Advanced Chiral Carbon Bond Catalysts for Efficient Pharmaceutical Intermediate Synthesis
The groundbreaking intellectual property disclosed in Chinese Patent CN115974792A introduces a paradigm-shifting approach to asymmetric catalysis through the engineering of novel chiral carbon bond catalysts. This technology leverages the unique electronic properties of carbon centers to create directional weak interactions, specifically sigma-holes, which drive chemical transformations with exceptional precision. Unlike traditional hydrogen bonding catalysts, these carbon bond catalysts offer dual activation sites and stronger directionality, addressing critical challenges in stereoselective synthesis. The core innovation lies in the structural modification of the catalyst backbone, replacing less efficient scaffolds with barbituric acid derivatives to enhance both solubility and catalytic turnover. 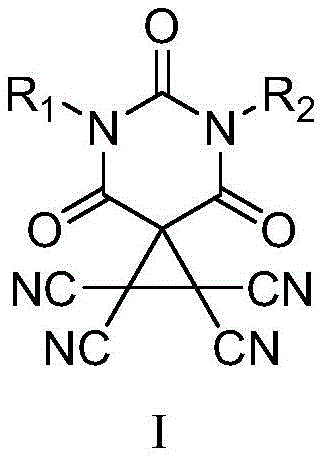 For procurement specialists and R&D directors seeking a reliable chiral organocatalyst supplier, this patent represents a significant leap forward in accessing high-purity specialty chemicals that were previously difficult to synthesize efficiently.
For procurement specialists and R&D directors seeking a reliable chiral organocatalyst supplier, this patent represents a significant leap forward in accessing high-purity specialty chemicals that were previously difficult to synthesize efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the field of carbon bond catalysis was heavily reliant on catalysts derived from Michaelis acid, which suffered from inherent physicochemical drawbacks that limited their industrial utility. These conventional catalysts often exhibited poor solubility in common organic solvents, leading to heterogeneous reaction mixtures that complicated process control and purification workflows. Furthermore, the catalytic efficiency of these earlier generations was frequently suboptimal, requiring higher catalyst loadings or extended reaction times to achieve acceptable conversion rates. The rigidity of the traditional scaffolds also restricted the ability to fine-tune steric and electronic properties, limiting the scope of substrates that could be effectively transformed. For supply chain heads managing the commercial scale-up of complex fine chemicals, these limitations translated into unpredictable batch consistency and increased operational costs due to inefficient material usage. Consequently, there was a pressing need in the pharmaceutical intermediates sector for a more robust and versatile catalytic platform.
The Novel Approach
The methodology outlined in CN115974792A overcomes these historical barriers by introducing a barbituric acid-based scaffold that fundamentally alters the solubility profile and reactivity of the catalyst. By incorporating chiral primary amines into the barbituric acid core, the inventors have created a library of catalysts that maintain high stereocontrol while dissolving readily in standard reaction media. This structural evolution allows for homogeneous catalysis, which ensures uniform heat and mass transfer during the reaction, thereby improving reproducibility on a large scale. The new approach also simplifies the synthetic route, utilizing readily available starting materials such as isocyanates and malonyl chloride, which are cost-effective and widely sourced. This strategic shift not only enhances the performance metrics of the catalyst but also aligns with green chemistry principles by reducing waste and energy consumption. For organizations focused on cost reduction in pharmaceutical intermediate manufacturing, this novel approach offers a clear pathway to more economical and sustainable production processes.
Mechanistic Insights into Carbon Bond Catalysis
The efficacy of these new catalysts stems from the sophisticated exploitation of sigma-hole interactions, where the electron-deficient region on the central carbon atom acts as a Lewis acid. This carbon center forms a directional non-covalent bond with electron-rich donors on the substrate, effectively lowering the activation energy for the transformation. In the context of the oxa-Pictet-Spengler reaction, the catalyst simultaneously activates both the electrophilic and nucleophilic components through these dual activation sites. 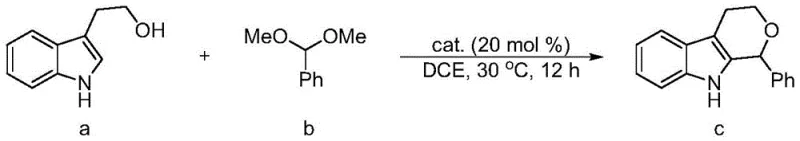 This bifunctional activation mode ensures that the reactants are held in a precise spatial arrangement, which is critical for achieving high enantioselectivity. The barbituric acid backbone provides the necessary acidity and structural rigidity to stabilize the transition state, while the chiral side chains impart the asymmetry required for enantioselective outcomes. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrate classes.
This bifunctional activation mode ensures that the reactants are held in a precise spatial arrangement, which is critical for achieving high enantioselectivity. The barbituric acid backbone provides the necessary acidity and structural rigidity to stabilize the transition state, while the chiral side chains impart the asymmetry required for enantioselective outcomes. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrate classes.
Furthermore, the impurity profile of reactions using these catalysts is significantly cleaner due to the high specificity of the carbon bond interaction. Unlike metal-based catalysts that can leave behind toxic residues requiring extensive downstream purification, these organocatalysts degrade into benign byproducts or can be easily removed via standard extraction techniques. The strong directionality of the carbon bond minimizes off-pathway reactions, thereby reducing the formation of regioisomers and other side products. This purity advantage is paramount for the production of high-purity organocatalysts intended for sensitive pharmaceutical applications where trace impurities can compromise drug safety. The ability to predict and control the impurity spectrum based on the catalyst structure provides a distinct competitive advantage in regulatory filings and quality assurance protocols.
How to Synthesize Chiral Carbon Bond Catalyst I Efficiently
The synthesis of these advanced catalysts follows a logical four-step sequence that is amenable to standard laboratory and pilot plant equipment. The process begins with the formation of a urea linkage, followed by cyclization to establish the barbituric acid core, bromination to activate the position for final functionalization, and concludes with the addition of tetracyanoethylene. Each step has been optimized to proceed under mild conditions, typically at room temperature or slightly below, minimizing thermal stress on the chiral centers. 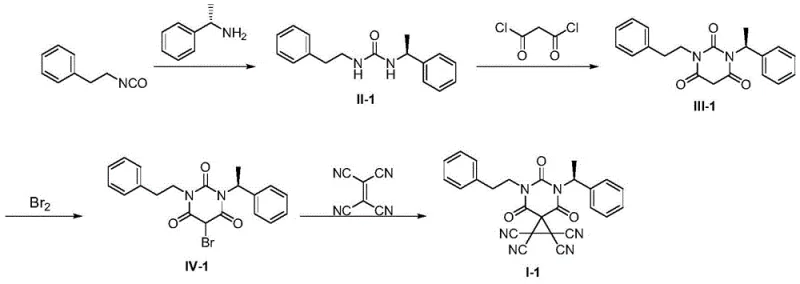 The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during implementation.
The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during implementation.
- React chiral primary amine with isocyanate or solid phosgene to form intermediate II.
- Perform condensation ring-closing reaction with malonyl chloride under argon protection to obtain intermediate III.
- Execute substitution reaction with Br2 in the presence of base at 0-5°C to generate intermediate IV.
- Complete cyclization by reacting intermediate IV with tetracyanoethylene in 1,4-dioxane to yield the final catalyst I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this technology addresses several critical pain points associated with the sourcing and production of specialized catalysts. The reliance on commodity chemicals like malonyl chloride and common chiral amines reduces dependency on exotic or single-source reagents, thereby enhancing supply chain resilience. The elimination of transition metals from the catalytic cycle removes the need for expensive scavenging resins and complex metal testing, streamlining the quality control workflow. For procurement managers, this translates into a more predictable cost structure and reduced risk of supply disruptions caused by geopolitical or logistical issues affecting rare metal markets. The overall process design prioritizes operational simplicity, which facilitates faster technology transfer from R&D to manufacturing units.
- Cost Reduction in Manufacturing: The synthetic route utilizes inexpensive and widely available raw materials, avoiding the high costs associated with precious metal ligands or complex chiral pools. By operating at ambient temperatures and pressures, the process significantly lowers energy consumption compared to high-temperature or high-pressure alternatives. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproduct. Additionally, the simplified workup procedures reduce the volume of solvents and consumables required for purification. These factors collectively contribute to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on product quality.
- Enhanced Supply Chain Reliability: The use of robust, non-sensitive reagents ensures that the supply chain is less vulnerable to fluctuations in the availability of specialized chemicals. Since the catalysts are organic in nature, they do not face the same regulatory scrutiny and import/export restrictions often applied to heavy metal compounds. This ease of logistics allows for more flexible inventory management and shorter lead times for high-purity organocatalysts. Manufacturers can maintain consistent production schedules knowing that the raw material base is stable and diversified. This reliability is crucial for meeting the just-in-time delivery requirements of modern pharmaceutical supply chains.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and more environmentally friendly. Waste streams are easier to treat, and the risk of environmental contamination is minimized, aligning with increasingly stringent global environmental regulations. The scalability of the process is supported by the use of common solvents like dichloromethane and acetonitrile, which are well-understood in large-scale operations. This facilitates the commercial scale-up of complex fine chemicals from gram-scale discovery to multi-ton production with minimal process re-engineering. Companies adopting this technology can demonstrate a strong commitment to sustainability while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbon bond catalyst technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. They serve to clarify the operational benefits and application scope for potential partners and licensees. Understanding these details is essential for making informed decisions about integrating this technology into existing production pipelines.
Q: What advantages do barbituric acid-based carbon bond catalysts offer over traditional Michaelis acid derivatives?
A: Barbituric acid derivatives exhibit significantly improved solubility and higher catalytic efficiency compared to traditional Michaelis acid-based catalysts, overcoming previous limitations in organic synthesis applications.
Q: What specific reactions can these novel carbon bond catalysts facilitate?
A: These catalysts are particularly effective in catalyzing the oxa-Pictet-Spengler reaction, demonstrating superior yields and reduced reaction times compared to existing benchmarks.
Q: Are the synthesis conditions for these catalysts suitable for large-scale production?
A: Yes, the synthesis utilizes mild reaction conditions, inexpensive chiral starting materials, and avoids toxic heavy metals, making it highly suitable for scalable commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Organocatalyst Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating cutting-edge academic research into commercially viable chemical solutions. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical capability to manufacture these complex carbon bond catalysts with stringent purity specifications. Our rigorous QC labs ensure that every batch meets the highest standards required for pharmaceutical and fine chemical applications. We understand the critical nature of catalyst performance in your synthesis and are committed to delivering products that drive your process efficiency forward.
We invite you to contact our technical procurement team to discuss how this technology can be tailored to your specific needs. Request a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this novel catalytic system. Our experts are ready to provide specific COA data and route feasibility assessments to support your project development. Partner with us to secure a sustainable and efficient supply of next-generation organocatalysts for your future projects.
