Advanced Synthesis of High-Purity Enantiomeric Phenylpropionic Acid Intermediates for Metabolic Therapies
Advanced Synthesis of High-Purity Enantiomeric Phenylpropionic Acid Intermediates for Metabolic Therapies
The pharmaceutical landscape for treating metabolic syndromes such as type II diabetes is undergoing a significant transformation driven by the demand for higher efficacy and reduced side effects. A pivotal development in this sector is detailed in patent CN112479977B, which discloses a novel enantiomeric compound, specifically (-)-2-[(2-(4-fluorobenzoyl)phenyl)amino]-3-[(4-(2-carbazolylethoxy)phenyl)]propionic acid, and its pharmaceutically acceptable salts. This innovation represents a critical leap forward from previous racemic formulations, offering a targeted therapeutic approach that maximizes biological activity while minimizing potential toxicity associated with inactive isomers. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the structural nuances and synthesis pathways of this molecule is essential for securing a competitive edge in the development of next-generation antidiabetic agents.
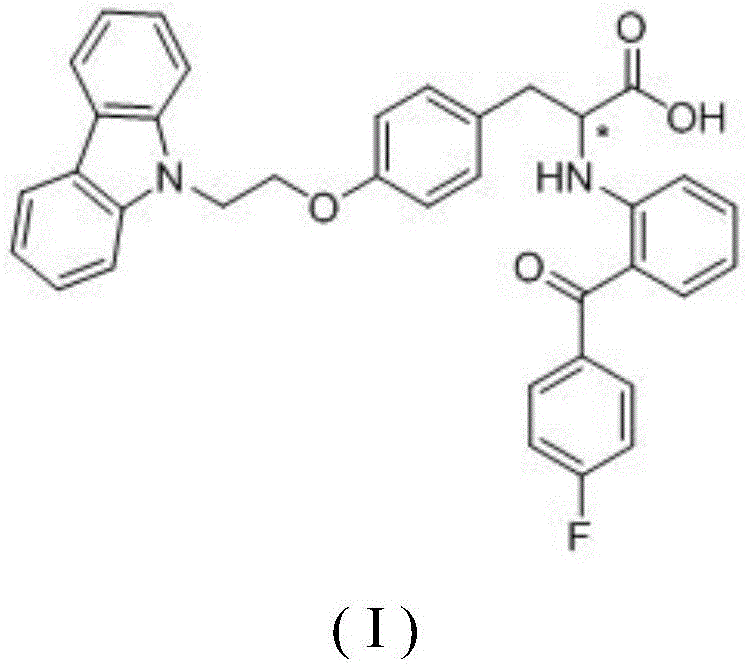
The core value proposition of this technology lies in the stereoselective optimization of PPAR activators. While the parent racemic compound was known to possess activity, the isolation of the specific levorotatory isomer has revealed unexpected pharmacological superiority. This compound functions as a potent activator of the RXR/PPAR heterodimers, which are nuclear receptors crucial for regulating lipid metabolism and glucose homeostasis. By focusing on the single active enantiomer, manufacturers can achieve cost reduction in API manufacturing by eliminating the processing and disposal costs associated with the therapeutically inferior (+)-enantiomer, thereby streamlining the production workflow and enhancing the overall economic viability of the drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of PPAR agonists often relied on racemic mixtures, where both the (+) and (-) enantiomers were present in equal proportions. As highlighted in the background of the patent, while racemates are easier to synthesize initially, they introduce significant inefficiencies in the later stages of drug development. The presence of the inactive or less active enantiomer acts essentially as an impurity, diluting the potency of the final formulation and potentially contributing to off-target effects or unpredictable metabolic clearance rates. Furthermore, regulatory agencies increasingly scrutinize chiral drugs, often requiring extensive data to prove the safety of the unwanted isomer, which prolongs the timeline for clinical approval and increases the financial burden on pharmaceutical developers seeking high-purity OLED material or pharmaceutical grade intermediates.
The Novel Approach
The novel approach presented in this patent circumvents these issues through a sophisticated chiral separation strategy using supercritical fluid chromatography (SFC). Instead of attempting a difficult asymmetric synthesis from scratch, the inventors utilized a resolution technique to isolate the desired (-)-enantiomer from the racemic precursor with exceptional precision. This method leverages the differential interaction of enantiomers with a chiral stationary phase (Chiralpak column) under supercritical conditions. The result is a highly pure product with an enantiomeric excess (ee%) reaching 100% for the free acid form. This level of purity is critical for ensuring consistent pharmacokinetic profiles and is a key selling point for any commercial scale-up of complex pharmaceutical intermediates intended for sensitive metabolic indications.
Mechanistic Insights into Chiral Separation via Supercritical Fluid Chromatography
The separation mechanism relies on the subtle differences in the three-dimensional arrangement of atoms around the chiral center, denoted by the asterisk in the chemical structure. In the supercritical fluid chromatography process, the mobile phase typically consists of carbon dioxide modified with polar co-solvents like methanol and additives such as ammonia. The supercritical CO2 provides low viscosity and high diffusivity, allowing for rapid mass transfer and efficient separation. The chiral selector on the stationary phase forms transient diastereomeric complexes with the enantiomers; the (-)-enantiomer forms a slightly less stable complex or interacts differently compared to the (+)-enantiomer, leading to distinct retention times. In the specific embodiment described, the desired enantiomer elutes at approximately 6 minutes, while the unwanted isomer elutes later at around 11 minutes, allowing for clean fraction collection.
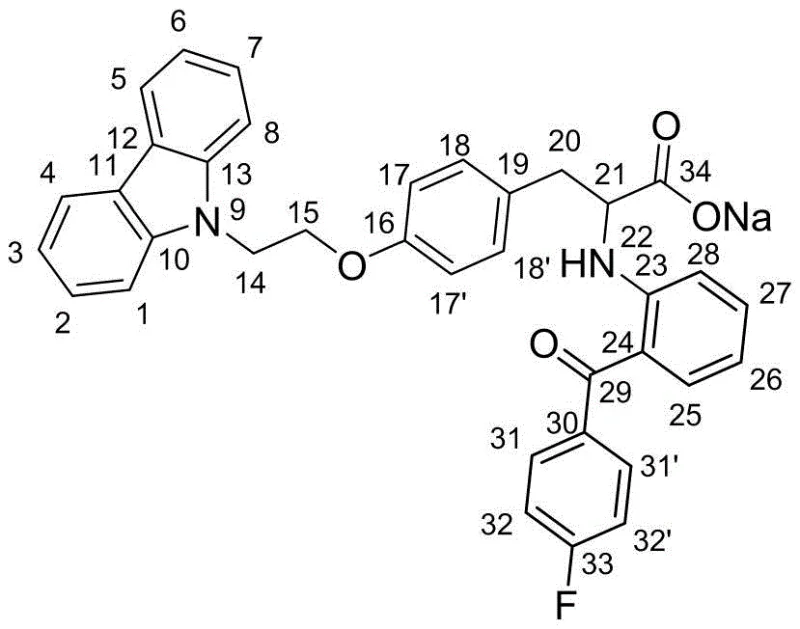
Following the isolation of the free acid, the process often involves conversion to a pharmaceutically acceptable salt to improve stability and solubility. The patent details the formation of the sodium salt, where the carboxylic acid group is neutralized by sodium hydroxide. Structural elucidation of this salt form, as evidenced by the comprehensive NMR data including COSY, HSQC, and HMBC correlations, confirms the integrity of the molecular framework post-salt formation. The preservation of the chiral center during this neutralization step is paramount, and the data confirms that the optical rotation remains consistent (e.g., [α]D = -119.5 for the sodium salt), indicating no racemization occurred. This robustness in the downstream processing is vital for maintaining the reducing lead time for high-purity pharmaceutical intermediates, as it eliminates the need for re-purification steps that often plague salt formation processes.
How to Synthesize (-)-2-[(2-(4-fluorobenzoyl)phenyl)amino]-3-[(4-(2-carbazolylethoxy)phenyl)]propionic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this high-value intermediate. The process begins with the dissolution of the racemic mixture in methanol, followed by injection into the SFC system. The operational parameters, such as flow rate (80 ml/min) and mobile phase composition (methanol/CO2 ratio of 4:1), are optimized to maximize resolution. After collecting the fractions containing the target enantiomer, the solvent is evaporated to yield the pure free acid. For the salt form, the acid is dissolved in tetrahydrofuran and treated with a methanolic solution of sodium hydroxide, followed by precipitation using isopropyl ether. This sequence ensures high recovery and purity, making it an attractive route for industrial adoption.
- Dissolve the racemic mixture of enantiomers in methanol and load onto a Chiralpak column within a supercritical chromatograph system.
- Elute the column using a methanol/carbon dioxide mixture followed by a methanol/carbon dioxide/ammonia mixture to separate the enantiomers based on retention time.
- Collect the specific elution peak corresponding to the desired (-)-enantiomer, evaporate the solvent, and optionally convert to the sodium salt using sodium hydroxide in tetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enantiomer-specific technology offers substantial strategic benefits beyond mere chemical efficacy. The shift from racemic to single-enantiomer production aligns with global trends towards greener and more efficient manufacturing practices. By utilizing SFC, the process significantly reduces the reliance on large volumes of hazardous organic solvents typically used in traditional liquid chromatography, thereby lowering waste disposal costs and environmental compliance burdens. This efficiency translates directly into a more resilient supply chain, as the simplified purification train reduces the risk of batch failures and ensures a consistent supply of the active ingredient.
- Cost Reduction in Manufacturing: The ability to isolate the active isomer with high efficiency means that the effective dose of the drug can potentially be lowered, reducing the total amount of API required per tablet. Furthermore, the elimination of the inactive isomer removes the cost associated with synthesizing, handling, and disposing of 50% of the reaction mass that would otherwise be waste in a racemic process. This qualitative improvement in atom economy and process efficiency drives down the overall cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of supercritical CO2 as a primary mobile phase component offers a distinct advantage in terms of raw material availability and cost stability. Unlike specialized chiral catalysts that may be subject to supply shortages or price volatility, CO2 is abundant and inexpensive. Additionally, the robustness of the SFC method allows for continuous processing capabilities, which enhances the reliability of supply for long-term commercial contracts and mitigates the risks associated with batch-to-batch variability.
- Scalability and Environmental Compliance: The described process is inherently scalable. Preparative SFC systems can be scaled up to handle kilogram to ton-scale quantities, facilitating the commercial scale-up of complex pharmaceutical intermediates. Moreover, the reduced solvent footprint and the ability to recycle CO2 contribute to a lower carbon footprint, helping pharmaceutical partners meet their sustainability goals and adhere to increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries regarding the production and application of this enantiomeric compound. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the compound's potential.
Q: What distinguishes the (-)-enantiomer from the racemic mixture in terms of biological activity?
A: The (-)-enantiomer demonstrates significantly superior activation of RXR/PPAR-α, RXR/PPAR-γ, and RXR/PPAR-δ heterodimers compared to the (+)-enantiomer and the racemate, leading to enhanced hypoglycemic effects in diabetic models.
Q: What is the optical purity achieved through the described separation process?
A: The process yields the (-)-enantiomer with an enantiomeric excess (ee%) of up to 100% for the free acid form and maintains high optical purity (ee% > 96%) even after conversion to the pharmaceutically acceptable sodium salt.
Q: Is the synthesis method scalable for commercial production?
A: Yes, the use of supercritical fluid chromatography (SFC) with carbon dioxide and methanol is a green chemistry approach that is highly amenable to scale-up, offering robust separation efficiency without the need for excessive organic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-2-[(2-(4-fluorobenzoyl)phenyl)amino]-3-[(4-(2-carbazolylethoxy)phenyl)]propionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the development of metabolic therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of this enantiomeric intermediate meets the highest international standards for pharmaceutical applications. Our commitment to quality assurance ensures that the biological activity observed in the lab is faithfully reproduced in the commercial product.
We invite you to collaborate with us to leverage this advanced chiral separation technology for your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your goal of bringing safer and more effective diabetes treatments to patients worldwide.
