Advanced Synthesis of LED-Matched Photoinitiators for High-Efficiency UV Curing Applications
Advanced Synthesis of LED-Matched Photoinitiators for High-Efficiency UV Curing Applications
The rapid industrial transition towards energy-efficient LED curing systems has exposed a critical limitation in traditional photopolymerization technologies, specifically regarding spectral matching. As detailed in patent CN111635301B, the prevailing market standard for LED light sources operates within an emission wavelength range exceeding 365nm, whereas conventional Type I photoinitiators like the widely used 184 initiator exhibit a maximum absorption peak at merely 333nm. This spectral mismatch results in suboptimal energy utilization and significantly reduced double bond conversion efficiency during the curing process. To address this technological bottleneck, the patent introduces a novel class of photoinitiators synthesized through the condensation reaction of hydroxycyclohexylacetophenone derivatives containing double alpha-hydrogen carbonyl groups with various aldehyde compounds. This strategic structural modification extends the conjugated pi-system of the molecule, effectively red-shifting the absorption maximum to align perfectly with the 365nm to 770nm output of modern LED arrays, thereby unlocking superior curing performance and broader industrial applicability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoinitiators, particularly the commercially ubiquitous 184 type (1-hydroxycyclohexyl phenyl ketone), were originally designed for medium-pressure mercury lamp systems which emit strongly in the UV-C and UV-B regions. When deployed in modern LED curing units that predominantly emit in the UV-A and visible regions (above 365nm), these legacy molecules suffer from poor molar extinction coefficients at the operating wavelength. Consequently, formulators are forced to increase the loading levels of the initiator to compensate for the low absorption, which can lead to issues with yellowing, odor, and migration of unreacted species in the final cured film. Furthermore, the inefficiency in photon absorption translates directly to slower line speeds in coating and printing operations, creating a bottleneck for high-throughput manufacturing environments that demand rapid tack-free times. The reliance on these mismatched initiators essentially forces a compromise between curing speed and final material properties, limiting the full potential of LED technology in sectors ranging from wood coatings to electronic encapsulation.
The Novel Approach
The innovative methodology presented in the patent overcomes these spectral limitations by chemically engineering the chromophore of the initiator itself. By reacting a hydroxycyclohexylacetophenone core with aromatic or heteroaromatic aldehydes via a base-catalyzed condensation, the process introduces an alpha,beta-unsaturated ketone moiety into the molecular structure. This extension of conjugation is the key mechanistic driver that shifts the absorption bathochromically into the desired LED range. As illustrated in the general reaction scheme below, this approach is highly modular, allowing for the incorporation of diverse substituents to fine-tune both solubility and reactivity.
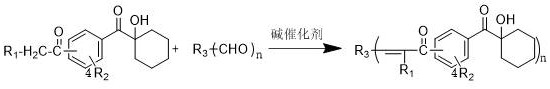
The resulting compounds, exemplified by structures such as those shown in Formula I through Formula V, demonstrate maximum absorption wavelengths ranging from 356nm up to 450nm, ensuring robust overlap with commercial LED emission profiles. This structural versatility means that a single synthetic platform can generate a library of initiators tailored for specific LED wavelengths, whether it be 365nm, 385nm, 395nm, or 405nm, providing a level of customization that simple blending of traditional initiators cannot achieve. Moreover, the synthesis avoids the use of expensive transition metal catalysts or harsh conditions, relying instead on accessible organic building blocks and mild alkaline promoters.
Mechanistic Insights into Base-Catalyzed Aldol Condensation
The core chemical transformation driving this innovation is the Claisen-Schmidt condensation, a specific type of aldol condensation between a ketone possessing alpha-hydrogens and an aldehyde lacking alpha-hydrogens. In the context of this patent, the hydroxycyclohexylacetophenone derivative serves as the nucleophilic donor after deprotonation at the alpha-carbon by a base catalyst. The patent specifies a wide array of suitable bases, including inorganic hydroxides like sodium hydroxide and potassium hydroxide, as well as stronger organic bases such as lithium diisopropylamide (LDA) and lithium hexamethyldisilazide (LiHMDS). The choice of base influences the reaction kinetics and the equilibrium position; for instance, using alkoxides like potassium tert-butoxide in alcoholic solvents can enhance the solubility of the enolate intermediate, facilitating the attack on the electrophilic carbonyl carbon of the aldehyde. Following the addition step, the intermediate beta-hydroxy ketone undergoes spontaneous or catalyzed dehydration to yield the thermodynamically stable alpha,beta-unsaturated ketone product, which constitutes the active photoinitiating species.
From an impurity control perspective, this mechanism offers distinct advantages for high-purity manufacturing. The reaction is highly selective for the formation of the trans-alkene isomer due to steric factors in the transition state, minimizing the formation of cis-isomers which might have different photochemical properties. Furthermore, the patent highlights that the products often possess low solubility in the reaction medium at lower temperatures, leading to spontaneous crystallization upon cooling. This phenomenon acts as an inherent purification step, as the crystallizing product excludes soluble impurities and unreacted starting materials from the crystal lattice. Subsequent recrystallization from solvents like ethanol or methanol further refines the purity profile, ensuring that the final photoinitiator meets the stringent specifications required for sensitive applications such as optical fiber coatings or food-contact packaging materials where extractables and leachables are critical concerns.
How to Synthesize Novel Photoinitiators Efficiently
The preparation of these advanced photoinitiators is designed for operational simplicity and robustness, making it highly suitable for transfer from laboratory bench to pilot plant and eventually to full commercial production. The process begins with the preparation of the key ketone intermediate, which may itself require functionalization steps such as methylation or halogenation depending on the desired final substitution pattern. Once the appropriate ketone and aldehyde partners are selected based on the target absorption wavelength, they are dissolved in a polar protic or aprotic solvent. The reaction is initiated by the addition of the catalyst, and the mixture is maintained at a moderate temperature, typically between 20°C and 80°C, for a duration of 1 to 6 hours. The progress of the reaction can be monitored via TLC or HPLC, looking for the disappearance of the aldehyde starting material. Upon completion, the workup is remarkably straightforward, often requiring only filtration of the precipitated solid followed by washing and drying, although liquid-liquid extraction is also described for certain variants.
- Prepare the key intermediate, hydroxycyclohexylacetophenone containing double alpha-H carbonyl groups, through methylation and oxidation sequences.
- Dissolve the ketone intermediate and the selected aldehyde compound in a suitable solvent such as ethanol or methanol.
- Add an alkaline catalyst like NaOH or t-BuOK to adjust pH to 8-12 and react at 20-80°C for 1-6 hours.
- Purify the resulting solid product through filtration, washing, recrystallization, and vacuum drying to obtain the final photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a compelling value proposition centered around cost stability and supply security. Unlike complex multi-step syntheses that rely on scarce precious metal catalysts or hazardous reagents, this condensation pathway utilizes commodity chemicals that are widely available in the global market. The ketone and aldehyde precursors are produced at massive scales for other industries, insulating the supply chain from the volatility often associated with specialty fine chemical intermediates. This abundance of raw materials ensures that production schedules can be maintained without the risk of bottlenecks caused by supplier shortages, a critical factor for manufacturers operating on just-in-time inventory models. Furthermore, the simplicity of the reaction conditions eliminates the need for specialized high-pressure or cryogenic equipment, allowing production to be scheduled on general-purpose multipurpose reactors without requiring dedicated capital investment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive catalytic systems and the reduction of unit operations. Traditional methods for modifying photoinitiator absorption often involve cross-coupling reactions requiring palladium or nickel catalysts, which not only add significant material cost but also necessitate costly downstream purification steps to remove trace metal residues to ppm levels. In contrast, the alkaline condensation described here uses inexpensive bases like NaOH or KOH, and the product isolation via crystallization avoids the need for column chromatography or complex distillation. This streamlined workflow drastically reduces solvent consumption, energy usage for heating and cooling, and labor hours per kilogram of output, translating into a substantially lower cost of goods sold (COGS) while maintaining high margins.
- Enhanced Supply Chain Reliability: The robustness of the chemistry contributes directly to supply chain resilience. The reaction tolerates a wide range of temperatures and pH levels, meaning that minor fluctuations in process parameters do not lead to batch failures or off-spec material. This forgiveness in the manufacturing process reduces waste and ensures consistent output quality, which is vital for maintaining long-term contracts with downstream formulators. Additionally, because the synthesis does not generate toxic heavy metal waste streams, the environmental compliance burden is significantly lighter, reducing the risk of regulatory shutdowns or permitting delays that could disrupt supply continuity. The ability to source raw materials from multiple geographic regions further diversifies the supply base, mitigating risks associated with geopolitical instability or regional logistics disruptions.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the homogeneous nature of the reaction mixture and the exothermic profile which is manageable under standard cooling capacities. The use of common solvents like ethanol and methanol simplifies solvent recovery and recycling loops, aligning with green chemistry principles and corporate sustainability goals. The absence of halogenated solvents in many embodiments reduces the generation of hazardous waste, lowering disposal costs and environmental impact. This eco-friendly profile is increasingly becoming a prerequisite for suppliers entering the supply chains of major multinational corporations who are aggressively pursuing carbon neutrality and waste reduction targets, thereby opening up premium market segments for manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel photoinitiator technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of integrating these materials into existing UV curing formulations. Understanding these details is crucial for R&D teams assessing compatibility and for procurement teams evaluating the total cost of ownership.
Q: Why is this novel photoinitiator superior to traditional 184 initiators for LED applications?
A: Traditional 184 photoinitiators have a maximum absorption wavelength of 333nm, which mismatches the 365nm+ emission of modern LED sources. This novel initiator, synthesized via condensation, extends the conjugation system, shifting the maximum absorption wavelength to above 356nm (up to 450nm in some embodiments), ensuring efficient energy capture and higher double bond conversion rates under LED irradiation.
Q: What are the scalability advantages of this condensation method?
A: The synthesis relies on a straightforward one-step condensation reaction between readily available ketones and aldehydes. It operates under mild temperatures (20-80°C) and uses common alkaline catalysts. The product often precipitates directly from the reaction mixture upon cooling, simplifying isolation to basic filtration and recrystallization, which significantly reduces processing complexity and facilitates large-scale manufacturing.
Q: Can the absorption properties be tuned for specific LED wavelengths?
A: Yes, the patent describes a versatile general formula where substituents on the benzene ring and the nature of the aldehyde component can be varied. By selecting different aldehydes (e.g., benzaldehyde, pyrrole carbaldehydes) and modifying the ketone core (e.g., introducing methoxy or cyano groups), the absorption maximum can be precisely tuned across the 365nm to 450nm range to match specific LED light sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Novel Photoinitiator Supplier
As the demand for high-performance LED-curable materials continues to surge across the coatings, inks, and electronics sectors, having a manufacturing partner with deep expertise in photo-active chemistry is essential. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-quality photoinitiators. Our state-of-the-art facilities are equipped to handle the specific requirements of base-catalyzed condensations, ensuring stringent purity specifications are met through our rigorous QC labs which utilize advanced analytical techniques to verify absorption spectra and impurity profiles. We understand that in the fast-paced world of UV curing, reliability is just as important as performance, and our dedicated technical team is ready to support your transition to next-generation LED systems.
We invite you to engage with our technical procurement team to discuss how this novel photoinitiator technology can optimize your specific application requirements. Whether you need a Customized Cost-Saving Analysis for switching from traditional initiators or require specific COA data and route feasibility assessments for new derivative structures, we are prepared to provide the data-driven insights you need. Contact us today to request samples and discover how our advanced synthesis capabilities can drive efficiency and performance in your UV curing operations.
