Advanced Stereo-Selective Synthesis of FACBC Precursors for Commercial PET Tracer Production
The escalating demand for precise oncological diagnostics has placed positron emission tomography (PET) tracers at the forefront of modern nuclear medicine, with [18F]-FACBC emerging as a critical agent for detecting prostate cancer and other proliferative diseases. However, the commercial viability of such tracers hinges entirely on the availability of high-purity labeling precursors, a challenge addressed innovatively in patent CN101573330A. This intellectual property discloses a groundbreaking process for producing precursor compounds of radioactive halogen-labeled organic compounds, specifically targeting the synthesis of trans-[18F]-FACBC. The core innovation lies in a stereo-selective strategy that bypasses the traditionally cumbersome purification of cis/trans isomeric mixtures. By introducing a specific base during the leaving group installation step, the process differentiates between stereoisomers based on their reactivity and solubility profiles. This technical advancement represents a paradigm shift for reliable radiopharmaceutical intermediate supplier networks, enabling the production of GMP-grade precursors with unprecedented efficiency and reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of labeling precursors for [18F]-FACBC has been plagued by significant stereochemical hurdles that inflate costs and extend lead times. Conventional routes, such as those described in earlier international publications, typically involve synthesizing a mixture of cis and trans hydroxy-intermediates which must then be separated before the crucial radiofluorination step. This separation often relies on labor-intensive techniques like preparative high-performance liquid chromatography (HPLC) or repeated recrystallization, which are inherently inefficient for large-scale manufacturing. Furthermore, the physical properties of these stereoisomers are often too similar to allow for easy fractionation, leading to substantial material loss and compromised overall yields. For procurement managers, these inefficiencies translate into volatile pricing and unreliable supply chains, as the inability to consistently isolate the correct stereoisomer creates bottlenecks in the production of the final active pharmaceutical ingredient. The reliance on complex purification also increases the risk of introducing impurities that could affect the specific activity of the final radiotracer, a critical quality attribute for diagnostic efficacy.
The Novel Approach
The methodology outlined in patent CN101573330A offers a sophisticated solution by integrating the purification logic directly into the chemical transformation step itself. Instead of attempting to separate the cis and trans hydroxy-compounds prior to triflation, this novel approach subjects the mixture directly to trifluoromethanesulfonic anhydride in the presence of a tailored organic base. The brilliance of this design is that the resulting cis-leaving group adduct remains chemically stable and insoluble in the aqueous workup phase, whereas the trans-leaving group adduct reacts readily with the base to form water-soluble derivatives. This differential reactivity allows for the selective isolation of the desired cis-precursor through simple liquid-liquid extraction, effectively eliminating the need for expensive chromatographic columns. For stakeholders focused on cost reduction in PET tracer manufacturing, this translates to a drastically simplified workflow that minimizes solvent usage and processing time. The ability to start with a crude mixture of stereoisomers and end with a highly pure single isomer demonstrates a level of process intensification that is rarely seen in fine chemical synthesis.
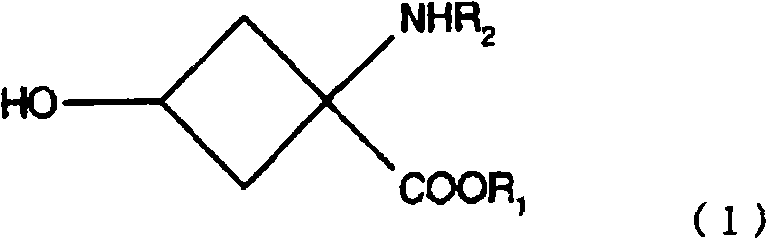
Mechanistic Insights into Base-Mediated Stereo-Differentiation
The mechanistic foundation of this invention rests on the subtle yet exploitable differences in steric environment and electronic density between the cis and trans isomers of the cyclobutane ring system. When the hydroxy-substituted intermediate is treated with a sulfonylating agent like triflic anhydride, both isomers initially form their respective triflate esters. However, the presence of a nitrogen-containing base, such as pyridine or specific alkylamines, triggers a secondary reaction pathway exclusively for the trans-isomer. It is hypothesized that the spatial arrangement of the trans-leaving group allows for a nucleophilic attack or elimination reaction facilitated by the base, converting the lipophilic triflate into a polar, water-soluble species, possibly an amine-substituted derivative or an elimination product. In stark contrast, the cis-configuration sterically hinders this secondary interaction, rendering the cis-triflate kinetically stable under the reaction conditions. This phenomenon effectively acts as a chemical filter, where the unwanted isomer is chemically modified into a form that can be easily washed away, while the desired product remains intact in the organic phase.
From an impurity control perspective, this mechanism provides a robust safeguard against cross-contamination of stereoisomers, which is a critical concern for regulatory compliance in radiopharmaceutical production. Traditional methods often struggle to reduce trans-impurities below acceptable thresholds without sacrificing yield, but this base-mediated approach ensures that any trans-material formed is actively removed from the product stream. The choice of solvent further enhances this selectivity; polar solvents like chloroform mixed with pyridine create an environment where the solubility differences are maximized. By maintaining the reaction temperature at strictly controlled low levels, typically around 0°C, the kinetic stability of the cis-isomer is preserved while preventing thermal degradation of the sensitive triflate functionality. This precise control over reaction parameters ensures that the final precursor meets the stringent purity specifications required for subsequent nucleophilic substitution with fluoride-18, guaranteeing high radiochemical yields in the final labeling step.
How to Synthesize cis-Triflate FACBC Precursor Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and thermal management to fully leverage the stereo-differentiation effect. The process begins with the preparation of a reaction mixture containing the protected hydroxy-cyclobutane carboxylate, which can be used as a crude cis/trans mixture, thereby saving upstream purification costs. To this mixture, a slight excess of trifluoromethanesulfonic anhydride is added in the presence of a molar excess of pyridine, which serves as both the base and the solvent medium. The detailed standardized synthesis steps see the guide below, which outlines the precise addition rates and quenching procedures necessary to maintain safety and reproducibility. Operators must ensure that the exothermic nature of the triflation is managed through efficient cooling systems, as temperature spikes could compromise the selectivity of the reaction. Following the reaction period, the workup involves a straightforward aqueous wash that leverages the solubility switch engineered into the trans-isomer, allowing for the recovery of the cis-precursor in high purity without the need for silica gel chromatography.
- Prepare a solution containing the cis/trans mixture of protected hydroxy-cyclobutane carboxylate, a sulfonylating agent like triflic anhydride, and a specific organic base such as pyridine.
- Maintain the reaction temperature below 4°C, preferably around 0°C, to ensure the stability of the desired cis-isomer while allowing the base to interact with the trans-isomer derivatives.
- Perform a liquid-liquid extraction using water and an organic solvent to selectively remove the water-soluble trans-isomer byproducts, isolating the stable cis-leaving group adduct.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend far beyond simple chemical yield improvements. By eliminating the dependency on complex separation technologies like preparative HPLC, manufacturers can significantly reduce the capital expenditure required for production facilities and lower the ongoing operational costs associated with column maintenance and solvent disposal. This simplification of the manufacturing process directly contributes to enhanced supply chain reliability, as the risk of batch failure due to poor separation efficiency is markedly decreased. Furthermore, the ability to utilize crude intermediate mixtures reduces the burden on upstream synthesis steps, allowing for faster throughput and shorter overall production cycles. These efficiencies collectively enable a more responsive supply chain capable of meeting the fluctuating demands of the global nuclear medicine market without the need for excessive inventory buffers.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps results in substantial cost savings by reducing solvent consumption and waste treatment expenses. Since the process relies on liquid-liquid extraction rather than solid-phase separation, the volume of organic waste generated is significantly lower, aligning with green chemistry principles and reducing environmental compliance costs. Additionally, the higher overall yield achieved by avoiding material loss during difficult separations means that less starting material is required to produce the same amount of final precursor, driving down the cost of goods sold. This economic efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a critical factor in the price-sensitive healthcare sector.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent quality output, which is vital for maintaining uninterrupted supply to radiopharmacies. By removing the variability associated with manual purification techniques, the process delivers a product with predictable specifications, reducing the need for extensive quality control testing and rework. This consistency fosters stronger partnerships between chemical suppliers and pharmaceutical companies, as it mitigates the risk of production delays caused by out-of-specification batches. Moreover, the simplicity of the process facilitates technology transfer to multiple manufacturing sites, creating a diversified supply network that is resilient to regional disruptions or capacity constraints.
- Scalability and Environmental Compliance: The transition from batch-wise chromatography to continuous or semi-continuous extraction processes greatly enhances the scalability of the production line. This scalability is essential for meeting the growing global demand for PET tracers as their clinical applications expand beyond oncology into neurology and cardiology. From an environmental standpoint, the reduction in hazardous solvent usage and the minimization of silica waste contribute to a smaller carbon footprint, helping manufacturers meet increasingly stringent sustainability targets. The process design inherently supports safe handling of reactive reagents like triflic anhydride through controlled addition protocols, ensuring adherence to occupational health and safety standards in large-scale chemical plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereo-selective synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers highlight the specific advantages related to purity, yield, and operational simplicity that distinguish this method from prior art.
Q: How does this patent improve the purity of FACBC precursors compared to conventional methods?
A: Unlike traditional methods requiring complex chromatographic separation of stereoisomers prior to labeling, this process utilizes a base-mediated kinetic differentiation during the triflation step. The unwanted trans-isomer derivatives react with the base to form water-soluble compounds, allowing them to be washed away easily, resulting in a highly pure cis-precursor essential for high-specific-activity PET imaging.
Q: What are the critical reaction conditions for maximizing the yield of the cis-isomer?
A: Temperature control is paramount; the reaction must be conducted at low temperatures, specifically not exceeding 4°C and optimally around 0°C. Additionally, the choice of base is critical, with nitrogen-containing heteroaromatic compounds like pyridine serving dual roles as both solvent and reagent to facilitate the selective solubilization of impurities.
Q: Can this synthesis method be scaled for industrial production of radiopharmaceutical intermediates?
A: Yes, the method is highly scalable because it replaces difficult solid-phase separations or preparative HPLC with standard liquid-liquid extraction. This simplification significantly reduces solvent consumption and processing time, making it ideal for commercial scale-up of complex radiopharmaceutical intermediates required for global diagnostic markets.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FACBC Precursor Supplier
As the global landscape of nuclear medicine evolves, the need for high-quality, cost-effective precursors for radiotracers like [18F]-FACBC has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies such as the one described in CN101573330A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of multinational pharmaceutical clients. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify the stereochemical integrity of every batch. This dedication to quality ensures that our precursors perform reliably in automated synthesis modules, maximizing the specific activity of the final radiopharmaceutical product.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your supply chain goals. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to reduce your raw material expenses without compromising on quality. We encourage you to request specific COA data and route feasibility assessments to verify our capabilities firsthand. Let us be your trusted partner in advancing the frontiers of diagnostic imaging through reliable chemistry and unwavering commitment to excellence.
