Scalable Synthesis Of Novel Aryl Pentadiene Amide ALDH2 Agonists For Cardiovascular Therapeutics
Scalable Synthesis Of Novel Aryl Pentadiene Amide ALDH2 Agonists For Cardiovascular Therapeutics
The pharmaceutical landscape for treating alcoholism and cardiovascular diseases is undergoing a significant transformation with the discovery of small molecule agonists targeting Aldehyde Dehydrogenase 2 (ALDH2). Patent CN114149386A introduces a groundbreaking class of aryl pentadiene amide aldehyde dehydrogenase inhibitors that demonstrate superior agonistic activity and enhanced water solubility compared to existing controls like Alda-1. This technological leap is critical for the East Asian population, where a significant percentage carries the less active ALDH2*2 variant, predisposing them to higher risks of oxidative stress-related diseases. As a leading manufacturer, we recognize that the commercial viability of these therapeutics hinges on robust, scalable synthetic routes that ensure high purity and consistent supply for global drug developers.
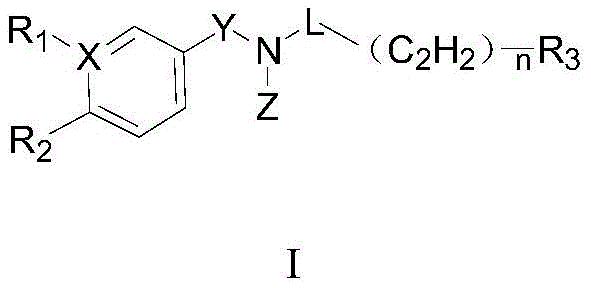
The core innovation lies in the structural versatility of the amide backbone, which allows for extensive modification to optimize pharmacokinetic properties. The patent details a comprehensive library of compounds where substituents such as halogens, alkoxy groups, and heteroaryl rings are strategically positioned to maximize binding affinity. For procurement managers and R&D directors, understanding the synthetic accessibility of these complex scaffolds is paramount. The disclosed methods utilize widely available starting materials and standard coupling protocols, suggesting a clear path toward cost-effective commercial production without compromising on the stringent quality standards required for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of ALDH2 agonists has been hampered by poor pharmacokinetic profiles, specifically low aqueous solubility and short half-lives in vivo. Traditional small molecule approaches often struggled to achieve the necessary balance between lipophilicity for membrane permeability and hydrophilicity for systemic distribution. Furthermore, earlier synthetic routes frequently relied on harsh reaction conditions or expensive transition metal catalysts that introduced difficult-to-remove impurities, complicating the purification process and inflating manufacturing costs. These limitations hindered the clinical translation of promising candidates, leaving a significant unmet medical need for effective treatments for acute alcoholism and ischemic heart disease.
The Novel Approach
The methodology outlined in CN114149386A overcomes these hurdles through a rational design strategy centered on amide bond formation using efficient coupling reagents. By employing reagents like HATU (1-[Bis(dimethylamino)methylene]-1H-1,2,3-triazolo[4,5-b]pyridinium 3-oxid hexafluorophosphate) in conjunction with organic bases like DIEA, the synthesis proceeds under mild conditions, often at room temperature. This approach not only preserves the integrity of sensitive functional groups but also minimizes the formation of side products. The resulting compounds, such as N-(benzo[d][1,3]dioxol-5-ylmethyl)anthracene-9-carboxamide, exhibit markedly improved solubility and potency, validating the efficacy of this synthetic platform for generating high-value pharmaceutical intermediates.
Mechanistic Insights into HATU-Mediated Amide Coupling
The cornerstone of this synthesis is the activation of the carboxylic acid moiety to facilitate nucleophilic attack by the amine component. In the presence of HATU and DIEA, the carboxylic acid is converted into a highly reactive O-acylisourea or active ester intermediate. This activated species is exceptionally electrophilic, allowing even sterically hindered or electron-deficient amines to react efficiently. The mechanism ensures rapid conversion rates, which is crucial for minimizing reaction times and energy consumption in a commercial setting. Moreover, the use of anhydrous DMF as a solvent provides a polar environment that stabilizes the transition state, further driving the reaction to completion with high yields, as evidenced by the 84.1% yield reported for Compound 1.
Impurity control is another critical aspect addressed by this mechanistic pathway. The specificity of the HATU coupling reduces the likelihood of racemization or over-alkylation, common pitfalls in peptide and amide synthesis. Post-reaction workup involves simple acid-base extraction or precipitation, effectively removing urea byproducts and unreacted starting materials. For instance, pouring the reaction mixture into dilute hydrochloric acid precipitates the product as a white solid, which can be further purified by recrystallization or silica gel column chromatography. This streamlined purification process is essential for meeting the rigorous purity specifications demanded by regulatory bodies for clinical-grade intermediates.
![Multi-step synthesis route for 2-((benzo[d][1,3]dioxol-5-ylmethyl)carbamoyl)-3-chlorobenzoic acid](/insights/img/aldh2-agonist-intermediate-synthesis-pharma-supplier-20260307050418-07.webp)
How to Synthesize ALDH2 Agonist Intermediates Efficiently
The synthesis of these high-potency intermediates follows a logical sequence of activation, coupling, and purification that is amenable to both laboratory and pilot-scale operations. The process begins with the dissolution of the carboxylic acid precursor in anhydrous DMF, followed by the sequential addition of the coupling agent and base to generate the active ester. Once the amine nucleophile is introduced, the mixture is stirred to allow the amide bond to form. Detailed standardized synthesis steps for specific derivatives are provided in the guide below, ensuring reproducibility and consistency across batches.
- Dissolve the carboxylic acid starting material in anhydrous DMF and activate it using HATU and DIEA at room temperature.
- Add the appropriate amine component (e.g., piperonylamine or substituted benzylamine) and stir the reaction mixture for 24 hours.
- Quench the reaction with dilute hydrochloric acid, filter the precipitated solid, and purify via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthetic routes described in this patent offer substantial advantages in terms of raw material availability and process safety. The starting materials, such as various substituted benzoic acids, phthalic anhydrides, and benzylamines, are commodity chemicals readily sourced from the global fine chemical market. This abundance mitigates the risk of supply disruptions and allows for competitive pricing strategies. Furthermore, the reactions typically proceed at room temperature or with mild heating, eliminating the need for specialized cryogenic equipment or high-pressure reactors, thereby reducing capital expenditure and operational complexity for manufacturing partners.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard coupling reagents significantly lower the direct material costs associated with production. Additionally, the high yields reported (often exceeding 80%) minimize waste generation and maximize the throughput of the final product. The simplified workup procedures, which often involve filtration rather than complex extractions, reduce solvent consumption and labor hours, contributing to a leaner and more cost-efficient manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The robustness of the synthetic method ensures consistent batch-to-batch quality, which is vital for maintaining long-term supply contracts with pharmaceutical clients. The reliance on stable, non-hazardous reagents simplifies logistics and storage requirements, reducing the regulatory burden associated with transporting dangerous goods. This stability translates into shorter lead times and greater flexibility in responding to fluctuating market demands, positioning suppliers who master this chemistry as reliable partners in the drug development value chain.
- Scalability and Environmental Compliance: The processes described are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to kilogram and ton-scale commercial production without significant re-optimization. The use of common solvents like ethyl acetate and ethanol facilitates solvent recovery and recycling, aligning with green chemistry principles and environmental regulations. This compliance not only avoids potential fines but also appeals to environmentally conscious stakeholders, adding a layer of corporate social responsibility value to the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these ALDH2 agonist intermediates. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and synthesis feasibility.
Q: How does the solubility of these novel amides compare to existing ALDH2 agonists?
A: According to patent CN114149386A, the disclosed amide compounds exhibit significantly improved water solubility and higher agonistic activity compared to the positive control Alda-1, addressing key pharmacokinetic limitations.
Q: What are the primary therapeutic applications for these intermediates?
A: These intermediates are designed for treating diseases related to ALDH2 activity, including acute alcoholism, cardiovascular diseases like myocardial ischemia, and oxidative stress-related disorders.
Q: Is the synthesis process suitable for large-scale manufacturing?
A: Yes, the synthesis utilizes standard coupling reagents like HATU and common solvents like DMF and ethyl acetate, operating often at room temperature, which facilitates easy scale-up and cost reduction in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ALDH2 Agonist Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to market-ready reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ALDH2 agonist intermediate meets the highest international standards. We understand the critical nature of timeline and quality in the pharmaceutical industry and are committed to delivering excellence in every shipment.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Whether you need specific COA data for regulatory filings or route feasibility assessments for novel analogs, our experts are ready to provide the support you need. Partner with us to leverage our technical expertise and secure a stable, high-quality supply of these vital cardiovascular and anti-alcoholism therapeutic intermediates.
