Advanced Aqueous Base Reduction for High-Purity Aniline Compounds and Commercial Scale-Up
Advanced Aqueous Base Reduction for High-Purity Aniline Compounds and Commercial Scale-Up
The chemical industry constantly seeks more efficient and safer pathways for synthesizing critical intermediates, and patent CN1429196A presents a significant breakthrough in the preparation of aniline compounds. This specific technology details a novel process where nitro compounds are reduced to their corresponding aniline derivatives using hydrazine at elevated temperatures, but crucially, in the presence of an aqueous base rather than traditional alcoholic solvents. This shift in solvent system represents a fundamental improvement in process chemistry, addressing long-standing challenges related to safety, selectivity, and industrial scalability. For R&D directors and technical leaders, understanding the nuances of this aqueous-based reduction is vital, as it offers a robust alternative to conventional methods that often rely on volatile organic compounds. The compounds produced through this method, specifically those of formula I as depicted in the patent, serve as essential intermediates in the preparation of isobenzofuranone type herbicides, highlighting their strategic importance in the agrochemical sector. By leveraging this patented approach, manufacturers can achieve superior control over reaction parameters, leading to consistent quality and reduced operational risks in large-scale production environments.
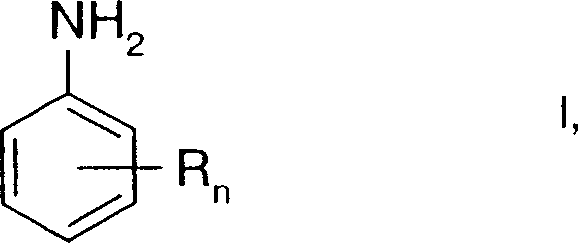
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitrobenzene to aniline compounds has been performed using hydrazine hydrate in alcoholic solutions, a method documented in literature dating back to the late 19th century. These conventional processes typically require elevated temperatures and often involve the use of ethanol or other organic solvents which introduce significant safety hazards due to flammability and volatility. Furthermore, reactions conducted in alcoholic media can sometimes suffer from slower reaction kinetics and lower selectivity, leading to the formation of unwanted by-products that complicate downstream purification. The reliance on organic solvents also necessitates extensive recovery and recycling systems, adding to the capital expenditure and operational complexity of the manufacturing plant. From a safety perspective, handling large volumes of flammable solvents at high temperatures increases the risk of fire and explosion, requiring stringent safety protocols and specialized equipment. Additionally, the environmental footprint of using volatile organic compounds is increasingly scrutinized, making these older methods less attractive for modern, sustainability-focused chemical production facilities.
The Novel Approach
In contrast, the novel approach described in patent CN1429196A utilizes an aqueous base solution as the solvent medium, which dramatically improves the reaction profile and operational safety. By replacing alcohol with water and a base such as sodium hydroxide or potassium carbonate, the process eliminates the fire hazards associated with organic solvents while simultaneously enhancing the reaction rate and selectivity. This aqueous system allows for better heat transfer and temperature control, which is critical for managing the exothermic nature of hydrazine reductions. The use of aqueous base also facilitates the handling of substrates containing acid groups, such as carboxyl or sulfo groups, which can be directly reduced in their salt forms without the need for protection and de-protection steps. This simplification of the synthetic route not only reduces the number of unit operations but also minimizes waste generation, aligning with green chemistry principles. The ability to conduct the reaction in a homogeneous aqueous phase ensures consistent mixing and reaction progress, leading to higher reproducibility and yield consistency across different batch sizes.
Mechanistic Insights into Hydrazine Reduction in Aqueous Base
The core of this technology lies in the interaction between the nitro compound, hydrazine, and the aqueous base, which creates a unique reaction environment conducive to efficient reduction. The mechanism involves the nucleophilic attack of hydrazine on the nitro group, facilitated by the basic conditions which help in the deprotonation steps and stabilization of intermediates. The presence of water as a solvent plays a crucial role in solvating the ionic species formed during the reaction, ensuring that the reactants remain in solution and available for transformation. The patent specifies that the reaction is preferably carried out at temperatures between 70-100°C, a range that balances reaction speed with the avoidance of excessive pressure build-up. The stoichiometry is also critical, with a preferred molar ratio of hydrazine to nitro compound ranging from 1.4 to 3, ensuring complete conversion while minimizing excess reagent waste. The base concentration is equally important, with 0.5 to 5 moles of base per mole of nitro compound recommended to maintain the necessary pH for optimal reaction kinetics. This precise control over reaction conditions allows for the suppression of side reactions, resulting in a cleaner product profile and reduced impurity levels.
Impurity control is further enhanced by the specific conditions of the aqueous base system, which influences the equilibrium between open-chain and ring-closed forms of the intermediates. For compounds with substituents capable of forming fused rings, such as those with ortho-carboxyl and meta-hydroxyalkyl groups, a pH-dependent hydrolytic equilibrium exists between the aniline form and the corresponding lactone form. The aqueous base environment allows for the manipulation of this equilibrium, enabling the selective formation of the desired product form. For instance, acidifying the reaction mixture post-reaction can drive the formation of the ring-closed lactone form, which may be the desired final product or a specific intermediate for downstream processing. This ability to toggle between structural forms by adjusting pH provides a powerful tool for purification and isolation, as different forms may have distinct solubility properties. The high chemical yields reported, often exceeding 97% in the reaction mixture, indicate that the side reactions are effectively minimized, ensuring that the majority of the starting material is converted into the target aniline compound.
How to Synthesize Aniline Compounds Efficiently
The synthesis of these high-value aniline compounds follows a streamlined protocol that is amenable to both batch and continuous processing, offering flexibility for different production scales. The general procedure involves dissolving or suspending the nitro compound in water, followed by the addition of the aqueous base solution to create the reaction medium. Once the mixture is heated to the target temperature, typically between 70°C and 100°C, the hydrazine or hydrazine hydrate is metered in over a controlled period to manage the exotherm and ensure safe operation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous base reduction technology offers substantial strategic advantages in terms of cost structure and supply reliability. The elimination of organic solvents like ethanol removes the need for complex solvent recovery systems and reduces the costs associated with solvent purchase, storage, and disposal. This simplification of the process infrastructure translates directly into lower capital investment and reduced operating expenses, making the production of these intermediates more economically viable. Furthermore, the enhanced safety profile of the aqueous system reduces insurance premiums and regulatory compliance costs, as the risks associated with flammable materials are significantly mitigated. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output and reducing the risk of batch failures. This reliability is crucial for maintaining steady supply chains, especially for critical agrochemical intermediates where downtime can have significant downstream impacts.
- Prepare an aqueous solution of the nitro compound and add the appropriate aqueous base solution.
- Heat the mixture to the desired reaction temperature between 70-100°C.
- Meter in hydrazine or hydrazine hydrate and maintain temperature until conversion is complete.
- Cost Reduction in Manufacturing: The shift to an aqueous solvent system fundamentally alters the cost equation by removing the expense and logistical burden of organic solvents. Without the need for solvent recovery distillation columns or explosion-proof equipment, the manufacturing footprint becomes more efficient and less capital intensive. The high yields achieved, often approaching theoretical maximums, mean that raw material utilization is optimized, reducing the cost per kilogram of the final product. Additionally, the ability to handle substrates with acid groups directly in their salt form avoids extra synthetic steps, further driving down the overall production cost. These cumulative savings allow for a more competitive pricing structure while maintaining healthy margins, which is essential in the price-sensitive agrochemical market.
- Enhanced Supply Chain Reliability: The use of common and readily available reagents such as hydrazine hydrate and sodium hydroxide ensures that the supply chain is not dependent on specialized or scarce materials. This availability reduces the risk of supply disruptions and allows for more flexible sourcing strategies, enhancing the overall resilience of the supply network. The process scalability, from laboratory to industrial tonnage, means that production can be ramped up quickly to meet surges in demand without requiring significant process re-engineering. The consistent quality and high purity of the output reduce the need for extensive rework or rejection, ensuring that delivery schedules are met reliably. This predictability is highly valued by downstream customers who depend on timely delivery of intermediates to keep their own production lines running smoothly.
- Scalability and Environmental Compliance: The aqueous nature of the process aligns well with increasingly stringent environmental regulations, as it minimizes the release of volatile organic compounds into the atmosphere. Waste treatment is simplified since the effluent is primarily aqueous, reducing the complexity and cost of wastewater management systems. The process can be easily scaled up in standard stirred vessels, allowing for seamless transition from pilot plant to full commercial production without significant technical barriers. This scalability ensures that the technology can support growing market demands for herbicides and other agrochemicals without compromising on quality or safety. The reduced environmental footprint also enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for major multinational corporations.
Frequently Asked Questions (FAQ)
Q: What is the primary advantage of using aqueous base over alcohol solvents?
A: Using aqueous base significantly improves reaction speed and selectivity while eliminating the need for flammable alcohol solvents, enhancing industrial safety.
Q: What yields can be expected from this reduction process?
A: The patent data indicates chemical yields in the reaction mixture are generally higher than 97%, with isolated yields ranging from 80% to 100%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method is designed for industrial scale-up, allowing for continuous or batch operation in standard stirred vessels with controlled metering.
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aniline Compounds Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the aqueous base reduction described in CN1429196A to deliver high-quality intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aniline compounds meets the exacting standards required for agrochemical synthesis. Our commitment to process excellence means that we can offer not just a product, but a reliable supply partnership that supports your long-term strategic goals. By leveraging our expertise in process optimization, we help our clients navigate the complexities of chemical manufacturing with confidence and security.
We invite you to engage with our technical procurement team to discuss how this technology can be integrated into your supply chain for maximum efficiency. Request a Customized Cost-Saving Analysis to understand the specific economic benefits applicable to your production volume and requirements. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your precise specifications. Partnering with us ensures access to cutting-edge chemistry backed by a robust manufacturing infrastructure dedicated to your success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
