Scalable Production of High-Purity Benzofuran Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those exhibiting neuroprotective properties. Patent CN100345835C introduces a groundbreaking methodology for the efficient production of benzofuran derivatives, specifically focusing on 2,3-dihydrobenzofuran structures substituted at the 5-position. This innovation addresses critical bottlenecks in the manufacturing of neurodegenerative disease therapeutics by providing a route that ensures high regioselectivity and operational simplicity. The core technology enables the introduction of amino or fused ring amino groups under significantly milder conditions compared to traditional approaches, thereby minimizing side reactions and enhancing the overall impurity profile. For R&D teams evaluating new chemical entities, this patent offers a validated framework for accessing high-purity intermediates essential for drug development pipelines. 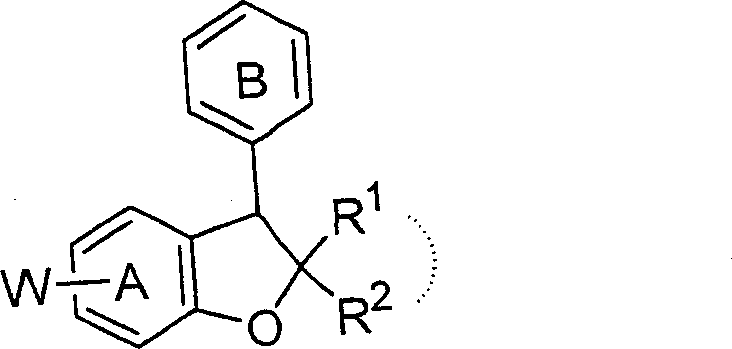
The strategic value of this synthesis lies in its adaptability to various substitution patterns on the benzene and heterocyclic rings, allowing for the generation of diverse libraries for SAR studies. By leveraging specific halogenation and coupling techniques, manufacturers can achieve precise control over the molecular architecture, which is paramount for meeting stringent regulatory standards in active pharmaceutical ingredient (API) production. The described process not only streamlines the synthetic sequence but also improves the economic viability of producing these high-value compounds. As a reliable benzofuran derivative supplier, understanding these mechanistic nuances is crucial for aligning production capabilities with the evolving demands of global pharmaceutical partners seeking cost-effective and scalable solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-substituted 2,3-dihydrobenzofurans has relied heavily on nitration or nitrosation of the parent ring followed by reduction, a pathway fraught with significant technical and safety challenges. These conventional methods often suffer from poor regioselectivity, leading to complex mixtures of isomers that require extensive and costly purification efforts to isolate the desired product. Furthermore, the use of strong nitrating agents poses substantial safety risks in large-scale operations and generates hazardous waste streams that complicate environmental compliance. The harsh reaction conditions associated with these traditional routes can also degrade sensitive functional groups, limiting the scope of compatible substrates and reducing the overall yield of the final active intermediate. For procurement managers, these inefficiencies translate into higher raw material costs and extended lead times due to the need for additional processing steps.
The Novel Approach
In contrast, the novel approach detailed in the patent utilizes a sequential strategy involving etherification, reduction, cyclization, and transition metal-catalyzed coupling to construct the target scaffold with superior precision. This method bypasses the need for direct nitration by employing selective halogenation at the 5-position, which serves as an excellent handle for subsequent palladium-catalyzed amination reactions. The ability to introduce diverse amine functionalities through cross-coupling allows for greater flexibility in molecular design while maintaining high reaction efficiency. Additionally, the cyclization step is facilitated by acid catalysis under relatively mild thermal conditions, preserving the integrity of the molecule and minimizing decomposition. This streamlined workflow significantly reduces the number of unit operations required, directly contributing to cost reduction in pharmaceutical intermediate manufacturing and enhancing the overall sustainability of the production process.
Mechanistic Insights into Palladium-Catalyzed Amination and Cyclization
The heart of this synthetic innovation lies in the precise execution of the ring-closing and amination steps, which dictate the quality and yield of the final benzofuran derivative. The cyclization mechanism involves the acid-mediated dehydration of a benzylic alcohol intermediate, promoting intramolecular nucleophilic attack by the phenolic oxygen to form the dihydrofuran ring. This step is critical for establishing the core stereochemistry and requires careful control of acid strength and temperature to prevent polymerization or rearrangement side reactions. Following cyclization, the introduction of the nitrogen substituent is achieved via a Buchwald-Hartwig type coupling, where a palladium catalyst facilitates the formation of the carbon-nitrogen bond between the aryl halide and the amine nucleophile. 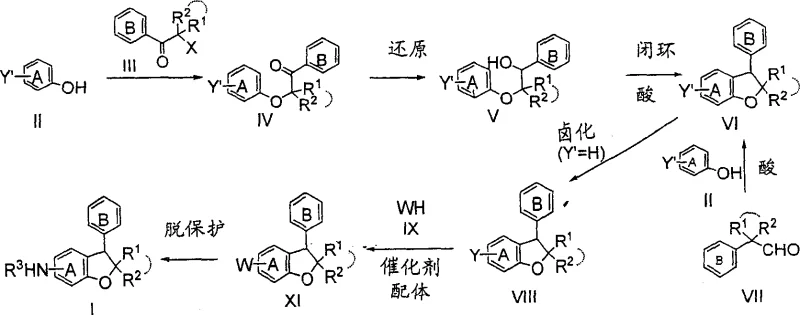
Impurity control is inherently built into this mechanism through the use of highly selective reagents and optimized reaction parameters that favor the desired transformation over competing pathways. The choice of ligands for the palladium catalyst, such as BINAP or tri-tert-butylphosphine, plays a pivotal role in accelerating the oxidative addition and reductive elimination steps, ensuring high turnover numbers and minimal metal residue in the product. Furthermore, the optional deprotection strategies outlined allow for the removal of protecting groups under mild conditions, preventing damage to the sensitive benzofuran core. For quality assurance teams, understanding these mechanistic details provides confidence in the robustness of the process and the consistency of the impurity profile across different batches, which is essential for regulatory filings and commercial supply agreements.
How to Synthesize 2,3-Dihydrobenzofuran Efficiently
Implementing this synthesis route requires a systematic approach to reaction engineering, starting with the preparation of the key ether intermediate from readily available phenol and alpha-halo ketone precursors. The process demands strict adherence to stoichiometric ratios and temperature profiles to maximize conversion during the initial alkylation and subsequent reduction phases. Operators must ensure that the reduction of the ketone to the alcohol is complete before proceeding to the acid-catalyzed cyclization, as residual ketone can lead to downstream impurities. The detailed standardized synthesis steps见下方的指南 provide a comprehensive roadmap for executing each transformation safely and effectively, ensuring that the critical quality attributes of the intermediate are maintained throughout the campaign.
- React phenol derivative with alpha-halo ketone in presence of base to form ether intermediate.
- Reduce the ketone group to alcohol using metal hydrides or catalytic hydrogenation.
- Perform acid-catalyzed ring-closing reaction to form the 2,3-dihydrobenzofuran core structure.
- Halogenate the 5-position selectively followed by palladium-catalyzed coupling with amines.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits for organizations looking to optimize their supply chain for complex heterocyclic intermediates. The elimination of hazardous nitration steps not only improves workplace safety but also reduces the burden on waste treatment facilities, leading to significant operational cost savings. The high yields reported in the examples, often exceeding ninety percent in key steps, indicate a highly efficient use of raw materials, which directly impacts the cost of goods sold. For supply chain heads, the robustness of the chemistry means fewer batch failures and more predictable production schedules, enhancing the reliability of supply for downstream API manufacturing. This process represents a strategic advantage in securing long-term contracts for high-purity 2,3-dihydrobenzofuran derivatives.
- Cost Reduction in Manufacturing: The streamlined synthetic sequence eliminates multiple purification stages and reduces solvent consumption, driving down the overall manufacturing expense per kilogram. By avoiding expensive and dangerous reagents associated with traditional nitration, the process lowers both material and disposal costs significantly. The high atom economy of the coupling reactions ensures that valuable starting materials are converted efficiently into the final product, minimizing waste. These factors combine to create a highly competitive cost structure that allows for better margin management in volatile market conditions.
- Enhanced Supply Chain Reliability: The use of stable and commercially available starting materials mitigates the risk of raw material shortages that often plague specialized synthetic routes. The robustness of the reaction conditions allows for scalability from pilot plant to commercial production without significant re-optimization, ensuring consistent supply continuity. Reduced lead time for high-purity benzofuran derivatives is achieved through faster cycle times and simplified workup procedures, enabling quicker response to market demand fluctuations. This reliability is crucial for maintaining uninterrupted production lines for critical neuroprotective medications.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing common solvents and equipment that are readily available in standard multipurpose plants. The avoidance of heavy metal waste from nitration and the ability to recover palladium catalysts contribute to a greener manufacturing footprint. Compliance with environmental regulations is simplified due to the reduced generation of hazardous byproducts, facilitating smoother permitting and auditing processes. This alignment with sustainability goals enhances the corporate reputation and meets the increasing ESG requirements of global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this benzofuran synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential manufacturing partners. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing production portfolios.
Q: What are the advantages of this benzofuran synthesis method over conventional nitration routes?
A: This method avoids harsh nitration and reduction steps, offering milder conditions, better position selectivity at the 5-position, and higher overall yields suitable for industrial scale-up.
Q: Is the process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the patent explicitly states industrial suitability, utilizing common solvents and catalysts like palladium with demonstrated high yields in multi-gram to kilogram scale examples.
Q: What is the purity profile of the resulting 2,3-dihydrobenzofuran compounds?
A: The process includes crystallization and purification steps that ensure high purity, with specific examples showing isolated yields exceeding 90% after standard workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Derivative Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this patented chemistry into commercial reality for your pharmaceutical projects. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benzofuran derivative meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for sourcing complex pharmaceutical intermediates that require advanced synthetic capabilities.
We invite you to discuss how our manufacturing capabilities can support your specific project requirements and help you achieve your development milestones. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain for success. Partner with us to leverage this innovative technology and secure a competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
