Advanced Dehydration Strategy for High-Purity 11-Alkene Steroid Intermediates Manufacturing
Advanced Dehydration Strategy for High-Purity 11-Alkene Steroid Intermediates Manufacturing
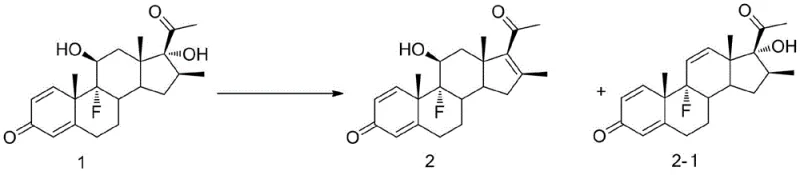
The pharmaceutical industry constantly seeks robust synthetic routes for complex steroid intermediates, particularly those featuring specific unsaturation patterns critical for biological activity. Patent CN110294782B introduces a transformative preparation method for 11-alkene steroid compounds that addresses long-standing challenges in regioselectivity and process efficiency. This innovation utilizes trimethyl halosilane as a specialized dehydrating agent to convert 11-hydroxy steroid substrates into the desired 11-alkene derivatives with exceptional precision. Unlike traditional methods that often struggle with isomer mixtures or require hazardous conditions, this novel approach operates under mild, aprotic conditions, delivering high molar yields and superior purity profiles essential for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 11-alkene steroids has been plagued by significant technical hurdles that compromise both yield and operational safety. Conventional literature describes the use of reagents such as N-bromosuccinimide (NBS) in pyridine or phosphorus oxychloride (POCl3) at elevated temperatures, often exceeding 90°C for extended periods. These harsh thermal conditions not only increase energy costs but also promote the formation of thermodynamically stable yet undesired 9,11-diene isomers, necessitating complex and costly chromatographic purification steps. Furthermore, existing methods utilizing fluorinating reagents like DAST, while effective, involve prohibitively expensive materials and generate hazardous byproducts, rendering them economically unviable for large-scale commercial manufacturing where cost efficiency and environmental compliance are paramount.
The Novel Approach
In stark contrast, the methodology disclosed in CN110294782B leverages the unique reactivity of trimethyl halosilanes—specifically trimethyl chlorosilane, bromosilane, or iodosilane—to achieve selective dehydration under remarkably mild conditions ranging from -10°C to 70°C. This process effectively overcomes steric hindrance at the 11-position, a common obstacle in steroid chemistry, by facilitating a smooth elimination reaction without degrading the sensitive steroid backbone. By simply adjusting the molar ratio of the silane reagent, manufacturers can precisely dictate the reaction pathway to favor the 11,12-alkene product over the 9,11-diene isomer, thereby drastically simplifying the purification workflow and enhancing overall process throughput.
Mechanistic Insights into Trimethyl Halosilane-Catalyzed Dehydration
The core of this technological breakthrough lies in the mechanistic interaction between the trimethyl halosilane and the 11-hydroxyl group of the steroid substrate. The silicon atom acts as a potent Lewis acid, coordinating with the hydroxyl oxygen to form a silyl ether intermediate in situ. This activation significantly lowers the energy barrier for the subsequent elimination of the leaving group, promoting the formation of the double bond at the 11,12-position. The choice of halogen on the silicon atom (Cl, Br, or I) influences the reaction kinetics, with trimethyliodosilane often providing superior results due to the better leaving group ability of iodide, ensuring rapid conversion even at lower temperatures which preserves the stereochemical integrity of adjacent chiral centers.
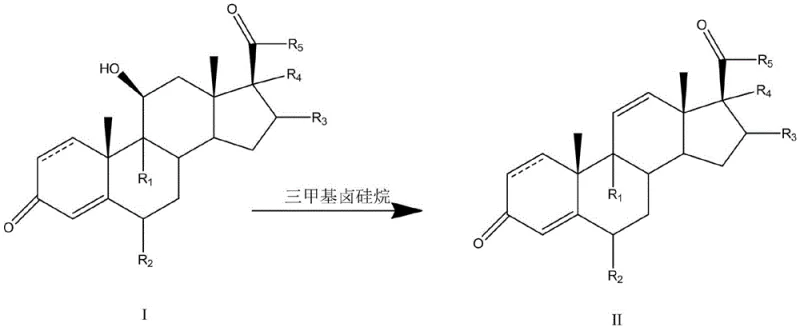
Crucially, the impurity profile is tightly controlled through stoichiometric regulation of the dehydrating agent. Experimental data indicates that maintaining a molar ratio of trimethyl halosilane to substrate between 1.0 and 2.5 selectively yields the 11,12-alkene isomer, whereas excess reagent can drive the reaction toward different elimination pathways. This tunability allows process chemists to minimize the generation of 9,11-diene impurities at the source rather than removing them post-reaction. The use of aprotic solvents such as acetonitrile or dichloromethane further stabilizes the transition state, preventing side reactions like hydrolysis or rearrangement, thus ensuring the final product meets the stringent purity specifications required for regulatory submission.
How to Synthesize 11-Alkene Steroid Compounds Efficiently
Implementing this synthesis route requires careful attention to reagent addition and temperature control to maximize the yield of the target 11-alkene isomer. The process begins by dissolving the 11-hydroxy steroid substrate in a dry aprotic solvent, followed by the slow addition of the trimethyl halosilane reagent under an inert atmosphere. Reaction progress is monitored via thin-layer chromatography (TLC) to determine the optimal endpoint, typically achieved within 1 to 3 hours depending on the specific substrate and temperature employed. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are outlined below.
- Dissolve the steroid substrate (Formula I) in an aprotic solvent such as acetonitrile or dichloromethane.
- Add trimethyl halosilane (e.g., trimethyliodosilane) as the dehydrating agent, controlling the molar ratio to direct regioselectivity.
- Stir the reaction mixture at mild temperatures (30-70°C), monitor progress via TLC, and quench with a reducing alkaline reagent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this trimethyl halosilane-mediated dehydration process represents a strategic opportunity to optimize manufacturing costs and secure a more reliable supply of critical steroid intermediates. By replacing expensive and hazardous fluorinating agents with commodity silane chemicals, the raw material cost structure is significantly improved. Furthermore, the mild reaction conditions reduce the demand for specialized high-temperature reactors and extensive cooling systems, lowering capital expenditure requirements for production facilities while simultaneously enhancing operator safety and reducing the risk of batch failures due to thermal runaway.
- Cost Reduction in Manufacturing: The substitution of high-cost fluorinating reagents like DAST with inexpensive trimethyl halosilanes results in substantial direct material savings. Additionally, the high selectivity of the reaction minimizes the loss of valuable starting materials to isomeric byproducts, effectively increasing the overall mass balance and reducing the cost per kilogram of the active pharmaceutical ingredient precursor without compromising quality standards.
- Enhanced Supply Chain Reliability: Trimethyl chlorosilane and related silanes are bulk commodities produced by major chemical suppliers globally, ensuring a stable and continuous supply chain free from the geopolitical or production bottlenecks often associated with specialty fluorine reagents. This availability allows for flexible sourcing strategies and reduces the lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional methods involving heavy metals or strong mineral acids, simplifying effluent treatment and reducing environmental compliance costs. The straightforward workup procedure, involving simple aqueous quenching and extraction, facilitates easy scale-up from 100 kgs to 100 MT annual commercial production, ensuring that the technology remains robust and efficient regardless of the production volume required.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel dehydration technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to legacy processes in terms of efficiency, safety, and product quality.
Q: How does this method control the formation of 9,11-diene isomers?
A: By precisely controlling the molar ratio of trimethyl halosilane to the substrate. A ratio between 1.0 and 2.5 favors the 11,12-alkene product, while higher ratios may shift selectivity, allowing for targeted synthesis without complex chromatographic purification.
Q: What are the advantages of using trimethyl halosilane over traditional fluorinating reagents?
A: Trimethyl halosilanes are significantly more cost-effective and easier to handle than expensive fluorinating agents like DAST. They operate under milder conditions, reducing energy consumption and safety risks associated with high-temperature dehydration processes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes readily available reagents, avoids high-risk reactions, and employs simple workup procedures involving standard extraction and recrystallization, making it highly amenable to commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-Alkene Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of steroid drug development. Our team of expert process chemists has thoroughly evaluated the trimethyl halosilane dehydration technology and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this innovation to your supply chain. We are committed to delivering high-purity 11-alkene steroid intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art manufacturing facilities designed for complex organic synthesis.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis method for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your production efficiency and reduce your overall time to market.
