Scalable Manufacturing of GP IIb/IIIa Antagonist Intermediates via Novel Crystallization
Scalable Manufacturing of GP IIb/IIIa Antagonist Intermediates via Novel Crystallization
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex cardiovascular therapeutics, particularly those targeting platelet aggregation. Patent CN1351600A discloses a highly efficient process for preparing [S-(R*,S*)]-beta-[[[1-[1-oxo-3-(4-piperidinyl)propyl]-3-piperidinyl]carbonyl]amino]-3-pyridinepropanoic acid and its derivatives, which function as potent antagonists of platelet fibrinogen receptors (GP IIb/IIIa). These compounds are critical for treating thrombotic disorders such as vascular restenosis following angioplasty, unstable angina, and myocardial infarction. The technical breakthrough lies not merely in the molecular structure but in the strategic shift from amorphous salt formation to a stable crystalline free base, addressing long-standing purification challenges in peptide-mimetic synthesis. This innovation offers a reliable pharmaceutical intermediate supplier pathway that aligns with modern GMP standards for high-purity active ingredients.
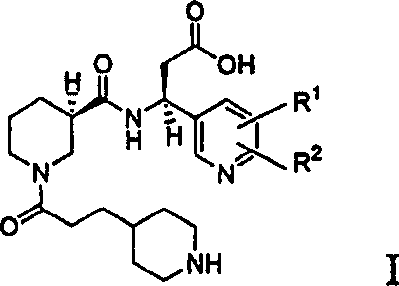
The structural complexity of Formula I, where R1 and R2 are independently selected from hydrogen, lower alkyl, and halogen, demands precise stereochemical control. Previous methodologies often struggled with the final isolation steps, resulting in hygroscopic salts that complicated downstream formulation. By establishing a route that culminates in a well-defined crystalline form, this patent provides a foundation for cost reduction in pharmaceutical intermediates manufacturing, ensuring that the supply chain remains resilient against quality fluctuations. The ability to produce these molecules with stringent purity specifications is paramount for regulatory approval and patient safety in the cardiovascular sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, specifically the methods disclosed in WO97/41102, relied heavily on the use of tert-butoxycarbonyl (Boc) protecting groups and subsequent removal using strong acids like trifluoroacetic acid (TFA). This approach inevitably led to the isolation of the final product as a bis-trifluoroacetate salt, which was described as a white amorphous solid. Amorphous solids present significant challenges in commercial scale-up of complex pharmaceutical intermediates, including variable solubility, difficulty in filtration, and potential stability issues during storage. Furthermore, the removal of residual TFA and the management of fluorinated waste streams add substantial environmental and processing burdens. The reliance on multiple protection and deprotection cycles with harsh reagents increases the risk of racemization and lowers the overall yield, making the process less attractive for large-scale production where efficiency is key.
The Novel Approach
The methodology presented in CN1351600A introduces a paradigm shift by utilizing benzyloxycarbonyl (Cbz) protection strategies combined with catalytic hydrogenation for the final deprotection step. This allows for the isolation of the compound as a free base rather than a salt. Crucially, the process incorporates a novel crystallization technique where the free base is dissolved in an organic solvent like n-butanol and treated with an organic amine base, such as tert-butylamine, to adjust the pH to a specific range (approximately 7-11). This induces the precipitation of the compound as irregular needle-like granular crystals, a form that is far superior for handling and formulation compared to the amorphous prior art. This transition represents a significant advancement in reducing lead time for high-purity pharmaceutical intermediates, as it simplifies the workup and eliminates the need for complex salt exchange procedures.
Mechanistic Insights into Chiral Resolution and Catalytic Hydrogenation
The core of this synthesis relies on the precise installation of stereocenters through classical resolution rather than expensive asymmetric catalysis. The patent details the preparation of key chiral intermediates, such as (S)-3-amino-3-(3-pyridyl)propionic acid methyl ester and (R)-(-)-3-piperidinecarboxylic acid esters, using (+)-tartaric acid. In this mechanism, the racemic amine or ester forms a diastereomeric salt with the chiral acid. Due to the differences in solubility between the diastereomers in specific solvent systems (e.g., ethanol/water or isopropanol/water), one enantiomer preferentially crystallizes out. For instance, the resolution of racemic 3-amino-3-(3-pyridyl)propionic acid methyl ester dihydrochloride involves liberating the free base, reacting it with (+)-tartaric acid, and carefully controlling the cooling profile to maximize the diastereomeric excess (de), which can reach levels greater than 98%. This approach ensures that the final API intermediate possesses the requisite optical purity without the need for chiral HPLC separation, which is often cost-prohibitive at scale.
Furthermore, the process leverages catalytic hydrogenation for both ring saturation and protecting group removal. The initial step involves the hydrogenation of 3-(4-pyridine)acrylic acid to 3-(4-piperidinyl)propionic acid using a rhodium catalyst on alumina (Rh/Al2O3) in an alkaline medium. The choice of pH (7-13) and temperature (80-95°C) is critical to prevent over-reduction or catalyst poisoning. Later in the sequence, the removal of the Cbz protecting group is achieved using palladium on carbon (Pd/C) under mild pressure (2-3 bar). This mechanistic choice is superior to acidolysis because it generates benign byproducts (toluene and CO2) instead of corrosive acids. The combination of resolution and hydrogenation creates a robust impurity profile, where side products are either volatile or easily removed by aqueous washes, thereby enhancing the overall purity of the high-purity pharmaceutical intermediates.
How to Synthesize [S-(R*,S*)]-beta-[[[1-[1-oxo-3-(4-piperidinyl)propyl]-3-piperidinyl]carbonyl]amino]-3-pyridinepropanoic acid Efficiently
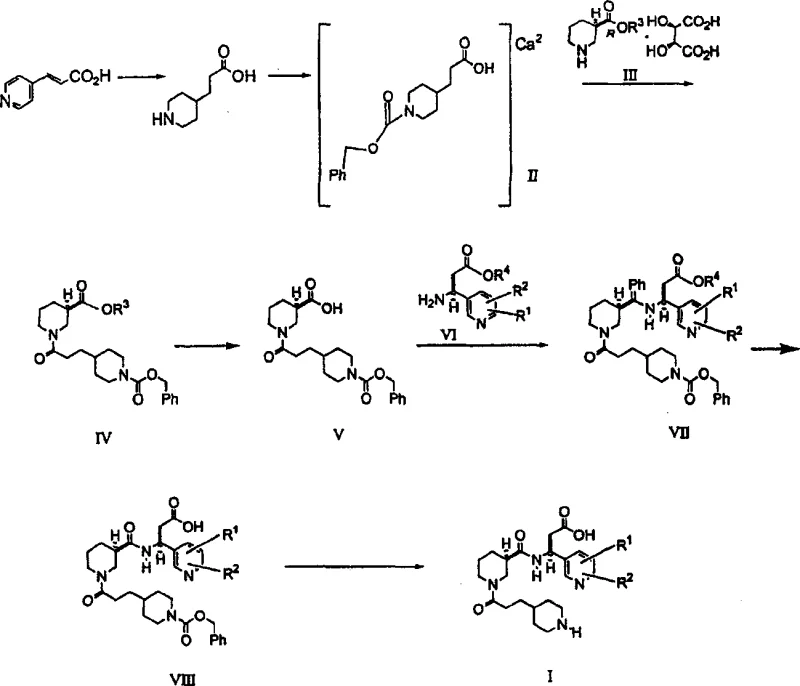
The synthesis outlined in the patent provides a clear roadmap for producing this complex molecule, starting from readily available commodity chemicals like 3-(4-pyridine)acrylic acid. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates. Key to the success of this route is the formation of the calcium salt of the protected piperidine propionic acid (Formula II), which serves as a stable handle for the subsequent peptide coupling reactions. The detailed standardized synthesis steps involve precise control of stoichiometry, particularly in the coupling steps using DCC and HOBt, to minimize the formation of urea byproducts.
- Catalytic hydrogenation of 3-(4-pyridine)acrylic acid using Rh/Al2O3 in alkaline solvent to form 3-(4-piperidinyl)propionic acid.
- Protection with benzyl chloroformate under basic calcium salt conditions to form the calcium salt intermediate.
- Coupling with chiral piperidine carboxylic acid tartrate and subsequent hydrolysis and hydrogenation to yield the final crystalline free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel process offers tangible benefits that extend beyond simple yield improvements. The elimination of trifluoroacetic acid (TFA) from the workflow is a major logistical win, as it removes the requirement for specialized corrosion-resistant equipment and the costly disposal of fluorinated waste. This directly translates to cost reduction in pharmaceutical intermediates manufacturing by lowering both capital expenditure on infrastructure and operational expenditure on waste management. Additionally, the use of classical resolution with tartaric acid, a cheap and abundant chiral pool material, avoids the volatility and high cost associated with proprietary chiral ligands or enzymes, stabilizing the raw material cost structure.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by replacing expensive and hazardous deprotection reagents with catalytic hydrogenation. The avoidance of TFA salts means that the final product does not require extensive ion-exchange chromatography or complex salt screening to achieve the desired solid-state properties. Furthermore, the high yields reported in the examples (e.g., 92% yield in the coupling step and 98% ee in resolution) indicate a highly efficient use of raw materials, reducing the cost of goods sold (COGS) per kilogram of active ingredient.
- Enhanced Supply Chain Reliability: By relying on commodity reagents such as calcium hydroxide, benzyl chloroformate, and palladium on carbon, the supply chain becomes less vulnerable to shortages of exotic chemicals. The robustness of the crystallization step ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance and avoiding production delays. The ability to produce a stable crystalline free base also extends the shelf-life of the intermediate, allowing for more flexible inventory management and reducing the risk of spoilage during transit.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the use of standard unit operations like filtration, distillation, and hydrogenation in stainless steel reactors. The environmental footprint is drastically reduced by eliminating fluorinated solvents and acids, aligning with green chemistry principles. The aqueous workups and the use of recyclable solvents like ethyl acetate and THF simplify the recovery and reuse of materials, further enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer.
Q: What is the primary advantage of the new crystallization method over the prior art?
A: The new process yields a stable crystalline free base (Formula Ia) rather than the amorphous bis-trifluoroacetate salt produced by previous methods, significantly improving purity and handling characteristics.
Q: How is chirality controlled in this synthesis?
A: Chirality is established through classical resolution of racemic intermediates using (+)-tartaric acid, which provides high enantiomeric excess (de > 98%) in an economically viable manner.
Q: Does this process avoid the use of trifluoroacetic acid (TFA)?
A: Yes, unlike the method disclosed in WO97/41102 which requires TFA for deprotection resulting in difficult-to-remove salts, this process utilizes Cbz protection and catalytic hydrogenation, eliminating TFA usage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable [S-(R*,S*)]-beta-[[[1-[1-oxo-3-(4-piperidinyl)propyl]-3-piperidinyl]carbonyl]amino]-3-pyridinepropanoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving cardiovascular medications. Our technical team has extensively analyzed the process described in CN1351600A and is fully equipped to execute this chemistry with precision. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facility is outfitted with state-of-the-art hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including chiral purity and residual solvent analysis, to guarantee that every batch meets global regulatory standards.
We invite you to collaborate with us to optimize your supply chain for this critical therapeutic area. By leveraging our expertise in chiral resolution and peptide coupling, we can help you achieve significant efficiencies in your manufacturing timeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your project requirements. Let us be your partner in delivering high-performance pharmaceutical solutions to the market.
