Advanced Long-Wavelength Carbonyl Alcohol Photoinitiators for Industrial LED Curing Applications
Advanced Long-Wavelength Carbonyl Alcohol Photoinitiators for Industrial LED Curing Applications
The global shift towards environmentally sustainable manufacturing processes has necessitated the rapid adoption of LED curing technologies, replacing traditional mercury vapor lamps in accordance with the Minamata Convention. Patent CN111574352A introduces a groundbreaking class of long-wavelength carbonyl alcohol photoinitiators specifically engineered to bridge the spectral gap between conventional chemistry and modern LED hardware. Unlike legacy initiators that struggle to absorb energy efficiently from LED sources, these novel compounds exhibit maximum absorption wavelengths exceeding 365nm, perfectly aligning with the emission profiles of industrial LED arrays. This technological leap ensures that photo-curing systems can achieve rapid cross-linking and high conversion rates without the environmental hazards associated with mercury. For R&D teams and procurement strategists, this represents a critical opportunity to future-proof supply chains while enhancing the performance of coatings, inks, and electronic materials.
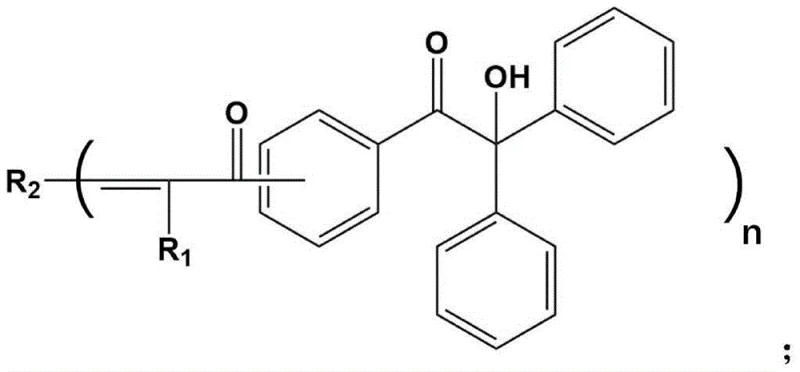
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photoinitiators, such as the widely used 1173 (2-hydroxy-2-methyl-1-phenyl-1-propanone), have served the industry well for decades but face obsolescence in the era of LED curing. The fundamental flaw lies in their absorption spectrum; 1173 exhibits a maximum absorption peak at approximately 331nm, which falls significantly short of the 365nm to 405nm range emitted by standard high-power LED sources. This spectral mismatch results in poor energy utilization, requiring higher initiator loadings or longer exposure times to achieve adequate curing, which can compromise the mechanical properties of the final polymer matrix. Furthermore, the reliance on mercury lamps to access shorter UV wavelengths is becoming legally and logistically untenable due to strict international environmental regulations banning mercury use. Consequently, manufacturers sticking to conventional initiators face increasing compliance risks and inefficiencies in their production lines.
The Novel Approach
The innovation disclosed in CN111574352A overcomes these spectral barriers by chemically modifying the core structure of carbonyl alcohol initiators to extend their conjugation systems. By introducing specific substituents and extending the molecular framework through condensation with aldehydes containing alpha-hydrogens, the new initiators shift their absorption maxima well into the long-wavelength UV region, reaching beyond 365nm and even up to 405nm in some embodiments. This structural adjustment allows the molecules to effectively harvest photons from LED sources, generating free radicals with high quantum efficiency. Experimental data from the patent demonstrates that these new initiators can achieve double bond conversion rates of up to 80-90% within 600 seconds under LED irradiation, significantly outperforming the roughly 50% conversion seen with traditional 1173 under similar conditions. This approach not only solves the technical mismatch but also opens the door for faster line speeds and lower energy consumption in industrial settings.
Mechanistic Insights into Base-Catalyzed Condensation and Spectral Tuning
The core synthetic strategy relies on a robust base-catalyzed condensation reaction, typically an aldol-type condensation, between a keto-functionalized 2-hydroxy-2-methyl-1-phenyl-1-propanone derivative and various aldehydes. The reaction mechanism involves the deprotonation of the alpha-carbon adjacent to the ketone group by a base such as sodium hydroxide, potassium tert-butoxide, or lithium diisopropylamide, generating a nucleophilic enolate species. This enolate then attacks the carbonyl carbon of the aldehyde, followed by dehydration to form an alpha,beta-unsaturated ketone system. This extension of the pi-conjugated system is the key electronic feature that lowers the energy gap between the HOMO and LUMO, thereby red-shifting the absorption spectrum to match LED wavelengths. The versatility of this mechanism allows for the incorporation of diverse functional groups, such as nitro, cyano, or halogen substituents, further fine-tuning the photophysical properties for specific application requirements.
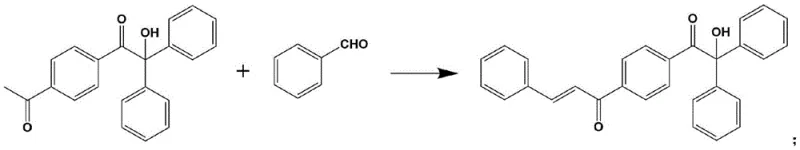
Impurity control in this synthesis is managed through precise pH regulation and temperature control during the reaction and subsequent crystallization steps. The patent specifies maintaining a pH between 8 and 12 and temperatures ranging from 0°C to 100°C, conditions that favor the formation of the desired trans-isomer while minimizing side reactions like polymerization of the aldehyde or over-condensation. Post-reaction purification is achieved through straightforward filtration and recrystallization, leveraging the low solubility of the final product in the reaction medium as it cools. This crystallization-driven purification is highly effective at removing unreacted starting materials and inorganic salts, ensuring a high-purity final product essential for preventing yellowing or stability issues in sensitive coating applications. The ability to obtain high-purity solids directly from the reaction mixture simplifies the downstream processing significantly compared to methods requiring column chromatography.
How to Synthesize Long-Wavelength Carbonyl Alcohol Photoinitiator Efficiently
The synthesis protocol outlined in the patent offers a streamlined pathway suitable for both laboratory optimization and large-scale manufacturing. The process begins with the preparation of the requisite keto-functionalized precursor, which can be achieved via Friedel-Crafts acylation or Grignard addition depending on the desired substitution pattern. Following this, the key condensation step is performed in common alcoholic solvents like ethanol or methanol, utilizing inexpensive inorganic or organic bases as catalysts. The detailed standardized synthesis steps for replicating this high-efficiency photoinitiator are provided in the guide below.
- Prepare the keto-functionalized 2-hydroxy-2-methyl-1-phenyl-1-propanone derivative via Friedel-Crafts acylation or Grignard reaction.
- Dissolve the derivative and an alpha-H containing aldehyde in a solvent such as ethanol or methanol.
- Add a basic catalyst like NaOH or potassium tert-butoxide, adjust pH to 8-12, and react at 0-100°C to form the final photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new photoinitiator technology offers substantial strategic advantages beyond mere technical performance. The synthesis route utilizes readily available commodity chemicals such as benzaldehyde derivatives and acetophenone precursors, mitigating the risk of supply bottlenecks often associated with exotic or highly specialized reagents. Furthermore, the reaction conditions are mild and do not require high-pressure equipment or cryogenic temperatures, allowing for production in standard glass-lined or stainless steel reactors found in most fine chemical facilities. This compatibility with existing infrastructure drastically reduces the capital expenditure required for technology transfer and scale-up, enabling a faster time-to-market for new LED-curable formulations.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of simple aqueous workups significantly lower the raw material and waste treatment costs. Since the purification relies on crystallization rather than complex distillation or chromatography, the energy consumption per kilogram of product is markedly reduced. Additionally, the higher curing efficiency means that formulators can potentially reduce the loading level of the photoinitiator in the final product while maintaining performance, leading to direct savings in bill-of-materials costs for the end-user.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining the reliability of downstream coating and ink production schedules. By diversifying the supply base with a domestically producible alternative to imported specialty initiators, companies can insulate themselves from geopolitical trade disruptions and logistics delays. The simplicity of the process also means that multiple contract manufacturing organizations (CMOs) can easily adopt the technology, creating a redundant and resilient supply network.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions that are easier to treat than organic solvent-heavy waste streams from other synthetic routes. This aligns perfectly with increasingly stringent environmental regulations, reducing the regulatory burden and potential fines associated with hazardous waste disposal. The scalability is proven by the straightforward exothermic profile of the condensation reaction, which can be safely managed in large reactors through controlled dosing, ensuring safe commercial scale-up of complex photoinitiators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this long-wavelength photoinitiator technology. These answers are derived directly from the experimental data and process descriptions found in the patent documentation to ensure accuracy and relevance for decision-makers.
Q: Why is this new photoinitiator better than traditional 1173 for LED curing?
A: Traditional 1173 has a maximum absorption at 331nm, which mismatches modern LED sources emitting above 365nm. This new initiator absorbs >365nm, ensuring efficient radical generation and higher double bond conversion rates under LED light.
Q: What are the key reaction conditions for synthesizing this photoinitiator?
A: The synthesis involves a condensation reaction under mild alkaline conditions (pH 8-12) at temperatures ranging from 0°C to 100°C, using common solvents like ethanol or methanol, which facilitates easy scale-up.
Q: Can this photoinitiator be used in mercury-free curing systems?
A: Yes, it is specifically designed to replace mercury lamp systems by matching the emission spectrum of environmentally friendly LED light sources, aligning with the Minamata Convention regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Long-Wavelength Photoinitiator Supplier
As the demand for LED-curable materials accelerates across the coatings, electronics, and adhesive sectors, having a partner with deep technical expertise in photochemistry is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to LED curing is supported by a secure and high-volume supply. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of photoinitiator meets the exacting standards required for high-performance industrial applications, minimizing the risk of cure failures or product defects.
We invite you to collaborate with our technical team to evaluate how this novel initiator can optimize your specific formulation needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your production volume. By partnering with us, you gain access to cutting-edge chemical solutions that drive both sustainability and profitability in your manufacturing operations.
