to form the benzimidazole ring. The choice of O-methyl isourea formate over methyl cyanocarbamate is significant; it allows the reaction to occur in a homogeneous organic phase, typically using toluene as a solvent. This homogeneity improves heat transfer and reaction kinetics, while the absence of bulk water prevents hydrolysis of the reagent and simplifies the workup. Upon completion, the addition of deionized water induces crystallization of the crude albendazole, which can then be further purified by slurry washing with solvents like methanol or acetone to remove residual impurities, yielding a final product with high optical clarity and minimal impurity content.
How to Synthesize Albendazole Efficiently
The synthesis of albendazole via this patented route requires precise control over reaction parameters to maximize yield and purity. The process is divided into three distinct stages: condensation with complexation separation, catalytic reduction, and ring-closing cyclization. Each stage demands specific stoichiometric ratios, temperature controls, and inert atmosphere conditions to ensure safety and reproducibility. For example, the condensation reaction must be monitored via HPLC to ensure complete consumption of the thiocyanato precursor before proceeding to the zinc chloride addition. Similarly, the reduction step requires careful addition of hydrazine to prevent exothermic runaway, while the final cyclization benefits from controlled acid catalysis to drive the equilibrium towards the benzimidazole product. Detailed operational procedures, including specific solvent volumes and drying conditions, are essential for translating this laboratory-scale success to industrial production.
- Condense 2-nitro-4-thiocyanatoaniline with bromopropane using PEG catalyst, then separate via zinc chloride complexation.
- Reduce the nitro group to an amine using hydrazine hydrate catalyzed by ferric chloride and activated carbon.
- Cyclize the diamine with O-methyl isourea formate in the presence of organic acid to form the benzimidazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthesis methodology offers substantial strategic advantages over legacy manufacturing routes. The elimination of sodium cyanide removes the need for expensive hazardous material handling certifications and reduces insurance liabilities associated with toxic reagent storage. Furthermore, the avoidance of ultra-low temperature crystallization equipment (-50°C to -70°C) significantly lowers capital expenditure (CAPEX) for new production lines and reduces operational expenditure (OPEX) related to energy consumption for refrigeration. This simplification of the process infrastructure enhances the reliability of the supply chain, as the manufacturing process becomes less dependent on specialized, high-maintenance cooling systems that are prone to failure. Consequently, partners can expect more consistent delivery schedules and reduced risk of production stoppages due to equipment limitations.
Cost reduction in pharmaceutical intermediates manufacturing is further driven by the improved efficiency of raw material utilization and waste management. The zinc chloride complexation method allows for the recovery of sodium bromide from the mother liquor, turning a waste product into a recoverable asset that can offset raw material costs. Additionally, the replacement of sodium sulfide with hydrazine hydrate drastically reduces the volume of wastewater requiring treatment, lowering environmental compliance costs. The high purity of the intermediates generated at each step minimizes the need for extensive downstream purification, thereby increasing the overall throughput of the facility. These factors combine to create a leaner manufacturing process that is inherently more cost-effective, allowing suppliers to offer competitive pricing without compromising on the quality specifications required by global regulatory bodies.
Scalability and environmental compliance are also significantly enhanced by this green chemistry approach. The process operates at moderate temperatures and pressures, making it easier to scale from pilot batches to multi-ton commercial production without encountering the heat transfer issues common in cryogenic processes. The reduction in hazardous waste generation aligns with global sustainability goals, making the supply chain more resilient to tightening environmental regulations. By adopting a process that inherently produces less pollution, manufacturers future-proof their operations against potential regulatory bans on sulfur-heavy or cyanide-based chemistries. This proactive approach to environmental stewardship ensures long-term supply continuity for customers who prioritize sustainable sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced albendazole synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in patent CN101270091A, providing clarity on how this method compares to traditional industrial practices. Understanding these distinctions is vital for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains or for R&D teams assessing the potential for process optimization. The focus remains on the practical implications of the zinc chloride separation and hydrazine reduction techniques on product quality and operational efficiency.
Q: How does the zinc chloride complexation method improve purity?
A: The zinc chloride complexation method allows for the direct separation of 2-nitro-4-propylthioaniline from the reaction mixture as a solid complex, avoiding the need for ultra-low temperature crystallization and significantly reducing organic impurities.
Q: Why is hydrazine hydrate preferred over sodium sulfide for reduction?
A: Hydrazine hydrate reduction eliminates the generation of large volumes of sulfur-containing wastewater associated with sodium sulfide, resulting in a cleaner process and higher product purity exceeding 99%.
Q: What are the environmental benefits of this synthesis route?
A: This route replaces toxic sodium cyanide and sodium sulfide with safer reagents like sodium hydroxide and hydrazine, and enables the recovery of sodium bromide byproducts, drastically reducing hazardous waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Albendazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to cleaner, more efficient synthetic routes is critical for the long-term viability of the pharmaceutical supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN101270091A can be successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific requirements of phase transfer catalysis and complexation separations, guaranteeing stringent purity specifications for every batch. With rigorous QC labs and a commitment to continuous improvement, we are positioned to deliver high-purity albendazole that meets the demanding standards of international pharmacopoeias.
We invite procurement leaders and technical directors to engage with our team to explore how this optimized synthesis route can benefit your specific product portfolio. By leveraging our expertise in process development, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating the tangible economic benefits of switching to this greener methodology. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your supply of this critical antiparasitic agent remains secure, compliant, and cost-effective for years to come.
