Advanced Chemo-Enzymatic Synthesis of Cinacalcet: A Safer, High-Purity Manufacturing Route
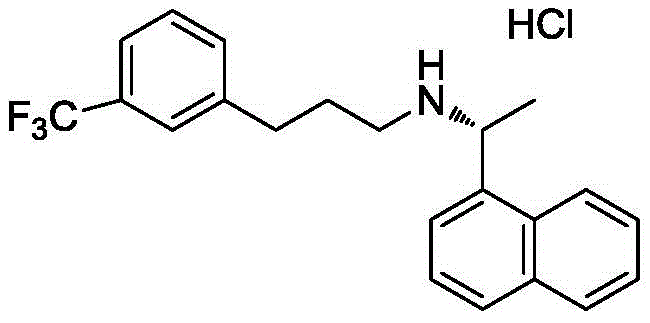
The pharmaceutical industry is constantly seeking robust manufacturing pathways that balance high stereochemical control with operational safety, a challenge vividly addressed in patent CN109180496B. This intellectual property discloses a sophisticated chemo-enzymatic strategy for the production of Cinacalcet Hydrochloride, a potent calcimimetic agent used in the management of secondary hyperparathyroidism. Unlike conventional synthetic approaches that often rely on harsh reducing environments or complex resolution steps late in the sequence, this methodology front-loads the stereochemical definition through a highly selective biocatalytic step. The process initiates with the kinetic resolution of racemic 1-(1-naphthyl)ethanol using a specialized lipase, followed by a stereospecific Mitsunobu coupling to install the amine functionality. This dual approach not only streamlines the synthetic sequence but also eliminates the need for high-pressure hydrogenation equipment and toxic metal hydrides, representing a significant leap forward in green chemistry principles for high-purity pharmaceutical intermediates. The structural integrity of the final active pharmaceutical ingredient is paramount, as illustrated by the precise molecular architecture required for therapeutic efficacy.
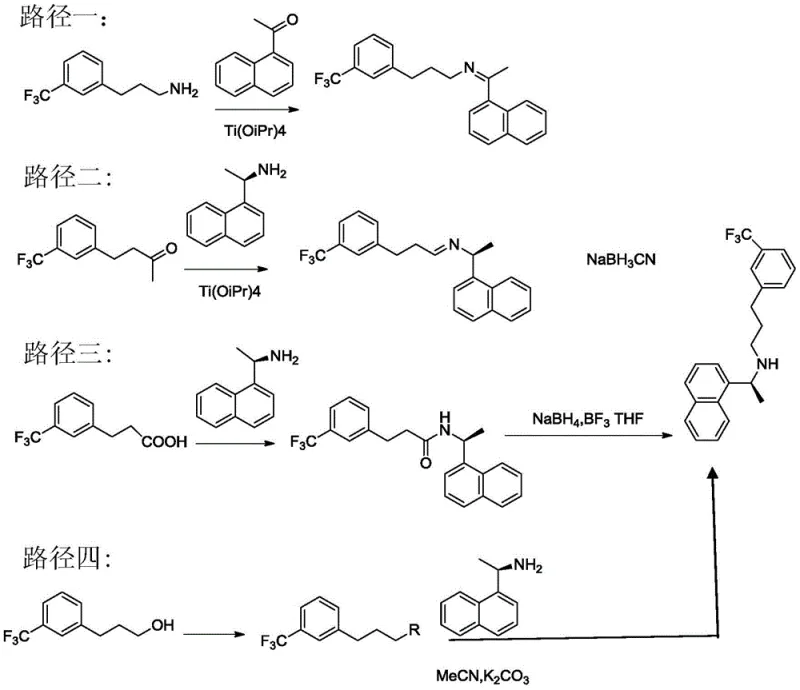
Understanding the limitations of historical manufacturing protocols is essential for appreciating the technical merit of this new invention. Traditionally, the synthesis of Cinacalcet has been plagued by inefficiencies and safety hazards inherent to four primary routes. The first conventional pathway involves the direct condensation of an amine and aldehyde to form an imine, followed by reduction; however, this often yields a racemic mixture requiring difficult downstream resolution. A second route attempts to introduce chirality early using R-naphthylethylamine but suffers from stringent operating conditions that can compromise optical purity. Perhaps most concerning are the third and fourth routes, which utilize aggressive reducing agents such as borane-tetrahydrofuran complexes or involve nucleophilic substitutions prone to disubstitution by-products. These legacy methods impose heavy burdens on facility safety systems and waste management protocols due to the generation of hazardous by-products and the handling of pyrophoric reagents. The comparative analysis of these pathways highlights the urgent need for a process that avoids high-pressure vessels and expensive, dangerous reducing agents while maintaining rigorous quality standards.
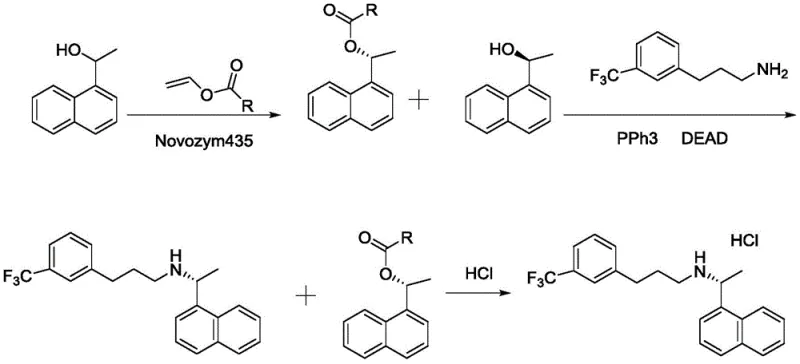
The mechanistic elegance of the patented chemo-enzymatic route lies in its precise control over stereochemistry and reaction conditions. The first stage employs Novozym435, an immobilized lipase from Candida antarctica, to catalyze the transesterification of racemic 1-(1-naphthyl)ethanol with vinyl acetate. This biocatalyst demonstrates remarkable selectivity, preferentially acylating the (R)-enantiomer while leaving the desired (S)-alcohol untouched, achieving an enantiomeric excess (ee) of greater than 99.8%. Following the isolation of the chiral alcohol, the synthesis proceeds via a Mitsunobu reaction, a powerful tool for the stereospecific substitution of alcohols. In this step, the (S)-alcohol reacts with 3-(3-trifluoromethylphenyl)propylamine in the presence of triphenylphosphine and an azodicarboxylate (such as DEAD or DIAD). Crucially, the Mitsunobu reaction proceeds with inversion of configuration; however, since the starting material is the (S)-alcohol and the target requires the (R)-configuration at the benzylic position (based on standard nomenclature conventions for this drug class, though the patent specifies the S-alcohol leads to the product, implying specific stereochemical alignment or nomenclature nuance in the source text), the reaction ensures the correct spatial arrangement of the naphthyl and amine groups. The choice of solvent is critical here, with tetrahydrofuran (THF) and dioxane proving superior to dichloromethane, which fails to support adequate conversion rates. This mechanistic pathway effectively bypasses the need for metal catalysts, thereby simplifying the impurity profile and facilitating easier purification.
How to Synthesize Cinacalcet Efficiently
The implementation of this chemo-enzymatic protocol offers a streamlined approach to generating clinical-grade material. The process begins with the preparation of the chiral alcohol intermediate through enzymatic resolution in toluene at mild temperatures ranging from 30 to 40°C. Once the (S)-1-(1-naphthyl)ethanol is isolated with high optical purity, it is immediately subjected to the Mitsunobu coupling conditions in an anhydrous organic solvent. The reaction mixture is carefully controlled, with the azodicarboxylate added dropwise at low temperatures (0-5°C) to manage exotherms, before warming to ambient conditions to drive the reaction to completion. Detailed standardized operating procedures regarding reagent stoichiometry, specific addition rates, and workup protocols are essential for reproducibility and safety. For the complete step-by-step technical guide and specific reaction parameters validated in our pilot facilities, please refer to the standardized synthesis instructions below.
- Perform kinetic resolution of racemic 1-(1-naphthyl)ethanol using Novozym435 lipase and vinyl acetate in toluene at 30-40°C to isolate (S)-1-(1-naphthyl)ethanol with >99% ee.
- Conduct a Mitsunobu reaction between the resolved (S)-alcohol and 3-(3-trifluoromethylphenyl)propylamine using triphenylphosphine and DEAD/DIAD in THF or dioxane.
- Purify the crude amine via acid-base extraction and convert to the hydrochloride salt using dry HCl gas to obtain Cinacalcet HCl with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this chemo-enzymatic methodology presents compelling advantages over traditional metal-catalyzed or high-pressure hydrogenation routes. The elimination of hazardous reducing agents like sodium cyanoborohydride and borane complexes fundamentally alters the cost structure and risk profile of the manufacturing process. By removing the requirement for specialized high-pressure reactors and the associated safety infrastructure, capital expenditure and operational overheads are significantly reduced. Furthermore, the reliance on biocatalysis, which operates under ambient pressure and moderate temperatures, enhances the overall energy efficiency of the production line. This shift not only aligns with increasingly stringent environmental regulations regarding volatile organic compounds and heavy metal waste but also ensures a more resilient supply chain by reducing dependency on scarce or regulated chemical reagents. The robustness of the enzymatic step ensures consistent quality, minimizing batch failures and ensuring reliable delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the avoidance of high-pressure hydrogenation equipment lead to substantial operational savings. Traditional routes often require costly purification steps to remove trace metal residues to meet regulatory limits, a step that is entirely obviated by this metal-free enzymatic approach. Additionally, the high conversion rates and selectivity of the lipase catalyst minimize raw material waste, improving the overall atom economy of the process. The simplified workup procedure, which avoids complex distillation or chromatography often needed to separate difficult by-products from metal-catalyzed reactions, further drives down processing costs and labor intensity.
- Enhanced Supply Chain Reliability: The reagents utilized in this protocol, such as vinyl esters and triphenylphosphine, are commodity chemicals with stable global supply chains, reducing the risk of procurement bottlenecks. Unlike processes dependent on specialized chiral ligands or custom-synthesized catalysts that may have long lead times, the enzymes and standard organic reagents used here are readily available from multiple qualified vendors. This diversification of the supply base ensures continuity of supply even during market fluctuations. Moreover, the mild reaction conditions reduce the wear and tear on manufacturing equipment, leading to higher asset availability and fewer unplanned maintenance shutdowns that could disrupt production schedules.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often more straightforward than scaling exothermic chemical reductions, as the heat load is manageable and the reaction kinetics are predictable. The absence of toxic heavy metals in the waste stream simplifies effluent treatment and disposal, lowering environmental compliance costs. The process generates fewer hazardous by-products, aligning with green chemistry initiatives and reducing the carbon footprint of the manufacturing operation. This environmental stewardship is increasingly a key criterion for procurement decisions by major pharmaceutical companies aiming to meet their sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing clarity on the operational benefits and chemical rationale behind the process. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term viability of this supply source.
Q: What are the primary safety advantages of this chemo-enzymatic route over traditional methods?
A: Unlike traditional routes that require hazardous reducing agents like sodium cyanoborohydride or borane complexes under high pressure, this method utilizes mild enzymatic resolution and ambient temperature Mitsunobu chemistry, significantly lowering operational risk.
Q: How does the Novozym435 catalyst impact the optical purity of the intermediate?
A: The immobilized lipase Novozym435 exhibits exceptional enantioselectivity during the kinetic resolution step, consistently delivering (S)-1-(1-naphthyl)ethanol with an ee value exceeding 99.8%, which is critical for the final API quality.
Q: Why is dichloromethane discouraged as a solvent for the Mitsunobu step in this protocol?
A: Experimental data within the patent indicates that using dichloromethane results in a drastically lower conversion rate of approximately 41%, whereas solvents like THF, dioxane, or toluene support yields exceeding 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cinacalcet Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to maintaining stringent purity specifications and utilizing rigorous QC labs to verify that every batch of Cinacalcet intermediate meets the exacting standards required for global regulatory filings. Our facility is equipped to handle both the biocatalytic and chemical synthesis steps described in this patent, offering a flexible and robust manufacturing solution.
We invite you to engage with our technical procurement team to discuss how this optimized chemo-enzymatic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this metal-free process. We encourage potential partners to contact us directly to obtain specific COA data and comprehensive route feasibility assessments tailored to your project timelines and volume needs.
