Advanced Synthesis of 2,5-Diamino Benzene Ethanol for Industrial Hair Dye Applications
The global demand for high-performance oxidative hair dye intermediates necessitates a rigorous re-evaluation of synthetic pathways to ensure both economic viability and chemical purity. Patent CN101698647B introduces a transformative methodology for the production of 2,5-diamino benzene ethanol, shifting the paradigm from expensive, low-yield precursors to a robust o-nitrotoluene-based architecture. This technical breakthrough addresses critical bottlenecks in the supply chain of personal care ingredients by leveraging a multi-step sequence that includes carboxylation, acetylation, nitration, esterification, reduction, hydrolyzation, and hydrogenation. By fundamentally altering the starting material strategy, this process mitigates the risks associated with volatile raw material pricing and complex purification protocols that have historically plagued the manufacturing of aromatic diamines. The strategic implementation of protective groups and catalytic hydrogenation ensures that the final product meets the stringent quality standards required for direct application in consumer cosmetic formulations. Furthermore, the scalability of this route offers a distinct competitive advantage for manufacturers seeking to optimize their production capacity without compromising on the integrity of the molecular structure. As the industry moves towards more sustainable and cost-efficient chemical manufacturing, this patented approach stands out as a benchmark for modern intermediate synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-diamino benzene ethanol has been hindered by reliance on suboptimal starting materials such as o-chlorophenylacetic acid or o-amino benzene ethanol, which introduce severe inefficiencies into the production workflow. When utilizing o-chlorophenylacetic acid, traditional routes often suffer from catastrophic yield losses during the benzylamine substitution step, where recovery rates can plummet to negligible levels, rendering the process economically unfeasible for large-scale operations. Alternative pathways attempting to reduce nitro-acetic acid derivatives frequently encounter the formation of indole byproducts, which are notoriously difficult to separate and significantly degrade the overall purity of the final batch. Moreover, routes initiating from o-amino benzene ethanol face substantial challenges during the nitration phase, where the generation of persistent byproducts like 2-amino-4-nitro ethanol diacetylation compounds complicates downstream processing and increases waste disposal costs. These conventional methods not only inflate the cost of goods sold due to poor atom economy but also introduce significant supply chain volatility due to the scarcity and high price of the requisite precursors. Consequently, manufacturers relying on these legacy technologies struggle to maintain consistent quality and competitive pricing in a rapidly evolving market.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent utilizes o-nitrotoluene as a foundational building block, offering a dramatic reduction in raw material costs and a significant enhancement in process stability. This innovative route employs a carefully orchestrated sequence of reactions that begins with a carboxylation step using carbon dioxide, followed by a highly efficient catalytic hydrogenation to establish the amino functionality early in the synthesis. The strategic use of acetylation serves as a protective measure before nitration, effectively directing the substitution to the desired position and minimizing the formation of unwanted isomers that plague other methods. Subsequent steps involving esterification and selective reduction with sodium borohydride allow for precise manipulation of the side chain without affecting the sensitive aromatic amine groups. The final stages involve hydrolysis and a concluding hydrogenation step to yield the target diamine, typically isolated as a stable sulfate salt. This comprehensive pathway ensures that each transformation proceeds with high conversion rates, eliminating the bottlenecks associated with low-yield steps found in prior art. 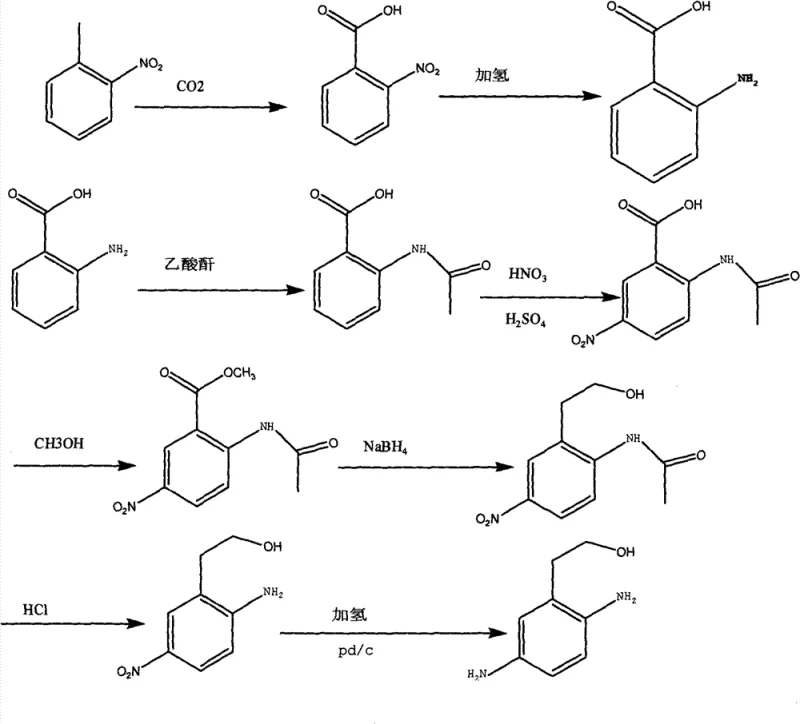
Mechanistic Insights into Pd/C Catalyzed Hydrogenation and Selective Reduction
The core of this synthesis lies in the precise control of reduction potentials and the strategic protection of reactive functional groups throughout the multi-step sequence. The initial hydrogenation of o-nitrophenylacetic acid utilizes a palladium on carbon (Pd/C) catalyst under controlled pressure and temperature conditions to selectively reduce the nitro group to an amine without affecting the carboxylic acid moiety. This chemoselectivity is paramount, as over-reduction or side reactions at this stage would compromise the entire synthetic trajectory and lead to complex mixtures that are difficult to purify. Following acetylation, the nitration step is conducted at low temperatures to manage the exothermic nature of the reaction and ensure regioselectivity, preventing the oxidation of the sensitive amino group which is now protected as an amide. The subsequent reduction of the ester group to a primary alcohol using sodium borohydride is another critical mechanistic feature, as it allows for the introduction of the hydroxyethyl side chain under mild conditions that preserve the integrity of the nitro group on the ring. This orthogonal reactivity ensures that the molecule evolves predictably through each stage, maintaining high structural fidelity. Finally, the removal of the acetyl protecting group via acid hydrolysis and the final catalytic hydrogenation of the remaining nitro group complete the transformation, yielding the diamine with exceptional purity profiles suitable for sensitive cosmetic applications.
Impurity control is rigorously managed through the physical properties of the intermediates and the specific reaction conditions employed at each juncture of the synthesis. The crystallization behaviors of intermediates such as o-aminophenylacetic acid and the various nitro-acetylated derivatives are leveraged to purge trace contaminants before they can propagate through the synthesis. By maintaining strict temperature controls during the nitration and hydrolysis steps, the formation of thermal degradation products and polymeric tars is effectively suppressed. The use of solvent systems like chloroform and methanol is optimized to maximize the solubility of desired products while precipitating inorganic salts and side products during workup phases. Furthermore, the final salification with sulfuric acid not only stabilizes the highly reactive diamine but also facilitates a final purification step where inorganic impurities and residual organic byproducts can be washed away. This multi-layered approach to quality assurance ensures that the final 2,5-diamino benzene ethanol sulfate meets the rigorous specifications demanded by international regulatory bodies for use in personal care products. The result is a material with a consistent impurity profile that minimizes the risk of skin sensitization and ensures uniform dyeing performance in end-user formulations.
How to Synthesize 2,5-Diamino Benzene Ethanol Efficiently
The operational execution of this synthesis requires careful attention to reaction parameters and safety protocols to maximize yield and ensure operator safety throughout the production cycle. The process begins with the carboxylation of o-nitrotoluene in a polar aprotic solvent, requiring precise gas flow control of carbon dioxide and maintenance of anhydrous conditions to prevent premature hydrolysis of reactive intermediates. Subsequent hydrogenation steps must be conducted in specialized pressure vessels with adequate venting and inert gas purging to mitigate the risks associated with hydrogen gas handling. The acetylation and nitration stages demand rigorous temperature monitoring to prevent runaway exotherms, while the reduction steps require the controlled addition of hydride reagents to manage gas evolution. Detailed standard operating procedures for each unit operation are essential to replicate the high yields reported in the patent literature consistently. For a comprehensive breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized guide below.
- Carboxylation of o-nitrotoluene with CO2 in DMF using sodium phenylate and sodium methylate to form o-nitrophenylacetic acid.
- Catalytic hydrogenation of o-nitrophenylacetic acid using Pd/C to produce o-aminophenylacetic acid.
- Protection via acetylation with acetic anhydride followed by nitration to introduce the second nitro group at the 5-position.
- Esterification, selective reduction of the ester to alcohol using NaBH4, hydrolysis of the amide, and final hydrogenation to yield the diamine sulfate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis route offers profound advantages by decoupling production costs from the volatile pricing of specialized fine chemical precursors. By anchoring the synthesis on o-nitrotoluene, a commodity chemical produced in massive volumes globally, manufacturers can secure a stable and predictable supply chain that is resilient to market fluctuations. This shift eliminates the dependency on scarce intermediates like o-amino benzene ethanol, which historically command premium prices due to their complex manufacturing requirements and limited supplier base. The robustness of the new process also translates into reduced operational expenditures, as the high yields per step minimize the volume of raw materials required to produce a given mass of final product. Additionally, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to lower utility costs and a smaller carbon footprint. These factors combine to create a compelling economic model that enhances margin potential while ensuring long-term supply continuity for downstream formulators.
- Cost Reduction in Manufacturing: The transition to o-nitrotoluene as a starting material represents a fundamental shift in cost structure, leveraging a raw material that is significantly less expensive than traditional precursors while offering superior availability. The elimination of low-yield steps, such as the problematic benzylamine substitution found in older routes, drastically reduces the loss of valuable intermediates and minimizes the cost of waste treatment. Furthermore, the high selectivity of the reactions reduces the need for extensive chromatographic purification, allowing for simpler crystallization-based isolation methods that are far more cost-effective at scale. This cumulative effect results in a substantially lower cost of goods sold, enabling competitive pricing strategies in the global market for hair dye ingredients.
- Enhanced Supply Chain Reliability: Utilizing a commodity feedstock like o-nitrotoluene ensures that production is not held hostage by the capacity constraints of niche intermediate suppliers, thereby guaranteeing consistent delivery schedules. The simplicity of the reaction sequence also means that the process can be easily replicated across multiple manufacturing sites, diversifying supply risk and preventing single-point failures. The stability of the intermediates allows for safer storage and transportation, reducing the logistical complexities associated with hazardous or unstable chemicals. This reliability is crucial for maintaining the production schedules of major cosmetic brands that depend on just-in-time delivery of high-quality ingredients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily managed in large-scale reactors without the need for exotic equipment or extreme pressures. The high atom economy and reduced generation of hazardous byproducts align with modern environmental regulations, simplifying the permitting process and reducing the burden on effluent treatment plants. The ability to recycle solvents and recover catalysts further enhances the sustainability profile of the manufacturing operation. This alignment with green chemistry principles not only mitigates regulatory risk but also appeals to increasingly eco-conscious consumers and brand partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced hair dye intermediate. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent documentation to ensure accuracy and relevance. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into their existing product lines. The responses cover aspects ranging from raw material sourcing to final product stability and regulatory compliance.
Q: Why is o-nitrotoluene preferred over o-amino benzene ethanol as a starting material?
A: o-Nitrotoluene is significantly more cost-effective and readily available compared to o-amino benzene ethanol, which commands a much higher market price and introduces nitration purification challenges.
Q: How does this process ensure high purity for cosmetic applications?
A: The route utilizes an acetylation protection strategy prior to nitration, which minimizes byproduct formation and allows for precise control over the substitution pattern, ensuring high purity specifications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions, stable high yields across all steps, and avoids hazardous low-yield transformations, making it highly suitable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diamino Benzene Ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for the personal care industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,5-diamino benzene ethanol meets the exacting standards required for oxidative hair dye formulations. Our commitment to quality extends beyond mere compliance; we actively collaborate with our clients to optimize the performance of these ingredients in their specific end-use applications. By partnering with us, you gain access to a supply chain that is both resilient and responsive to the dynamic needs of the global cosmetics market.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific manufacturing requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this superior grade of intermediate. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your production volume and quality targets. Let us help you engineer a more profitable and sustainable supply chain for your next generation of hair care products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
