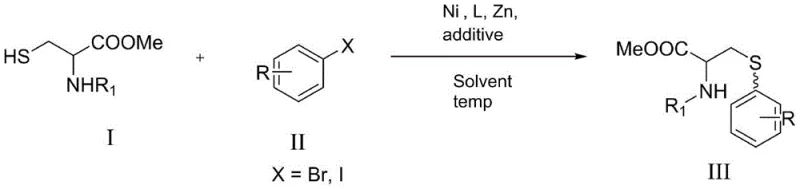
Advanced Nickel-Catalyzed Synthesis of Cysteine Derivatives for Commercial Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient and cost-effective pathways for synthesizing essential amino acid derivatives, particularly cysteine derivatives which hold significant potential in drug development. Patent CN112028800B introduces a groundbreaking synthetic methodology that utilizes a nickel-catalyzed cross-coupling reaction to produce these valuable compounds under remarkably mild conditions. This technical innovation addresses the critical need for sustainable and economically viable manufacturing processes by replacing expensive noble metal catalysts with abundant nickel sources. The method employs cysteine esters and aryl bromides as primary substrates, reacting them in the presence of a specific ligand system and reducing agents to achieve high yields without the need for rigorous inert gas protection. This development represents a significant leap forward for any reliable pharmaceutical intermediates supplier aiming to optimize their production capabilities and reduce dependency on volatile precious metal markets. By leveraging this patented technology, manufacturers can ensure a more stable supply chain while maintaining the stringent quality standards required for active pharmaceutical ingredients and their precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cysteine derivatives has relied heavily on palladium-catalyzed coupling reactions, which present substantial economic and operational challenges for large-scale manufacturing. The primary drawback of using palladium is its exorbitant cost, which significantly inflates the overall production expenses and makes the final product less competitive in the global market. Furthermore, conventional methods often involve the use of mercaptans as starting materials, which are notorious for their extremely unpleasant odors and handling difficulties, posing safety and environmental hazards in a production facility. Alternative routes utilizing amino alcohols are also problematic due to the high cost of these raw materials compared to cysteine, creating a bottleneck for cost reduction in pharmaceutical intermediates manufacturing. These traditional processes frequently require complex reaction conditions, including strict anhydrous environments and elevated temperatures, which increase energy consumption and operational complexity. Consequently, the industry has long suffered from a lack of scalable, safe, and economically efficient methods for producing high-purity cysteine derivatives, necessitating a shift towards more innovative catalytic systems.
The Novel Approach
The novel approach detailed in the patent data utilizes a nickel-catalyzed system that effectively overcomes the economic and operational barriers associated with traditional palladium chemistry. By employing nickel complexes such as Ni(acac)2, the method drastically lowers the catalyst cost while maintaining high catalytic efficiency and selectivity for the desired coupling transformation. The reaction proceeds smoothly at room temperature under an air atmosphere, eliminating the need for expensive inert gas setups and reducing the energy footprint of the manufacturing process. This one-step reaction strategy simplifies the operational workflow, minimizing the risk of human error and enhancing the overall safety profile of the synthesis. The use of readily available aryl bromides and protected cysteine esters ensures that raw material sourcing is both reliable and cost-effective, supporting the commercial scale-up of complex pharmaceutical intermediates. This method not only improves the economic viability of the process but also aligns with green chemistry principles by reducing waste and energy usage, making it an ideal choice for modern chemical production facilities.
Mechanistic Insights into Ni-Catalyzed Cross-Coupling
The core of this synthetic breakthrough lies in the intricate catalytic cycle facilitated by the nickel complex in conjunction with a nitrogen-containing ligand and zinc powder as a reductant. The reaction initiates with the oxidative addition of the aryl bromide to the low-valent nickel species, forming an organonickel intermediate that is crucial for the subsequent bond formation. The presence of zinc powder serves to regenerate the active nickel catalyst from higher oxidation states, ensuring the continuity of the catalytic cycle without the accumulation of inactive species. Anhydrous magnesium chloride acts as a critical additive, likely coordinating with the sulfur atom of the cysteine ester to enhance its nucleophilicity and facilitate the transmetallation step. This synergistic interaction between the catalyst, ligand, reductant, and additive allows the reaction to proceed with high turnover numbers and excellent functional group tolerance. The mechanism avoids the formation of common side products often seen in palladium chemistry, resulting in a cleaner reaction profile and simplified downstream purification processes for the final cysteine derivatives.
Controlling impurity profiles is paramount in the synthesis of pharmaceutical intermediates, and this nickel-catalyzed method offers superior selectivity compared to conventional routes. The mild reaction conditions prevent the decomposition of sensitive functional groups present on the aryl bromide or the cysteine ester, such as esters, aldehydes, and nitriles, which might otherwise degrade under harsher conditions. The specific choice of ligand, such as 4,4'-dimethoxy-2,2'-bipyridine, stabilizes the nickel center and prevents the formation of nickel black or other inactive aggregates that could lead to product contamination. By operating under air atmosphere, the system demonstrates robustness against oxygen and moisture, which are common sources of variability and impurity generation in sensitive organometallic reactions. This high level of control ensures that the resulting high-purity cysteine derivatives meet the rigorous specifications required for downstream drug synthesis, reducing the burden on quality control laboratories. The ability to maintain such high purity without extensive purification steps is a key advantage for any reliable pharmaceutical intermediates supplier looking to streamline their production.
How to Synthesize Cysteine Derivatives Efficiently
To implement this synthesis effectively, operators must adhere to the specific molar ratios and reaction conditions outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of a reaction solution containing the aryl bromide and N-protected cysteine ester dissolved in anhydrous dimethyl sulfoxide (DMSO) as the solvent. The catalyst system is then introduced, consisting of Ni(acac)2, the bipyridine ligand, zinc powder, and magnesium chloride, all added in precise stoichiometric amounts to drive the reaction to completion. The mixture is stirred at room temperature for a period ranging from 6 to 12 hours, allowing the coupling to proceed fully without the need for external heating or cooling. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining aryl bromide and N-protected cysteine ester in anhydrous DMSO solvent under an air atmosphere.
- Add the nickel catalyst Ni(acac)2, nitrogen-containing ligand, zinc powder reductant, and anhydrous magnesium chloride additive in specific molar ratios.
- Stir the solution at room temperature for 6 to 12 hours, then isolate the product via column chromatography without complex post-treatment.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this nickel-catalyzed methodology offers transformative benefits that directly impact the bottom line and operational reliability of chemical manufacturing. The shift from palladium to nickel represents a substantial cost saving opportunity, as the price volatility of precious metals is a major risk factor in long-term supply contracts. By utilizing base metal catalysts, manufacturers can stabilize their raw material costs and offer more competitive pricing to their clients without compromising on quality or performance. The simplicity of the reaction conditions, particularly the ability to run the process under air at room temperature, reduces the capital expenditure required for specialized reactor equipment and inert gas infrastructure. This accessibility allows for faster deployment of production lines and greater flexibility in responding to market demand fluctuations for high-purity cysteine derivatives. Furthermore, the high yields reported in the patent examples indicate a highly efficient use of raw materials, minimizing waste disposal costs and maximizing the output per batch.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with affordable nickel complexes leads to a significant decrease in the direct material cost of the synthesis. This cost optimization is further amplified by the elimination of complex post-treatment steps, as the reaction mixture can often be purified directly via column chromatography without extensive workup procedures. The use of inexpensive zinc powder as a reductant and common solvents like DMSO also contributes to the overall economic efficiency of the process. These factors combined allow for a drastic simplification of the cost structure, making the production of cysteine derivatives more financially sustainable in a competitive market environment.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as aryl bromides and cysteine esters ensures a stable supply chain that is less susceptible to geopolitical or logistical disruptions. Unlike specialized reagents that may have long lead times, the components for this reaction are commodity chemicals that can be sourced from multiple vendors globally. The robustness of the reaction under air atmosphere also reduces the risk of batch failures due to equipment leaks or inert gas supply issues, enhancing the consistency of production schedules. This reliability is crucial for reducing lead time for high-purity cysteine derivatives, ensuring that downstream pharmaceutical manufacturers receive their materials on time and without quality deviations.
- Scalability and Environmental Compliance: The mild conditions and simple operation of this method make it highly amenable to scale-up from laboratory to industrial production scales without significant re-engineering. The absence of toxic mercaptans and the use of less hazardous nickel salts improve the environmental profile of the process, facilitating compliance with increasingly strict environmental regulations. Reduced energy consumption due to room temperature operation lowers the carbon footprint of the manufacturing process, aligning with corporate sustainability goals. This scalability ensures that the method can meet growing market demand for complex pharmaceutical intermediates while maintaining safety and environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for R&D and procurement teams evaluating this technology for adoption in their own manufacturing workflows. The information provided here clarifies the operational advantages and quality outcomes associated with the nickel-catalyzed route.
Q: Why is nickel preferred over palladium for this cysteine derivative synthesis?
A: Nickel catalysts are significantly cheaper and more abundant than palladium, reducing raw material costs for mass production while maintaining high catalytic activity under mild conditions.
Q: What are the purity specifications for the synthesized cysteine derivatives?
A: The method yields high-purity products, often exceeding 90% yield with minimal byproducts, suitable for stringent pharmaceutical intermediate standards after standard chromatographic purification.
Q: Is this synthesis method scalable for industrial manufacturing?
A: Yes, the reaction operates at room temperature under air atmosphere with simple operation steps, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cysteine Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic technologies to deliver superior chemical solutions to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this nickel-catalyzed coupling are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of cysteine derivatives meets the highest industry standards. Our commitment to technical excellence allows us to offer high-purity cysteine derivatives that are ready for immediate integration into complex drug synthesis pipelines. By partnering with us, clients gain access to a supply chain that is both technologically advanced and commercially reliable.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to their specific production needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of switching to this optimized synthesis method. Contact us today to discuss how we can support your supply chain with high-quality intermediates and drive efficiency in your manufacturing operations. Let us help you engineer the next breakthrough in your pharmaceutical development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
