Advanced Rhodium-Catalyzed Asymmetric Hydrogenation for Commercial Scale-Up of Complex Chiral Esters
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct chiral centers, which are fundamental building blocks for bioactive molecules. Patent CN114436831A introduces a groundbreaking synthetic method for chiral 1-phenylpropyl acetate compounds, utilizing a highly sophisticated rhodium-catalyzed asymmetric hydrogenation strategy. This technology represents a significant leap forward in organic synthesis, addressing the critical need for high enantioselectivity and operational simplicity in the production of valuable pharmaceutical intermediates. By employing a chiral rhodium catalyst generated in situ from a rhodium salt precursor and a specialized ferrocene phosphine-phosphoramidite ligand, the process achieves remarkable stereocontrol. The reaction proceeds under mild conditions, specifically at room temperature and a hydrogen pressure of 20 bar, using a mixed solvent system of 1,2-dichloroethane and tert-butanol. This approach not only ensures high yields but also drastically simplifies the operational complexity typically associated with asymmetric catalysis, making it an attractive option for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of chiral ester structures has relied on methods that often suffer from significant drawbacks regarding efficiency, cost, and environmental impact. Conventional routes frequently involve the use of stoichiometric chiral auxiliaries, which require additional synthetic steps for attachment and removal, thereby increasing material costs and waste generation. Furthermore, many existing catalytic systems demand extreme reaction conditions, such as cryogenic temperatures or excessively high pressures, to achieve acceptable levels of enantioselectivity. These harsh conditions pose substantial challenges for commercial scale-up of complex polymer additives and pharmaceutical intermediates, as they necessitate specialized equipment and rigorous safety protocols. Additionally, older catalyst systems often exhibit limited substrate scope, failing to perform consistently across a diverse range of substituted alkenyl esters. This lack of versatility can lead to inconsistent batch quality and prolonged development timelines, ultimately hindering the ability to meet the demanding purity specifications required by global regulatory bodies for drug substances.
The Novel Approach
In stark contrast to these traditional limitations, the novel approach detailed in the patent utilizes a highly active chiral rhodium catalyst system that operates efficiently under ambient conditions. The core innovation lies in the specific combination of a rhodium salt precursor with a chiral ferrocene phosphine-phosphoramidite ligand, which creates a robust catalytic environment capable of inducing high stereoselectivity without the need for extreme thermal inputs. 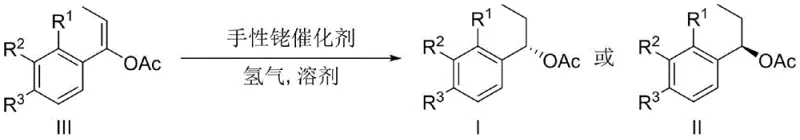 This method allows for the direct asymmetric hydrogenation of unsaturated alkenyl esters, streamlining the synthesis into a single, high-yielding step. The process demonstrates excellent functional group tolerance, accommodating various substituents on the phenyl ring without compromising reaction performance. By eliminating the need for stoichiometric chiral reagents and reducing energy consumption through room temperature operation, this technology offers a pathway for substantial cost savings in fine chemical manufacturing. The simplicity of the work-up procedure, involving standard solvent removal and column chromatography, further enhances its appeal for industrial adoption, ensuring a reliable supply chain for high-value chiral building blocks.
This method allows for the direct asymmetric hydrogenation of unsaturated alkenyl esters, streamlining the synthesis into a single, high-yielding step. The process demonstrates excellent functional group tolerance, accommodating various substituents on the phenyl ring without compromising reaction performance. By eliminating the need for stoichiometric chiral reagents and reducing energy consumption through room temperature operation, this technology offers a pathway for substantial cost savings in fine chemical manufacturing. The simplicity of the work-up procedure, involving standard solvent removal and column chromatography, further enhances its appeal for industrial adoption, ensuring a reliable supply chain for high-value chiral building blocks.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The exceptional performance of this synthesis route is rooted in the precise molecular architecture of the catalyst system, which dictates the stereochemical outcome of the hydrogenation. The chiral rhodium catalyst is formed in situ, where the rhodium center coordinates with the bidentate ferrocene phosphine-phosphoramidite ligand to create a well-defined chiral pocket. 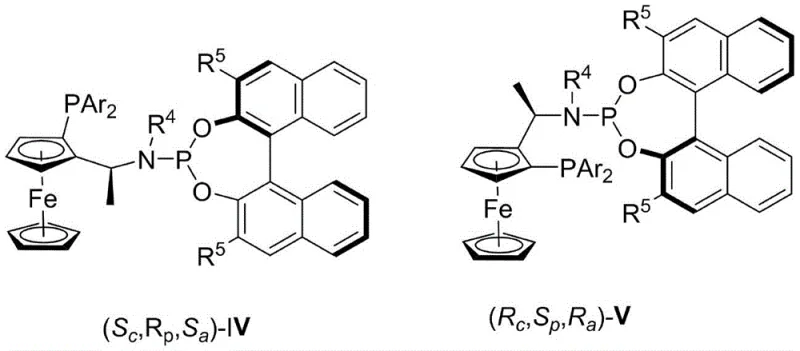 This ligand framework, featuring a ferrocene backbone and bulky binaphthyl-derived phosphoramidite groups, exerts significant steric influence over the substrate binding site. During the catalytic cycle, the alkene moiety of the 1-phenylprop-1-en-1-yl acetate coordinates to the rhodium center, followed by the oxidative addition of hydrogen. The chiral environment imposed by the ligand forces the hydrogen atoms to add to the double bond from a specific face, thereby establishing the new chiral center with high fidelity. The electronic properties of the ligand also play a crucial role in stabilizing the transition state, ensuring that the reaction proceeds rapidly even at room temperature. This mechanistic understanding is vital for R&D directors focusing on impurity profiles, as the high selectivity minimizes the formation of unwanted diastereomers and regioisomers, leading to a cleaner crude product profile.
This ligand framework, featuring a ferrocene backbone and bulky binaphthyl-derived phosphoramidite groups, exerts significant steric influence over the substrate binding site. During the catalytic cycle, the alkene moiety of the 1-phenylprop-1-en-1-yl acetate coordinates to the rhodium center, followed by the oxidative addition of hydrogen. The chiral environment imposed by the ligand forces the hydrogen atoms to add to the double bond from a specific face, thereby establishing the new chiral center with high fidelity. The electronic properties of the ligand also play a crucial role in stabilizing the transition state, ensuring that the reaction proceeds rapidly even at room temperature. This mechanistic understanding is vital for R&D directors focusing on impurity profiles, as the high selectivity minimizes the formation of unwanted diastereomers and regioisomers, leading to a cleaner crude product profile.
Furthermore, the presence of tert-butanol as an additive in the reaction medium contributes significantly to the stabilization of the active catalytic species and the enhancement of enantioselectivity. While the exact mechanistic role of the alcohol additive can vary, it is believed to facilitate proton transfer steps or stabilize specific intermediates within the catalytic cycle through hydrogen bonding interactions. The use of 1,2-dichloroethane as the primary solvent provides an optimal balance of polarity and solubility for both the organic substrate and the metal complex, ensuring homogeneous reaction conditions throughout the process. This homogeneity is critical for maintaining consistent reaction kinetics on a large scale, preventing localized hot spots or concentration gradients that could lead to variability in product quality. For procurement managers, understanding these mechanistic nuances underscores the robustness of the process, as it relies on well-defined chemical principles rather than empirical trial-and-error, reducing the risk of batch failures and ensuring cost reduction in pharmaceutical intermediate manufacturing through predictable outcomes.
How to Synthesize Chiral 1-Phenylpropyl Acetate Efficiently
The implementation of this synthesis route involves a straightforward sequence of operations that can be easily adapted for both laboratory-scale optimization and pilot-plant production. The process begins with the preparation of the active catalyst under an inert atmosphere to prevent oxidation of the sensitive rhodium species. Following catalyst activation, the substrate is introduced along with the specific solvent mixture and additive, and the system is pressurized with hydrogen gas. The reaction is then allowed to proceed with stirring until completion, monitored by standard analytical techniques such as GC or HPLC. Detailed standardized synthesis steps see the guide below.
- Prepare the chiral rhodium catalyst in situ by mixing a rhodium salt precursor with a chiral ferrocene phosphine-phosphoramidite ligand in a solvent mixture under nitrogen protection.
- Introduce the 1-phenylprop-1-en-1-yl acetate substrate and a tert-butanol additive to the catalyst solution within a pressurized reactor.
- Conduct the asymmetric hydrogenation at room temperature under 20 bar hydrogen pressure for 24 hours, followed by solvent removal and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this rhodium-catalyzed hydrogenation technology offers transformative benefits for procurement and supply chain stakeholders seeking to optimize their sourcing strategies. The primary advantage lies in the drastic simplification of the manufacturing process, which directly translates to enhanced supply chain reliability and reduced operational overheads. By operating at room temperature and moderate hydrogen pressures, the process eliminates the need for expensive cryogenic cooling systems or high-pressure autoclaves rated for extreme conditions, thereby lowering capital expenditure requirements for production facilities. This accessibility allows for a broader base of qualified manufacturers to produce the intermediate, reducing the risk of supply bottlenecks and ensuring continuity of supply for downstream drug manufacturers. Moreover, the high conversion rates and selectivity minimize the need for extensive purification steps, such as repeated recrystallizations or complex chromatographic separations, which are often time-consuming and costly.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the use of a catalytic amount of rhodium significantly lower the raw material costs associated with the synthesis. Since the catalyst loading is low and the turnover number is high, the overall consumption of precious metals is minimized, contributing to substantial cost savings. Additionally, the mild reaction conditions reduce energy consumption related to heating or cooling, further driving down the utility costs per kilogram of product. The simplified work-up procedure also reduces solvent usage and waste disposal costs, aligning with green chemistry principles and reducing the environmental compliance burden. These factors collectively enable a more competitive pricing structure for the final chiral ester, providing a clear economic advantage in cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system and the use of commercially available starting materials ensure a stable and predictable production schedule. Unlike processes that rely on bespoke reagents with long lead times, the substrates and ligands for this method are accessible from multiple global suppliers, mitigating the risk of raw material shortages. The high yield and consistency of the reaction mean that production targets can be met with fewer batches, reducing the logistical complexity of managing multiple production runs. This reliability is crucial for reducing lead time for high-purity chiral esters, allowing pharmaceutical companies to accelerate their drug development timelines and bring products to market faster without compromising on quality or supply security.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction parameters that are easily transferable from bench scale to multi-ton production. The use of standard solvents and moderate pressures simplifies the engineering requirements for scale-up, facilitating the commercial scale-up of complex chiral intermediates without significant process redesign. Furthermore, the high atom economy of the hydrogenation reaction and the reduction in waste generation support stringent environmental regulations. The ability to recycle solvents and the minimal generation of heavy metal waste due to low catalyst loading enhance the sustainability profile of the manufacturing process. This alignment with environmental standards not only ensures regulatory compliance but also enhances the corporate social responsibility profile of the supply chain, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of the method for potential adopters. Understanding these details is essential for making informed decisions about integrating this route into existing manufacturing workflows.
Q: What are the key advantages of the rhodium-ferrocene ligand system described in CN114436831A?
A: The system offers exceptional enantioselectivity (up to 95% ee) and high conversion rates (up to 99%) under mild room temperature conditions, significantly simplifying the thermal management requirements for commercial scale-up of complex chiral intermediates.
Q: How does this method improve supply chain reliability for high-purity chiral esters?
A: By utilizing readily available substrates and eliminating the need for extreme cryogenic conditions or stoichiometric chiral auxiliaries, the process reduces lead time for high-purity chiral esters and minimizes dependency on specialized low-temperature infrastructure.
Q: Is this synthesis route scalable for industrial pharmaceutical intermediate production?
A: Yes, the use of robust rhodium catalysts and standard hydrogenation pressures (20 bar) alongside common solvents like 1,2-dichloroethane ensures the commercial scale-up of complex chiral intermediates is feasible with existing reactor setups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 1-Phenylpropyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN114436831A can be successfully translated into robust industrial processes. We are committed to delivering high-purity chiral esters that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex catalytic reactions safely and efficiently, guaranteeing a consistent supply of high-quality intermediates for your drug development programs.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget without sacrificing quality. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of high-performance chiral building blocks for your next breakthrough therapy.
