Advanced Synthesis of Triphenylethylene Carbazole Derivatives for Next-Generation OLED Displays
The rapid evolution of the display industry has placed unprecedented demand on the performance and longevity of organic light-emitting diode (OLED) components, particularly within the critical blue emission spectrum. Patent CN101343537A introduces a groundbreaking class of organic luminescent materials featuring a triphenylethylene carbazole derivative structure, specifically engineered to overcome the historical limitations of blue emitters. This technology represents a significant leap forward in material science, offering a robust synthetic pathway that combines high thermal stability with exceptional luminous intensity. By integrating carbazole units with a triphenylethylene core, the invention achieves a unique molecular architecture that not only facilitates efficient charge transport but also exhibits a remarkable aggregation-induced emission (AIE) effect. This specific structural motif ensures that the material maintains high quantum efficiency even in the solid state, a common failure point for many conventional fluorescent dyes. For R&D directors and procurement specialists seeking a reliable OLED material supplier, understanding the nuances of this patented synthesis is crucial for securing a competitive edge in next-generation display manufacturing.
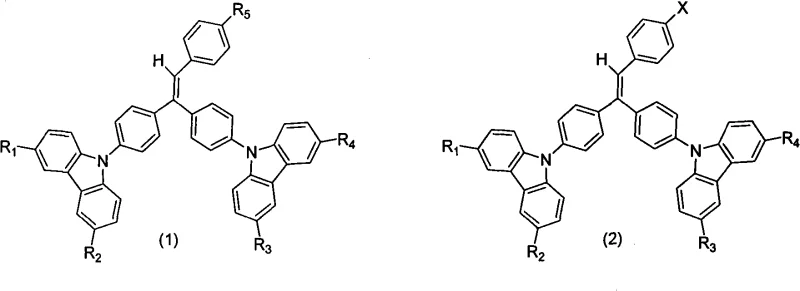
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of stable and efficient blue light-emitting materials has been plagued by significant technical hurdles, primarily centered around the instability of distyryl derivatives which were once the industry standard. These conventional materials often suffer from inherently low glass transition temperatures (Tg), which leads to morphological instability and crystallization within the thin film layers of an OLED device during operation. Such crystallization creates non-emissive dark spots that rapidly degrade device performance and shorten the operational lifespan, rendering them unsuitable for high-end commercial applications requiring thousands of hours of reliability. Furthermore, traditional distyryl compounds frequently exhibit poor fluorescence efficiency in the aggregated solid state due to aggregation-caused quenching (ACQ), where intermolecular interactions dissipate excited state energy as heat rather than light. This phenomenon necessitates complex device engineering or doping strategies to mitigate efficiency losses, thereby increasing manufacturing complexity and cost. The synthesis of these older generations of materials often involves harsh conditions or difficult purification steps that generate substantial chemical waste, posing challenges for environmental compliance and cost-effective cost reduction in electronic chemical manufacturing.
The Novel Approach
The methodology outlined in patent CN101343537A fundamentally addresses these deficiencies by employing a triphenylethylene carbazole scaffold that intrinsically possesses high thermal stability and resistance to crystallization. The novel approach leverages the rigid carbazole moiety to elevate the glass transition temperature significantly, often exceeding 120°C, which ensures the amorphous stability of the emitting layer under thermal stress. Crucially, this new class of materials harnesses the aggregation-induced emission (AIE) effect, a counter-intuitive photophysical phenomenon where the molecules become highly emissive upon aggregation, effectively turning a traditional weakness into a strength. This allows for the fabrication of highly efficient non-doped emitting layers or simplified doped systems without the severe efficiency roll-off seen in conventional systems. The synthetic strategy is modular, allowing for the precise tuning of emission wavelengths and energy levels through the variation of substituents at the R1 through R5 positions, providing a versatile platform for developing full-color displays. This flexibility, combined with a streamlined three-step synthesis involving nucleophilic substitution, olefination, and cross-coupling, offers a clear pathway for the commercial scale-up of complex polymer additives and small molecule emitters alike.
Mechanistic Insights into Wittig-Horner Olefination and Suzuki Coupling
The core of this synthetic innovation lies in the strategic conversion of a ketone intermediate into a conjugated alkene system using the Wittig-Horner reaction, followed by palladium-catalyzed functionalization. The process begins with the formation of a carbazolyl benzophenone intermediate via a nucleophilic aromatic substitution, where a halogenated benzophenone reacts with carbazole or its derivatives in the presence of a strong base like potassium tert-butoxide. This step establishes the electron-rich carbazole donors attached to the central benzophenone acceptor, setting the stage for intramolecular charge transfer characteristics essential for blue emission. Subsequently, the ketone carbonyl group is transformed into a carbon-carbon double bond using a phosphonate ester reagent under basic conditions. This Wittig-Horner transformation is preferred over the classic Wittig reaction due to the higher stability of the phosphonate carbanions and the water-soluble nature of the phosphate byproducts, which simplifies the workup and purification process significantly. The reaction proceeds through a betaine intermediate that collapses to form the alkene with high stereoselectivity, ensuring the formation of the desired trans-isomer which is critical for maintaining planarity and conjugation length.
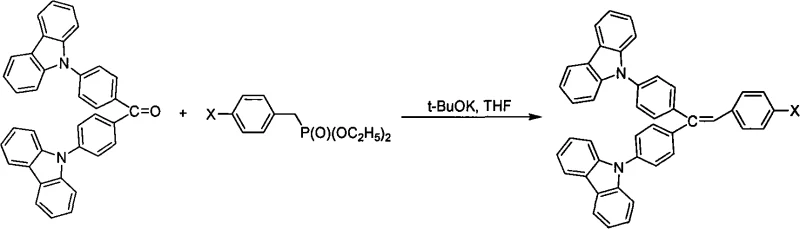
Following the formation of the vinyl backbone, the final diversification of the molecule is achieved through transition-metal catalyzed cross-coupling reactions, specifically the Suzuki-Miyaura coupling. In this step, a halogenated vinyl intermediate (typically containing a bromine atom) reacts with various aromatic boronic acids in the presence of a palladium catalyst such as tetrakis(triphenylphosphine)palladium(0). This mechanistic step involves the oxidative addition of the aryl halide to the palladium center, followed by transmetallation with the boronic acid species activated by a base like potassium carbonate, and finally reductive elimination to forge the new carbon-carbon bond. This robust coupling method allows for the introduction of diverse aromatic groups (R5), such as naphthyl, biphenyl, or pyrenyl units, which can further modulate the HOMO-LUMO gap and packing properties of the final material. Impurity control is managed through careful selection of solvents and purification techniques; for instance, the intermediates are often purified by precipitation from non-solvents like ethanol or acetone, while the final products are refined using silica gel column chromatography to remove trace palladium residues and unreacted starting materials, ensuring the high-purity OLED material specifications required for device fabrication.
How to Synthesize Triphenylethylene Carbazole Derivatives Efficiently
The synthesis of these advanced luminescent materials is designed to be operationally simple yet chemically precise, utilizing standard laboratory equipment and readily available reagents to ensure reproducibility and scalability. The process typically initiates with the preparation of the carbazolyl benzophenone core, followed by the olefination step to create the triphenylethylene backbone, and concludes with the coupling of the final aromatic substituent. Each step has been optimized in the patent examples to maximize yield and minimize side reactions, such as the use of dry tetrahydrofuran and argon protection during the Wittig-Horner reaction to prevent hydrolysis of sensitive intermediates. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures for producing gram-to-kilogram quantities, are outlined in the guide below for technical reference.
- Synthesize carbazolyl benzophenone intermediates by reacting 4-halobenzophenone with carbazole derivatives using potassium tert-butoxide in DMF.
- Convert the ketone carbonyl group into a double bond using the Wittig-Horner reaction with phosphonate esters and a strong base.
- Introduce diverse aromatic groups (R5) via palladium-catalyzed Suzuki coupling reactions to finalize the triphenylethylene structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical performance, primarily driven by the simplicity and robustness of the chemical transformations involved. The reliance on fundamental organic reactions such as nucleophilic substitution and palladium coupling means that the manufacturing process does not require exotic catalysts or extreme operating conditions, which translates directly into lower capital expenditure for reactor infrastructure and reduced operational risks. The ease of purification, often achievable through simple precipitation and filtration rather than energy-intensive distillation or complex crystallization sequences, significantly lowers the utility costs associated with solvent recovery and waste treatment. Furthermore, the modular nature of the synthesis allows for the use of commodity chemicals like carbazole and fluorobenzophenone as starting materials, which are produced at a massive global scale, ensuring a stable and resilient supply chain that is less susceptible to the volatility of niche precursor markets.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of high-yielding reactions like the Wittig-Horner olefination drastically reduce the overall cost of goods sold by minimizing material loss and labor hours. The ability to purify intermediates via precipitation rather than chromatography on a large scale represents a significant saving in silica gel and solvent consumption, directly impacting the bottom line. Additionally, the high thermal stability of the final product reduces the risk of batch failure due to thermal degradation during processing, further enhancing overall process economics and yield consistency.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials such as substituted carbazoles and halogenated benzophenones, manufacturers can source raw materials from multiple qualified vendors, mitigating the risk of single-source supply disruptions. The synthetic route avoids the use of highly unstable or hazardous reagents that require special handling and storage, simplifying logistics and reducing regulatory compliance burdens associated with transporting dangerous goods. This stability in the supply chain ensures consistent lead times and allows for better inventory planning, which is critical for meeting the just-in-time delivery requirements of major display panel manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids unit operations that are difficult to translate from bench to plant, such as cryogenic reactions or high-pressure hydrogenations. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through established recycling and treatment protocols, facilitating compliance with increasingly stringent environmental regulations. The high atom economy of the coupling reactions and the potential for solvent recovery loops contribute to a greener manufacturing profile, aligning with the sustainability goals of modern electronics corporations and enhancing the brand value of the final display products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these triphenylethylene carbazole derivatives, based on the specific embodiments and data provided in the patent documentation. These answers are designed to clarify the feasibility of implementation and the expected performance metrics for potential partners evaluating this technology for their supply chains.
Q: What are the key advantages of triphenylethylene carbazole derivatives over traditional distyryl blue emitters?
A: Unlike traditional distyryl derivatives which suffer from low glass transition temperatures and poor stability, triphenylethylene carbazole derivatives exhibit high thermal stability, high glass transition temperatures (Tg > 120°C), and significant aggregation-induced emission (AIE) effects, leading to superior solid-state luminous efficiency.
Q: How does the Wittig-Horner method improve the synthesis yield compared to standard Wittig reactions?
A: The Wittig-Horner method utilizes phosphonate esters which generally offer better stereo-selectivity and easier removal of byproducts compared to traditional phosphonium ylides, resulting in higher purity intermediates and simplified downstream purification processes.
Q: Can these materials be scaled for commercial OLED production?
A: Yes, the synthesis relies on robust, standard organic transformations such as nucleophilic aromatic substitution and palladium-catalyzed cross-coupling, which are well-established in industrial settings, allowing for reliable scale-up from laboratory grams to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylethylene Carbazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic luminescent materials play in the advancement of display technologies, and we are uniquely positioned to support your development and production needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of triphenylethylene carbazole derivative meets the exacting standards required for OLED device fabrication. Our commitment to quality and consistency makes us a trusted partner for global electronics manufacturers seeking to secure their supply of next-generation emitting materials.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthetic routes can reduce your overall material costs without compromising on performance. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive innovation and profitability in your OLED product lines.
